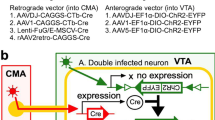
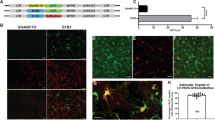
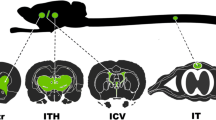
Abstract
To evaluate the potential for adenovirus–mediated central nervous system (CMS) gene transfer, the replication deficient recombinant adenovirus vectors Ad.RSVβgal (coding for β–galactosidase) and Ad–α1AT (coding for human α1–antitrypsin) were administered to the lateral ventricle of rats. Ad.RSVβgal transferred β–galactosidase to ependymal cells lining the ventricles whereas Ad–α1AT mediated α1 –antitrypsin secretion into the cerebral spinal fluid for 1 week. These observations, together with β–galactosidase activity in the globus pallidus and substantia nigra following stereotactic administration of Ad.RSVβgal to the globus pallidus, suggest that adenovirus vectors will be useful for CNS gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Harbaugh, R.E., Novel CNS-directed drug delivery systems in Alzheimer's disease and other neurological disorders. Neurobiol. Aging 10, 623–629 (1989).
Krieger, D.T. & Martin, J.B. Brain peptides. New Engl. J. Med. 304, 944–951 (1981).
Pardridge, W.M. Recent advances in blood-brain barrier transport. Ann. Rev. Pharmacol. Toxicol. 28, 25–39 (1988).
Wood, J.H. in Neurobiology of Cerebrospynal Fluid (Plenum Press, New York, 1983).
Brumback, R.R. in The Cerebrospinal Fluid (eds Herndon, R. & Brumback, R.) 15–43 (Kluwer Academic Publishers, 1989).
Rosemberg, M.B. et al. Grafting genetically modified cells to the damaged brain: restorative effects of NGF expression. Science 2, 1575–1578 (1988).
Shimohama, S. et al. Grafting genetically modified cells into the rat brain: characteristic of E coli β-galactosidase as a reporter gene. Brain Res. molec. Brain Res. 5, 271–278 (1989).
Dobson, A.T., Margolis, T.P., Sedarati, F., Stevens, J.G. & Feldman, L.T. A latent, nonpathogenetic HSV-1-derived vector stably express β-galactosidase in mouse neurons. Neuron 5, 353–360 (1990).
Gage, F.H. et al. Grafting genetically modified cells to the brain: conceptual and technical issues. Prog. Brain Res. 82, 1–10 (1990).
Kawaja, M.D., Rosemberg, M.B., Yoshida, K. & Gage, F.H. Somatic gene transfer of nerve growth factor promotes the survival of axotomized septal neurons and the regeneration of their axons in adult rats. J. Neurosci. 12, 2849–2864 (1992).
Wolfe, J.H., Deshmane, S.L. & Frazer, N.W. Herpesvirus vector gene transfer and expression of β-glucuronidase in the central nervous system of MPS VII mice. Nature Genet. 1, 379–384 (1992).
Culver, K.W. et al. In vivo gene transfer with retroviral vector-producer cells for treatment of experimental brain tumors. Science 256, 1550–1552 (1992).
Andersen, J.K., Garber, D.A., Meaney, C.A. & Breakfield, X.O. Gene transfer into mammalian nervous system using herpes virus vectors: extended expression of bacterial lacZ in neurons using the neuron-specific enolase promoter. Hum. Gene Ther. 3, 487–499 (1992).
Huang, Q. et al. Introduction of a foreign gene (Escherichia coli lacZ) into rat neostriatal neurons using herpes simplex virus mutants: a light and electron microscopy study. Exp. Neurol. 115, 303–316 (1992).
Federoff, H.J., Geshwind, M.D., Geller, A.I. & Kessler, J.A. Expression of nerve growth factor in vivo from defective herpes simplex virus 1 vector prevents effects of axotomy on sympathetic ganglia. Proc. natn. Acad. Sci. U.S.A. 89, 1636–1640 (1992).
Ono, T., Fujino, Y., Tsuchiya, T. & Tsuda, M. Plasmid DNAs directly injected into mouse brain with lipofectin can be incorporated and expressed by brain cells. Neurosci. Lett. 117, 259–263 (1990).
Rosenfeld, M.A. et al. Adenovirus-mediated transfer of a recombinant α1-antitrypsin gene to the lung epithelium in vivo. Science 252, 431–434 (1991).
Rosenfeld, M.A. et al. In vivo transfer of the human cystic fibrosis transmembrane conductance regulator gene to the airway epithelium. Cell 68, 143–155 (1992).
Jaffe, H.A. et al. Adenovirus-mediated in vivo gene transfer and expression in normal rat liver. Nature Genet. 1, 372–378 (1992).
Mastrangeli, A. et al. Diversity of airway epithelial cell targets for in vivo recombinant adenovirus-mediated gene transfer. J. clin. Invest. 91, 225–234 (1993).
Miettinen, M., Clark, R. & Virtanen, I. Intermediate filament proteins in choroid plexus and ependyma and their tumors. Am. J. Pathol. 123, 231–240 (1986).
Kasper, M. Cytokeratin in intracranial and intraspinal tissue. Adv. Anat Embryol. Cell. Biol. 126, 1–82 (1992).
Balercia, G., Bentivoglio, M. & Kruger, L. Fine structural organization of the ependymal region of the paraventricular nucleus of the rat thalamus and its relation with projection neurons. J. Neurocytol. 21, 105–109 (1991).
Bass, N. & Lundborg, P. Postnatal development of bulk flow in the cerebro-spinal fluid system of the albino rat: clearance of carboxyl-[14C]inulin after intrathecal infusion. Brain Res. 52, 323–332 (1973).
Pardrige, W. Strategies for drug delivery through the blood-brain barrier. Neurobiol. Aging 10, 636–637 (1989).
Bowsher, D. Pathway of absorption of proteins from the cerebrospinal fluid: an autoradiographic study in the cat. Anat. Rec. 128, 23–39 (1957).
Lemarchand, P. et al. Adenovirus-mediated transfer of a recombinant human α1-antitrypsin cDNA to human endothelial cells. Proc. natn. Acad. Sci. U.S.A. 89, 6482–6486 (1992).
Horwitz, M.S. Adenoviridae and Their Replication. in Fundamental Virology (eds Fields, B. N. & Knipe, D.M.) 1679–1721 (Raven Press, New York, 1991).
Rotman, B. Measurement of activity of single molecules of β-D-galactosidase. Proc. natn. Acad. Sci. U.S.A. 47, 1981–1991 (1961).
Paxinos, G. & Watson, C. in The Rat Brain in Stereotaxic Coordinates (Academic Press, Sidney, 1982).
Dannenberg, A.M. & Suga, M. in Methods for Studying Mononuclear Phagocytes, (eds Adams, D.O., Edelson, P.J. & Koren, H.S.) 375–395 (Academic Press, New York, 1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bajocchi, G., Feldman, S., Crystal, R. et al. Direct in vivo gene transfer to ependymal cells in the central nervous system using recombinant adenovirus vectors. Nat Genet 3, 229–234 (1993). https://doi.org/10.1038/ng0393-229
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0393-229
This article is cited by
-
Retargeted adenoviruses for radiation-guided gene delivery
Cancer Gene Therapy (2016)
-
Electroporation markedly improves Sleeping Beauty transposon-induced tumorigenesis in mice
Cancer Gene Therapy (2014)
-
Targeting choroid plexus epithelia and ventricular ependyma for drug delivery to the central nervous system
BMC Neuroscience (2011)
-
Forebrain ependymal cells are Notch-dependent and generate neuroblasts and astrocytes after stroke
Nature Neuroscience (2009)
-
Genetic manipulation of adult mouse neurogenic niches by in vivo electroporation
Nature Methods (2008)