Abstract
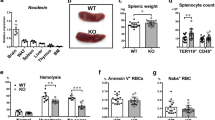
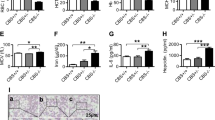
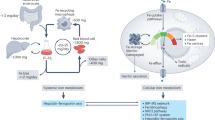
Plasma iron circulates bound to transferrin (Trf), which solubilizes the ferric ion and attenuates its reactivity. Diferric Trf interacts with cell-surface Trf receptor (Trfr) to undergo receptor-mediated endocytosis into specialized endosomes. Endosomal acidification leads to iron release, and iron is transported out of the endosome through the activity of divalent metal transporter 1 (DMT1, formerly Nramp2), a transmembrane iron transporter that functions only at low pH (ref. 1). Trf and Trfr then return to the cell surface for reuse, completing a highly efficient cycle. Although the Trf cycle is assumed to be the general mechanism for cellular iron uptake, this has not been validated experimentally. Mice with hypotransferrinaemia (hpx) have little or no plasma Trf (Refs 2,3). They have severe anaemia, indicating that the Trf cycle is essential for iron uptake by erythroid cells3. Other hpx tissues, however, are generally normal, and there is a paradoxical increase in intestinal iron absorption and iron storage3,4. To test the hypothesis that the Trf cycle has unique importance for erythropoiesis, we disrupted the Trfr gene in mice. This results in elimination of the Trf cycle, but leaves other Trf functions intact. Mice lacking Trfr have a more severe phenotype than hpx mice, affecting both erythropoiesis and neurologic development. Furthermore, haploinsufficiency for Trfr results in impaired erythroid development and abnormal iron homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fleming, M.D. et al. Nramp2 is mutated in the anemic Belgrade (b) rat: evidence of a role for Nramp2 in endosomal iron transport. Proc. Natl Acad. Sci. USA 95, 1148–1153 ( 1998).
Huggenvik, J.I. et al. A splicing defect in the mouse transferrin gene leads to congenital atransferrinemia. Blood 74, 482– 486 (1989).
Bernstein, S.E. Hereditary hypotransferrinemia with hemosiderosis, a murine disorder resembling human atransferrinemia. J. Lab. Clin. Med. 110, 690–705 (1987).
Craven, C.M. et al. Tissue distribution and clearance kinetics of non-transferrin-bound iron in the hypotransferrinemic mouse: a rodent model for hemochromatosis. Proc. Natl Acad. Sci. USA 84, 3457– 3461 (1987).
Dickinson, T.K. & Connor, J.R. Histological analysis of selected brain regions of hypotransferrinemic mice. Brain Res. 635, 169–178 (1994).
Inman, R.S. & Wessling-Resnick, M. Characterization of transferrin-independent iron transport in K562 cells. Unique properties provide evidence for multiple pathways of iron uptake. J. Biol. Chem. 268, 8521–8528 (1993).
Sturrock, A., Alexander, J., Lamb, J., Craven, C.M. & Kaplan, J. Characterization of a transferrin-independent uptake system for iron in HeLa cells. J. Biol. Chem. 265, 3139–3145 (1990).
Gelvan, D., Fibach, E., Meyron-Holtz, E.G. & Konijn, A.M. Ferritin uptake by human erythroid precursors is a regulated iron uptake pathway. Blood 88, 3200–3207 (1996).
Andrews, N.C. & Bridges, K.R. in Hematology of Infancy and Childhood (eds Nathan, D.G. & Orkin, S.H.) 423–461 (W.B. Saunders, Philadelphia, 1997).
Anderson, G.J., Powell, L.W. & Halliday, J.W. Transferrin receptor distribution and regulation in the rat small intestine. Effect of iron stores and erythropoiesis. Gastroenterology 98, 576–585 (1990).
Fleming, M.D. et al. Microcytic anaemia mice have a mutation in Nramp2, a candidate iron transporter gene. Nature Genet. 16 , 383–386 (1997).
Parkkila, S. et al. Association of the transferrin receptor in human placenta with HFE, the protein defective in hereditary hemochromatosis. Proc. Natl Acad. Sci. USA 94, 13198– 13202 (1997).
Feder, J.N. et al. The hemochromatosis gene product complexes with the transferrin receptor and lowers its affinity for ligand binding. Proc. Natl Acad. Sci. USA 95, 1472–1477 (1998).
Lebron, J.A. et al. Crystal structure of the hemochromatosis protein HFE and characterization of its interaction with transferrin receptor. Cell 93, 111–123 (1998).
Zhou, X.Y. et al. HFE gene knockout produces mouse model of hereditary hemochromatosis. Proc. Natl Acad. Sci. USA 95, 2492– 2497 (1998).
Nehls, M. et al. Two genetically separable steps in the differentiation of thymic epithelium. Science 272, 886– 889 (1996).
Laird, P.W. et al. Simplified mammalian DNA isolation procedure. Nucleic Acids Res. 19, 4293 ( 1991).
Fujiwara, Y., Browne, C.P., Cuniff, K., Goff, S.C. & Orkin, S.H. Arrested development of embryonic red cell precursors in mouse embryos lacking transcription factor GATA-1. Proc. Natl Acad. Sci. USA 93, 12355–12358 (1996).
Wang, L.C. et al. Yolk sac angiogenic defect and intra-embryonic apoptosis in mice lacking the Ets-related factor TEL. EMBO J. 16 , 4374–4383 (1997).
Joiner, C.H. et al. Increased cation permeability in mutant mouse red blood cells with defective membrane skeletons. Blood 86, 4307–4314 (1995).
Torrance, J.D. & Bothwell, T.H. in Methods in Hematolory: Iron (ed. Cook, J.D.) 104–109 (Churchill Livingstone Press, New York, 1980).
Acknowledgements
We thank A. Sharpe for providing ES cells; R. Mortenson for providing the targeting vector and advice on its use; M. Kim for technical assistance in sequencing phage clones; C. Browne for tissue culture advice; K. Cuniff for help with karyotyping; M. McDevitt for advice on embryo dissections; C. Brugnara for assistance with haematological measurements; Brigham and Women's Hospital Pathology and Hematology labs for sample analysis; S. Orkin for sharing equipment; and M. Fleming, V. Blank, V. Sellers and other members of the Andrews laboratory for reviewing the manuscript and discussing these results. N.C.A. is an investigator of the Howard Hughes Medical Institute. This work was partially supported by National Institutes Health grants HL51057 to N.C.A. and HL03503 to J.E.L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Levy, J., Jin, O., Fujiwara, Y. et al. Transferrin receptor is necessary for development of erythrocytes and the nervous system. Nat Genet 21, 396–399 (1999). https://doi.org/10.1038/7727
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/7727
This article is cited by
-
Mechanisms controlling cellular and systemic iron homeostasis
Nature Reviews Molecular Cell Biology (2024)
-
A Novel Homozygous Germline Mutation in Transferrin Receptor 1 (TfR1) Leads to Combined Immunodeficiency and Provides New Insights into Iron-Immunity Axis
Journal of Clinical Immunology (2024)
-
Molecular insights into placental iron transfer mechanisms and maternofetal regulation
Archives of Gynecology and Obstetrics (2023)
-
Ferroptosis As a Mechanism for Health Effects of Essential Trace Elements and Potentially Toxic Trace Elements
Biological Trace Element Research (2023)
-
The muscle-enriched myokine Musclin impairs beige fat thermogenesis and systemic energy homeostasis via Tfr1/PKA signaling in male mice
Nature Communications (2023)