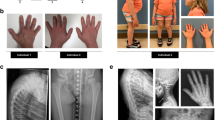
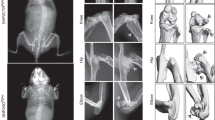
Abstract
Osteopetrosis includes a group of inherited diseases in which inadequate bone resorption is caused by osteoclast dysfunction. Although molecular defects have been described for many animal models of osteopetrosis, the gene responsible for most cases of the severe human form of the disease (infantile malignant osteopetrosis) is unknown. Infantile malignant autosomal recessive osteopetrosis (MIM 259700) is a severe bone disease with a fatal outcome, generally within the first decade of life. Osteoclasts are present in normal or elevated numbers in individuals affected by autosomal recessive osteopetrosis1, suggesting that the defect is not in osteoclast differentiation, but in a gene involved in the functional capacity of mature osteoclasts. Some of the mouse mutants have a decreased number of osteoclasts, which suggests that the defect directly interferes with osteoclast differentiation2,3. In other mutants, it is the function of the osteoclast that seems to be affected, as they show normal or elevated numbers of non-functioning osteoclasts2,4,5,6. Here we show that TCIRG1, encoding the osteoclast-specific 116-kD subunit of the vacuolar proton pump, is mutated in five of nine patients with a diagnosis of infantile malignant osteopetrosis. Our data indicate that mutations in TCIRG1 are a frequent cause of autosomal recessive osteopetrosis in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gerritsen, E.J.A. et al. Autosomal recessive osteopetrosis: variability of findings at diagnosis and during the natural course. Pediatrics 93, 247–253 (1994).
Lazner, F., Gowwn, M., Pavasovic, D. & Kola, I. Osteopetrosis and osteoporosis: two sides of the same coin. Hum. Mol. Genet. 8, 1839–1846 (1999).
Kong, Y.-Y. et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymphnode organogenesis. Nature 397, 315–323 (1999).
Marks, S., Seifert, M. & Lane, P. Osteosclerosis: a recessive skeletal mutation on chromosome 19 in the mouse. J. Hered. 76, 171–176 (1985).
Soriano, P., Montgomery C., Geske, R. & Bradley, A. Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64, 693–702 (1991).
Saftig, P. et al. Impaired osteoclastic bone resorption leads to osteopetrosis in cathepsin-K-deficient mice. Proc. Natl Acad. Sci. USA 95, 13453–13458 (1998).
Heaney, C. et al. Human autosomal recessive osteopetrosis maps to 11q13, a position predicted by comparative mapping of the murine osteosclerosis (oc) mutation. Hum. Mol. Genet. 7, 1407–1414 (1998).
Forgac, M. Structure and properties of the vacuolar (H+)- ATPases. J. Biol. Chem. 274, 12951–12954 (1999).
Li, Y.P., Chen, W., Liang, Y., Li, E. & Stashenko, P. ATP6i-deficient mice exhibit severe osteopetrosis due to loss of osteoclast-mediated extracellular acidification. Nature Genet. 23, 447–451 (1999).
Scimeca, J.C. et al. The gene encoding the mouse homologue of the human osteoclast-specific 116-kD V-ATPase subunit bears a deletion in osteosclerotic (oc/oc) mutants. Bone 26, 207–213 (2000).
Väänänen, K.H., Zhao, H., Mulari, M. & Halleen, J.M. The cell biology of osteoclast function. J. Cell. Sci. 113, 377–381 (2000).
Li, Y.P., Chen, W. & Stashenko, P. Molecular cloning and characterization of a putative novel human osteoclast-specific 116-kD vacuolar proton pump subunit. Biochem. Biophys. Res. Commun. 218, 813–821 (1996).
Heinemann, T. et al. Genomic organization of the gene coding for TIRC7, a novel membrane protein essential for T cell activation. Genomics 57, 398–406 (1999).
Baron, R., Neff, L., Louvard, D. & Courtoy, P.J. Cell-mediated extracellular acidification and bone resorption: evidence for a low pH in resorbing lacunae and localization of a 100-kD lysosomal membrane protein at the osteoclast ruffled border. J. Cell Biol. 101, 2210–2222 (1985).
Blair, H.C., Teitelbaum, S.L., Ghiselli, R. & Gluck, S. Osteoclastic bone resorption by a polarized vacuolar proton pump. Science 245, 855–857 (1989).
Utku, N. et al. Prevention of acute allograft rejection by antibody targeting of TIRC7, a novel T cell membrane protein. Immunity 9, 509–518 (1998).
Maquat, L.E. Defects in RNA splicing and the consequence of shortened translational reading frames. Am. J. Hum. Genet. 59, 279–286 (1996).
Eapen, M., Davies, S.M., Ramsay N.K. & Orchard, P.J. Hematopoietic stem cell transplantation for infantile osteopetrosis. Bone Marrow Transplant. 22, 941–946 (1998).
Villa, A. et al. Partial V(D)J recombination activity leads to Omenn syndrome. Cell 93, 885–896 (1998).
Macchi, P. et al. Mutations of JAK3 gene in patients with autosomal severe combined immunodeficiency (SCID). Nature 377, 65–68 (1995).
Acknowledgements
We thank R. Dulbecco, L. Rossi Bernardi and D. Brown for their encouragement; M. Perrella for comments; S. Sencer and D. Unruh for assistance with samples from family members; V. Starnes for typing of the manuscript; and D. Strina, L. Susani, M. Littardi, J. Hatton and E.M. Catò for technical assistance. This work was partially supported by grants from Telethon, Italy (E.917 to A.V.; E.C0682 to A.F. and E.668 to L.D.N.), Biomed2 grant CT-983007 (to L.D.N.) and the Minnesota Medical Foundation and Vikings Children Fund (P.J.O.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frattini, A., Orchard, P., Sobacchi, C. et al. Defects in TCIRG1 subunit of the vacuolar proton pump are responsible for a subset of human autosomal recessive osteopetrosis. Nat Genet 25, 343–346 (2000). https://doi.org/10.1038/77131
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77131
This article is cited by
-
Osteopetrorickets: two contradictory patterns—one unifying diagnosis
Skeletal Radiology (2024)
-
Glycolysis-related biomarker TCIRG1 participates in regulation of renal cell carcinoma progression and tumor immune microenvironment by affecting aerobic glycolysis and AKT/mTOR signaling pathway
Cancer Cell International (2023)
-
Alterations in the microenvironment and the effects produced of TRPV5 in osteoporosis
Journal of Translational Medicine (2023)
-
Atp6i deficient mouse model uncovers transforming growth factor-β1 /Smad2/3 as a key signaling pathway regulating odontoblast differentiation and tooth root formation
International Journal of Oral Science (2023)
-
Physiology and diseases of tissue-resident macrophages
Nature (2023)