Abstract
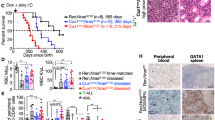
The Ras signal transduction pathway is often deregulated in human myeloid leukaemia. For example, activating point mutations in RAS genes are found in some patients with juvenile chronic myelogenous leukaemia (JCML), while other patients with JCML show loss of the neurofibromatosis type 1 (NF1) gene, a Ras GTPase activating protein. By generating mice whose haematopoietic system is reconsituted with NF1 deficient haematopoietic stem cells we show that NF1 gene loss, by itself, is sufficient to produce the myeloproliferative symptoms associated with human JCML. We also provide evidence to indicate that NF1 gene loss induces myeloproliferative disease through a Ras-mediated hypersensitivity to granulocyte/macrophage-colony stimulating factor (GM-CSF). Finally, we describe a genetic screen for identifying genes that cooperate with NF1 gene loss during progression to acute myeloid leukaemia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Riccardi, V.M. In Neurofibromatosis: phenotype, natural history, and pathogenesis. (Johns Hopkins Press, Baltimore, 1992).
Bader, J.L. Neurofibromatosis and cancer. Ann. N. Y. Acad. Sci. 486, 56–65 (1986).
Hope, D.G. & Mulvihill, J.J. Malignancy in neunofibromatosis. Adv. Neurol. 29, 33–56 (1981).
Bader, J.L. & Miller, R.W. Neurofibromatosis and childhood leukemia. J. Pediat. 92, 925–929 (1978).
Gutmann, D.H. & Collins, F.S. The neurofibromatosis type 1 gene and its protein product, neurofibromin. Neuron 10, 335–343 (1993).
Barbacid, M. Ras oncogenes: their role in neoplasia. Eur. J. Clin. Invest. 20, 225–235 (1990).
Johnson, M.R., Look, A.T., DeClue, J.E., Valentine, M.B. & Lowy, D.R. Inactivation of the NF1 gene in human melanoma and neuroblastoma cell lines without impaired regulation of GTP–Ras. Proc. Natl. Acad. Sci. USA 90, 5539–5543 (1993).
Johnson, M.R. et al. Neurofibromin can inhibit ras-dependent growth by a mechanism independent of its GTPase-accelerating function. Molec. cell. Biol. 14, 641–645 (1994).
Shannon, K.M. et al. Loss of the normal NF1 allele from the bone marrow of children with type 1 neurofibromatosis and malignant myeloid disorders. New Engl. J. Med. 330, 597–601 (1994).
Jacks, T. et al. Tumor predisposition in mice heterozygous for a targeted mutation in Nf1. Nature Genet. 7, 353–361 (1994).
Buchberg, A.M., Bedigian, H.G., Jenkins, N.A. & Copeland, N.G., Evi-2, a common integration site involved in murine myeloid leukemogenesis. Molec. Cell. Biol. 10, 4658–4666 (1990).
Largaespada, D.A., Shaughnessy, J.D., Jenkins, N.A. & Copeland, N.G. Retroviral insertion at the Evi-2 locus in BXH-2 myeloid leukemia cell lines disrupts Nf1 expression without changes in steady state ras-GTP levels. J. Virol. 69, 5095–5102 (1995).
Kalra, R., Paderanga, D.C., Olson, K. & Shannon, K.M. Genetic analysis is consistent with the hypothesis that NF1 limits myeloid growth through p21 ras. Blood 84, 3435–3439 (1994).
Brannan, C.I. et al. Targeted disruption of the neurofibromatosis type-1 gene leads to developmental abnormalities in heart and various neural crest-derived tissues. Genes Dev. 8, 1019–1029 (1994).
Moore, M.A.S., McNeill, T.A. & Haskill, J.S. Density distribution analysis of in vivo and in vitro colony forming cells in developing fetal liver. J. Cell. Physiol. 75, 181–192 (1970).
Dunn, A.R. et al. In Biological characterization of regulators encoded by cloned hemopoietic growth factor gene sequences. (Cold Spring Harbor Laboratory, New York, 1985).
Emanuel, P.O., Bates, L.J., Castleberry, R.B., Gualtieri, R.J. & Zuckerman, K.S. Selective hypersensitivity to granulaocyte-macrophage colony-stimulating factor by juvenile chronic myeloid leukemia hematopoietic progenitors. Blood 77, 925–929 (1991).
Miyajima, A., Mui, A.L., Ogorochi, T. & Sakamaki, K. Receptors for granulocyte-macrophage colony-stimulating factor, interleukin-3, and interleukin-5. Blood 82, 1960–1974 (1993).
Castro-Malaspina, H. et al.Subacute and chronic myelomonocytic leukemia in children (Juvenile CML): Clinical and hematologic observations, and identification of prognostic factors. Cancer 54, 675–686 (1984).
Hestdal, K. et al. Characterization and regulation of RB6-8C5 antigen expression on murine bone marrow cells. J. Immunol. 147, 22–28 (1991).
Lang, R.A. et al. Transgenic mice expressing a hemopoietic growth factor gene (GM-CSF) develop accumulations of macrophages, blindness, and a fatal syndrome of tissue damage. Cell 51, 675–686 (1987).
Johnson, G.R., Gonda, T.J., Metcalf, D., Hariharan, I.K. & Cory, S. A lethal myeloproliferative syndrome in mice transplanted with bone marrow cells infected with a retrovirus expressing granulocyte-macrophage colony stimulating factor. EMBO J. 8, 441–448 (1989).
Satoh, I., Nakafuku, M., Miyajima, A. & Kaziro, Y. Involvement of ras p21 protein in signal-transduction pathways from interleukin 2, interleukin 3, and granulocyte/macrophage colony-stimulating factor, but not from interleukin 4. Proc. Natl. Acad. Sci. USA 88, 3314–3318 (1991).
DeClue, J.E. et al. Abnormal regulation of mammalian p21 ras contrinutes to malignant tumor growth in von Recklinghausen (Type 1)neurofibromatosis. Cell 69, 265–273 (1992).
Basu, T.N. et al. Aberrant regulation of ras proteins in malignant tumor cells from type 1 neurofibromatosis patients. Nature 356, 713–715 (1992).
Gonda, T.J., E.M., Macmillan, Townsend, P.V. & Hapel, A.J. Differentiation state and responses to hematopoietic growth factors of murine myeloid cells transformed by myb. Blood 82, 2813–2822 (1993).
Gonda, T.J. Molecular and cellular activities of Myb: regulation in normal hemopoiesis and transformation. Seminars in virology 2, 351–361 (1991).
Janssen, J.W.G. et al. RAS gene mutations in acute and chronic myelocytic leukemias, chronic myeloproliferative disorders, and myelodysplastic syndromes. Proc. Natl. Acad. Sci. USA 84, 9228–9232 (1987).
The, I. et al. Neurofibromatosis type 1 gene mutations in neuroblastoma. Nature Genet. 3, 62–66 (1993).
Bedigian, H.G., Johnson, D.A., Jenkins, N.A., Copeland, N.G. & Evans, R. Spontaneous and induced leukemias of myeloid origin in recombinant inbred BXH–2 mice. J. Virol. 51, 586–594 (1984).
Jenkins, N.A., Copeland, N.G., Taylor, B.A., Bedigian, H.G. & Lee, B.K. Ecotropic murine leukemia virus DNA content of normal and lymphomatous tissues of BXH–2 recombinant inbred mice. J. Virol. 42, 379–388 (1982).
von Lohuizen, M. & Berns, A. Tumorigenesis by slow transforming retroviruses—an update. Biochim. Biophys. Acta. 1032 (2–3), 213–235 (1990).
Peters, G. Oncogenes at viral integration sites. Cell. Growth Differ. 1, 503–510 (1990).
Puil, L. et al. Bcr-abl oncoproteins bind directly to activators of the ras signalling pathway. EMBO J. 13, 764–773 (1994).
Pendergast, A.M. et al. BCR-ABL-induced oncogenesis is mediated by direct interaction of the SH2 domain of the GRB-2 adaptor protein. Cell 75, 175–185 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Largaespada, D., Brannan, C., Jenkins, N. et al. Nf1 deficiency causes Ras-Dediated granulocyte/macrophage colony stimulating factor hypersensitivity and chronic myeloid leukaemia. Nat Genet 12, 137–143 (1996). https://doi.org/10.1038/ng0296-137
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0296-137
This article is cited by
-
NF1 mutations are recurrent in adult acute myeloid leukemia and confer poor outcome
Leukemia (2018)
-
Aberrant Myeloid Differentiation Contributes to the Development of Osteoporosis in Neurofibromatosis Type 1
Current Osteoporosis Reports (2016)
-
Modulation of Ras signaling alters the toxicity of hydroquinone, a benzene metabolite and component of cigarette smoke
BMC Cancer (2014)
-
Neurofibromatosis type 1-related gastrointestinal stromal tumors: a special reference to loss of heterozygosity at 14q and 22q
Journal of Cancer Research and Clinical Oncology (2009)
-
Juvenile myelomonocytic leukemia and chronic myelomonocytic leukemia
Leukemia (2008)