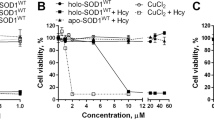
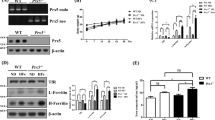
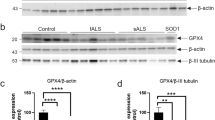
Abstract
Reactive oxygen species (ROS) have been implicated in a wide range of degenerative processes including amyotrophic lateral sclerosis, ischemic heart disease, Alzheimer disease, Parkinson disease and aging1–5. ROS are generated by mitochondria as the toxic by-products of oxidative phosphorylation, their energy generating pathway6,7. Genetic inactivation of the mitochondrial form of superoxide dismutase in mice results in dilated cardiomyopathy, hepatic lipid accumulation and early neonatal death8. We report that treatment with the superoxide dismutase (SOD) mimetic Manganese 5,10,15, 20-tetrakis (4-benzoic acid) porphyrin (MnTBAP) rescues these Sod2tm1cje(−/−) mutant mice from this systemic pathology and dramatically prolongs their survival. The animals instead develop a pronounced movement disorder progressing to total debilitation by three weeks of age. Neuropathologic evaluation reveals a striking spongiform degeneration of the cortex and specific brain stem nuclei associated with gliosis and intramyelinic vacuolization similar to that observed in cytotoxic edema and disorders associated with mitochondrial abnormalities such as Leighs disease and Canavans disease. We believe that due to the failure of MnTBAP to cross the blood brain barrier progressive neuropathology is caused by excessive mitochondrial production of ROS. Consequently, MnTBAP-treated Sod2tm1cje(−/−) mice may provide an excellent model for examining the relationship between free radicals and neurodegenerative diseases and for screening new drugs to treat these disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yim, M.B. et al. A gain-of-function of an amyotrophic lateral sclerosis-associated Cu,Zn-superoxide dismutase mutant: An enhancement of free radical formation due to a decrease in Km for hydrogen peroxide. Proc. Natal. Acad. Sci. USA 93, 5709–5714 (1996).
Wallace, D.C. Mitochondrial DMA Mutations and Bioenergetic Defects in Aging and Degenerative Diseases. in Molecular and Genetic Basis of Neurologic Disease (eds Rosenburg, D. N., Prusiner, S.B., DiMauro, S. & Barchi, R.L.) 237–269 (Butterworth Heinemann, Boston, 1996).
Szabo, C. Physiological and Pathophysiological Roles of Nitric Oxide in the Central Nervous System. Brain Research Bulletin 41, 131–141 (1996).
Schulz, J.B. & Seal, M.F. Neuroprotective effects of free radical scavengers and energy repletion in animal models of neurodegenerative disease. Ann. N.Y.Acad. Sci. 765, 100–118 (1995).
Simonian, N.A. & Coyle, J.T. Oxidative stress in neurodegenerative diseases. Annu. Rev. Pharmacol. Toxicol. 36, 83106 (1996).
Turrens, J.F. & Boveris, A. Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem. J. 191, 421–427 (1980).
Turrens, J.F., Alexandre, A. & Lehninger, A.L. Ubisemiquinone is the electron donor for superoxide formation by complex III of heart mitochondria. Arch. Biochem. Biophys. 237, 408–414 (1985).
Li, Y. et al. Dilated cardiomyopathy and neonatal lethality in mutant mice lacking manganese superoxide dismutase. Nature Genet. 11, 376–381 (1995).
Day, B.J., Shawen, S., Liochev, S.I. & Crapo, J.D. A metalloporphyrin superoxide dismutase mimetic protects against paraquat-induced endothelial cell injury, in vitro. J. Pharmacol. Exp. Ther. 275, 1227–1232 (1995).
Day, B.J. & Crapo, J.D. A metalloporphyrin superoxide dismutase mimetic protects against paraquat-induced lung injury in vivo. Toxicol. Appl. Pharmacol. 140, 94–100 (1996).
Szabo, C., Day, B.J. & Salzman, A.L. Evaluation of the relative contribution of nitric oxide and peroxynitrite to the suppression of mitochondrial respiration in immunostimulated macrophages using a manganese mesoporphyrin superoxide dismutase mimetic and peroxynitrite scavenger. FEBS Lett. 381, 82–86 (1996).
Zingarelli, B., Day, B.J., Crapo, J.D., Salzman, A.L. & Szabo, C. The potential role of peroxynitrite in the vascular contractile and cellular energetic failure in endotoxic shock, Br. J. Pharmacol. 120, 259–267 (1997).
Lebovitz, R.M. et al Neurodegeneration, myocardial injury, and perinatal death in mitochondria! superoxide dismutase-deficient mice. Proc. Natal. Acad. Sci. USA 93, 9782–9787 (1996).
Hirano, A., Zimmerman, H.M. & Levine, S. Intramyelinic and extracellular spaces in triethyltin intoxication. J. Neuropathol. Exp. Neurol. 27, 571−580 (1968).
Kimura, S., Kobayashi, T. & Amemiya, F. Myelin splitting in the spongy lesion in Leigh encephalopathy. Pediatr. Neurol. 7, 56–58 (1991).
Adachi, M., Wallace, B.J. & Volk, B.W. Ultramicroscopic and histochemical studies of spongy degeneration (van Bogaert and Bertrand type). J. Neuropathol. Exp. Neurol. 26, 164–165 (1967).
Melov, S., Hinerfeld, D., Esposito, L. & Wallace, D.C. Multi-organ characterization of mitochondrial genomic rearrangements in ad libitum and caloric restricted mice show striking somatic mitochondrial DNA rearrangments with age. Nucleic Acids Res. 25, 974–982 (1997).
Wong, P.C. et al. An adverse property of a familial ALS-linked SOD1 mutation causes motor neuron disease characterized by vacuolar degeneration of mitochondria. Neuron 14, 1105–1116 (1995).
Beal, M.F., ging, energy, and oxidative stress in neurodegenerative diseases. Ann. Neurol. 38, 357–366 (1995).
Halliwell, B. Reactive oxygen species and the central nervous system. J Neurochem. 59, 1609−1623 (1992).
Hensley, K. et al. A model for beta-amyloid aggregation and neurotoxicity based on the free radical generating capacity of the peptide: implications of molecular shrapnel for Alzheimers disease. Proc. West. Pharmacol. Soc. 38, 113–120 (1995).
Mattson, M.P. Calcium and neuronal injury in Alzheimers disease. Contributions of beta-amyloid precursor protein mismetabolism, free radicals, and metabolic compromise. Ann. N.Y Acad. Sci. 747, 50–76 (1994).
Bancroft, J.D. & Stevens, A. Theory and Practice of Histological Techniques (Churchill Livingstone, New York, 1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Melov, S., Schneider, J., Day, B. et al. A novel neurological phenotype in mice lacking mitochondrial manganese superoxide dismutase. Nat Genet 18, 159–163 (1998). https://doi.org/10.1038/ng0298-159
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0298-159
This article is cited by
-
Molecular and cellular pathways contributing to brain aging
Behavioral and Brain Functions (2021)
-
The Spinal Cord Damage in a Rat Asphyxial Cardiac Arrest/Resuscitation Model
Neurocritical Care (2021)
-
Detection of 8-oxoguanine and apurinic/apyrimidinic sites using a fluorophore-labeled probe with cell-penetrating ability
BMC Molecular and Cell Biology (2019)
-
Drosophila Voltage-Gated Calcium Channel α1-Subunits Regulate Cardiac Function in the Aging Heart
Scientific Reports (2018)
-
Connexin 43 and ATP-sensitive potassium channels crosstalk: a missing link in hypoxia/ischemia stress
Cell and Tissue Research (2018)