Abstract
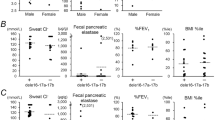
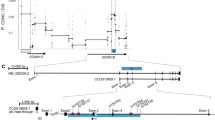
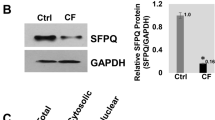
Variable in–frame skipping of exon 9 in cystic fibrosis transmembrane conductance regulator (CFTR) mRNA transcripts (exon 9−) occurs in the respiratory epithelium. To explore the genetic basis of this event, we evaluated respiratory epithelial cells and blood leukocytes from 124 individuals (38 with cystic fibrosis (CF), 86 without CF). We found an inverse relationship between the length of the polythymidine tract at the exon 9 splice branch/acceptor site and the proportion of exon 9− CFTR mRNA transcripts. These results strongly indicate a genetic basis in vivo modulating post–transcriptional processing of CFTR mRNA transcripts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rommens, J.M. et al. Identification of the cystic fibrosis gene: chromosome walking and jumping. Science 245, 1059–1065 (1989).
Riordan, J.R. et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245, 1066–1073 (1989).
Kerem, B.-S. et al. Identification of the cystic fibrosis gene: genetic analysis. Science 245, 1073–1080 (1989).
Zielenski, J. et al. Genomic DMA sequence of the cystic fibrosis transmembrane conductance regulator (CFTR) gene. Genomics 10, 214–228 (1991).
Rich, D.P. et al. Expression of cystic fibrosis transmembrane conductance regulator corrects defective chloride channel regulation in cystic fibrosis airway epithelial cells. Nature 347, 358–363 (1991).
Anderson, M.P., Rich, D.P., Gregory, R.J., Smith, A.E. & Welsh, M.J. Generation of cAMP-activated chloride currents by expression of CFTR. Science 251, 679–682 (1990).
Kartner, N. et al. Expression of the cystic fibrosis gene in non-epithelial invertebrate cells produces a regulated anion conductance. Cell 64, 681–691 (1991).
Anderson, M.P. et al. Demonstration that CFTR is a chloride channel by alteration of its anion selectivity. Science 253, 202–205 (1991).
Tabcharani, J.A., Chang, X.-B., Riordan, J.R. & Hanrahan, J.W. Phosphorylation-regualted Cl− channel in CHO cells stably expressing the cystic fibrosis gene. Nature 352, 628–631 (1991).
Bear, C.E. et al. Cl− channel activity in Xenopus oocytes expressing the cystic fibrosis gene. J. biol. Chem. 266, 19142–19145 (1991).
Bear, C.E. et al. Purification and functional reconstitution of the cystic fibrosis transmembrane conductance regulator (CFTR). Cell 68, 809–818 (1992).
Denning, G.M., Ostedgaard, L.S., Cheng, S.H., Smith, A.E. & Welsh, M.J. Localization of cystic fibrosis transmembrane conductance regulator in chloride secretory epithelia. J. clin. Invest. 89, 339–349 (1992).
Boat, T.F., Welsh, M.J. & Beaudet, A.L. in The Metabolic Basis of Inherited Diseases 6th edn (eds Scriver, C.R., Beaudet, A.L., Sly, W.S. & Valle, D.) 2649–2680 (McGraw-Hill, New York, 1989).
Welsh, M.J. & Fick, R.B. Cystic fibrosis. J. clin. Invest. 80, 1523–1526 (1987).
Quinton, P.M. Cystic fibrosis: a disease in electrolyte transport. FASEB J. 4, 2709–2717 (1990).
Drumm, M.L. et al. Chloride conductance expressed by ΔF508 and other mutant CFTRs in Xenopus oocytes. Science 254, 1797–1799 (1991).
Barinaga, M. Novel function discovered for the cystic fibrosis gene. Science 256, 444–445 (1992).
Yoshimura, K. et al. Expression of the cystic fibrosis transmembrane conductance regulator gene in cells of non-epithelial origin. Nucl. Acids Res. 19, 5417–5423 (1991).
Yoshimura, K. et al. The cystic fibrosis gene has a “housekeeping”-type promoter and is expressed at low levels in cells of epithelial origin. J. biol. Chem. 266, 9140–9144 (1991).
Trapnell, B.C. et al. Expression of the cystic fibrosis transmembrane conductance regulator gene in the respiratory tract of normal individuals and individuals with cystic fibrosis. Proc. natn. Acad. Sci. U.S.A. 88, 6565–6569 (1991).
Trapnell, B.C. et al. Down-regulation of cystic fibrosis gene mRNA transcript levels and induction of the cystic fibrosis chloride secretory phenotype in epithelial cells by phorbol ester. J. biol. Chem. 266, 10319–10323 (1991).
Bargon, J. et al. Down-regulation of cystic fibrosis transmembrane conductance regulator gene expression by agents that modulate intracellular divalent cations. Molec. cell Biol. 12, 1872–1878 (1992).
Bargon, J. et al. Expression of the cystic fibrosis transmembrane conductance regulator gene can be regulated by protein kinase C. J. biol. Chem. 267, 16056–16060 (1992).
Chu, C.-S. et al. Variable deletion of exon 9 coding sequences in cystic fibrosis transmembrane conductance regulator gene mRNA transcripts in normal bronchial epithelium. EMBO J. 10, 1355–1363 (1991).
Chu, C.-S., Trapnell, B.C., Curristin, S.M., Cutting, G.R. & Crystal, R.G. Extensive post-transcriptional deletion of the coding sequences for part of nucelotide-binding fold 1 in respiratory epithelial mRNA transcripts of the cystic fibrosis transmembrane conductance regulator gene is not associated with the clinical manifestations of cystic fibrosis. J. clin. Invest. 90, 785–790 (1992).
Cutting, G.R. et al. A cluster of cystic fibrosis mutations in the first nucleotide-binding fold of the cystic fibrosis conductance regulator protein. Nature 346, 366–369 (1990).
Kerem, B.-S. et al. Identification of mutations in regions corresponding to the two putative nucleotide (ATP)-binding folds of the cystic fibrosis gene. Proc. natn. Acad. Sci. U.S.A. 87, 8447–8451 (1990).
Cuppens, H. et al. A child, homozygous for a stop codon in exon 11, shows milder cystic fibrosis symptoms than her heterozygous nephew. J. med. Genet. 27, 717–719 (1990).
Gregory, R.J. et al. Maturation and function of cystic fibrosis transmembrane conductance regulator variants bearing mutations in putative nucleotide-binding domains 1 and 2. Molec. cell Biol. 11, 3886–3893 (1991).
Padgett, R.A. et al. Splicing of messenger RNA precursors. Ann. Rev. Biochem. 55, 1119–1150 (1986).
Krainer, A.R. & Maniatis, T. in Transcription and Splicing (eds Hames, B.D. & Glover, D.M.) 131–206 (IRL Press, Oxford, 1988).
Guthrie, C. Messenger RNA splicing in yeast: clues to why the spliceosome is a ribonucleoprotein. Science 253, 157–163 (1991).
Ruby, S.W. & Abelson, J. Pre-mRNA splicing in yeast. Trends Genet. 7, 79–85 (1991).
Smith, C.W.J., Porro, E.B., Patton, J.G. & Nadal-Ginard, B. Scanning from an independently specified branch point defines the 3′ splice site of mammalian introns. Nature 342, 243–247 (1989).
Reed, R. The organization of 3′ splice-site sequences in mammalian introns. Genes Dev. 3, 2113–2123 (1989).
Reed, R. & Maniatis, T. Intron sequences involved in lariat formation during pre-mRNA splicing. Cell 41, 95–105 (1985).
Frendewey, D. & Keller, W. Stepwise assembly of a pre-mRNA splicing complex requires U-snRNPs and specific intron sequences. Cell 42, 355–367 (1985).
Ruskin, B. & Green, M.R. Role of the 3′ splice site consensus sequence in mammalian pre-mRNA splicing. Nature 317, 732–734 (1985).
Bindereif, A. & Green, M.R. Ribonucleoprotein complex formation during pre-mRNA splicing in vitro. Molec. cell Biol. 6, 2582–2592 (1986).
Patterson, B. & Guthrie, C. A U-rich tract enhances usage of an alternative 3′ splice site in yeast. Cell 64, 181–187 (1991).
Kerem, E. et al. The relation between genotype and phenotype in cystic fibrosis – analysis of the most common mutation (ΔF508). New Engl. J. Med. 323, 1517–1522 (1990).
Zamore, P.D. & Green, M.R. Identification, purification, and biochemical characterization of U2 small nuclear ribonucleoprotein auxiliary factor. Proc. natn. Acad. Sci. U.S.A. 86, 9243–9247 (1989).
Garcia-Blanco, M.A., Jamison, S.F. & Sharp, P.A. Identification and purification of a 62,000-dalton protein that binds specifically to the polypyrimidine tract of introns. Genes Dev. 3, 1874–1886 (1989).
Wang, J. & Pederson, T. A 62,000 molecular weight spliceosome prote in crosslinks to the intron polypyrimidine tract. Nucl. Acids Res. 18, 5995–6001 (1990).
Chehab, F.F. et al. A dimorphic 4-bp repeat in the cystic fibrosis gene is in absolute linkage disequilibrium with the ΔF508 mutation: implications for prenatal diagnosis and mutation origin. Am. J. hum. Genet. 48, 223–226 (1991).
Estivill, X. et al. Patterns of polymorphism and linkage disequilibrium for cystic fibrosis. Genomics 1, 257–163 (1987).
Estivill, X. et al. A candidate for the cystic fibrosis locus isolated by selection for methylation-free islands. Nature 326, 840–845.
Morral, N., Nunes, V., Casals, T. & Estivill, X. CA/GT microsatellite alleles within the cystic fibrosis transmembrane conductance regulator (CFTR) gene are not generated by unequal crossingover. Genomics 10, 692–698 (1991).
Saiki, R.K. et al. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239, 487–491 (1988).
Chomczynski, P. & Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162, 156–159 (1987).
Southern, E.M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. molec. Biol. 98, 503–517 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chu, CS., Trapnell, B., Curristin, S. et al. Genetic basis of variable exon 9 skipping in cystic fibrosis transmembrane conductance regulator mRNA. Nat Genet 3, 151–156 (1993). https://doi.org/10.1038/ng0293-151
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0293-151
This article is cited by
-
The dominant findings of a recessive man: from Mendel’s kid pea to kidney
Pediatric Nephrology (2023)
-
Novel mutation c.1210-3C > G in cis with a poly-T tract of 5T affects CFTR mRNA splicing in a Chinese patient with cystic fibrosis
Frontiers of Medicine (2022)
-
Genetics of the congenital absence of the vas deferens
Human Genetics (2021)
-
The CFTR gene variants in Japanese children with idiopathic pancreatitis
Human Genome Variation (2019)
-
Genetics of male infertility
Nature Reviews Urology (2018)