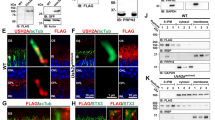
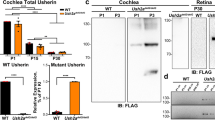
Abstract
Usher syndrome type I (USH1) is an autosomal recessive disorder characterized by congenital sensorineural hearing loss, vestibular dysfunction and visual impairment due to early onset retinitis pigmentosa1 (RP). So far, six loci (USH1A–USH1F) have been mapped, but only two USH1 genes have been identified: MYO7A (ref. 2) for USH1B and the gene encoding harmonin3,4 for USH1C. We identified a Cuban pedigree linked to the locus for Usher syndrome type 1D (MIM 601067) within the q2 region of chromosome 10 (ref. 5). Affected individuals present with congenital deafness and a highly variable degree of retinal degeneration. Using a positional candidate approach, we identified a new member of the cadherin gene superfamily, CDH23. It encodes a protein of 3,354 amino acids with a single transmembrane domain and 27 cadherin repeats. In the Cuban family, we detected two different mutations: a severe course of the retinal disease was observed in individuals homozygous for what is probably a truncating splice-site mutation (c.4488G→C), whereas mild RP is present in individuals carrying the homozygous missense mutation R1746Q. A variable expression of the retinal phenotype was seen in patients with a combination of both mutations. In addition, we identified two mutations, ΔM1281 and IVS51+5G→A, in a German USH1 patient. Our data show that different mutations in CDH23 result in USH1D with a variable retinal phenotype. In an accompanying paper6, it is shown that mutations in the mouse ortholog cause disorganization of inner ear stereocilia and deafness in the waltzer mouse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Keats, B.J. & Corey, D.P. The usher syndromes. Am. J. Med. Genet. 89, 158–166 (1999).
Weil, D. et al. Defective myosin VIIA gene responsible for Usher syndrome type 1B. Nature 374, 60–61 (1995).
Verpy, E. et al. A defect in harmonin, a PDZ domain-containing protein expressed in the inner ear sensory hair cells, underlies usher syndrome type 1C. Nature Genet. 26, 51–55 (2000).
Bitner-Glindzicz, M. et al. A recessive contiguous gene deletion causing infantile hyperinsulinism, enteropathy and deafness identifies the usher type 1C gene. Nature Genet. 26, 56–60 ( 2000).
Wayne, S. et al. Localization of the Usher syndrome type ID gene (Ush1D) to chromosome 10. Hum. Mol. Genet. 5, 1689– 1692 (1996).
Di Palma, F. et al. Mutations in Cdh23, encoding a new type of cadherin, cause stereocilia disorganization in waltzer, the mouse model for Usher syndrome type 1D. Nature Genet. 27, 103– 107 (2001).
Bryda, E.C., Ling, H. & Flaherty, L. A high-resolution genetic map around waltzer on mouse chromosome 10 and identification of a new allele of waltzer. Mamm. Genome 8, 1–4 ( 1997).
Yagi, T. & Takeichi, M. Cadherin superfamily genes: functions, genomic organization, and neurologic diversity. Genes Dev. 14, 1169–1180 (2000).
Legan, P.K. & Richardson, G.P. Extracellular matrix and cell adhesion molecules in the developing inner ear. Semin. Cell. Dev. Biol. 8, 217–224 ( 1997).
Noben-Trauth, K., Zheng, Q.Y., Johnson, K.R. & Nishina, P.M. mdfw: a deafness susceptibility locus that interacts with deaf waddler (dfw) . Genomics 44, 266–272 (1997).
Nollet, F., Kools, P. & van Roy, F. Phylogenetic analysis of the cadherin superfamily allows identification of six major subfamilies besides several solitary members. J. Mol. Biol. 299, 551–572 (2000).
Zhang, M.Q. Statistical features of human exons and their flanking regions. Hum. Mol. Genet. 7, 919–932 (1998).
Mount, S.M. Genomic sequence, splicing, and gene annotation. Am. J. Hum. Genet. 67, 788–792 ( 2000).
Jones, C.T., McIntosh, I., Keston, M., Ferguson, A. & Brock, D.J. Three novel mutations in the cystic fibrosis gene detected by chemical cleavage: analysis of variant splicing and a nonsense mutation. Hum. Mol. Genet. 1, 11– 17 (1992).
Alagramam, K.N. et al. The mouse Ames waltzer hearing-loss mutant is caused by mutation of Pcdh15, a new protocadherin gene. Nature Genet. 27, 99–102 (2001).
Carrasquillo, M.M., Zlotogora, J., Barges, S. & Chakravarti, A. Two different connexin 26 mutations in an inbred kindred segregating non-syndromic recessive deafness: implications for genetic studies in isolated populations . Hum. Mol. Genet. 6, 2163– 2172 (1997).
Liu, X.Z. et al. Mutations in the myosin VIIA gene cause a wide phenotypic spectrum, including atypical Usher syndrome. Am. J. Hum. Genet. 63, 909–912 (1998).
Weil, D. et al. The autosomal recessive isolated deafness, DFNB2, and the Usher 1B syndrome are allelic defects of the myosin-VIIA gene. Nature Genet. 16, 191–193 ( 1997).
Liu, X.Z. et al. Autosomal dominant non-syndromic deafness caused by a mutation in the myosin VIIA gene. Nature Genet. 17, 268–269 (1997).
Jain, P.K. et al. A gene for recessive nonsyndromic sensorineural deafness (DFNB18) maps to the chromosomal region 11p14–p15.1 containing the Usher syndrome type 1C gene. Genomics 50, 290– 292 (1998).
Chaib, H. et al. Mapping of DFNB12, a gene for a non-syndromal autosomal recessive deafness, to chromosome 10q21–22. Hum. Mol. Genet. 5, 1061–1064 (1996).
Chothia, C. & Jones, E.Y. The molecular structure of cell adhesion molecules. Annu. Rev. Biochem. 66, 823–862 (1997).
Eudy, J.D. et al. Mutation of a gene encoding a protein with extracellular matrix motifs in Usher syndrome type IIa. Science 280, 1753–1757 (1998).
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 ( 1990).
Thompson, J.D., Gibson, T.J., Plewniak, F., Jeanmougin, F. & Higgins, D.G. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 25, 4876– 4882 (1997).
Saitou, N. & Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425 (1987)
Dib, C. et al. A comprehensive genetic map of the human genome based on 5,264 microsatellites. Nature 380, 152– 154 (1996).
Acknowledgements
We thank B. Ressler and I. Jantke for technical assistance. This study was financially supported by Mrs. B. Höll, The Foundation Fighting Blindness, Faun-Stiftung, Forschung contra Blindheit, Initiative Usher-Syndrom, Deutsche Forschungsgemeinschaft, Pro Retina Deutschland and BONFOR.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Table A
Detailed clinical data of patients from the USH Cuban family. Genotypes referring to the two mutations (R1746Q and c.4488G --> C) described in the paper are given for each individual. (PDF 19 kb)
Table B
95°C for 3 min; three cycles at 95°C for 15 s, AT+4°C for 10 s, 72°C for 30 s; three cycles at 95°C for 15 s, AT+2°C for 10 s, 72°C for 30 s; 30 cycles at 95°C for 15 s, AT for 10 s, 72°C for 30 s and 72°C for 10 min. (DOC 76 kb)
Table C
Non-synonymous sequence variants in the coding region of the human CDH23 gene. All substitutions were present in unaffected control individuals and are therefore unlikely to represent pathogenic mutations. (DOC 26 kb)
Figure B
Characterization of the human CDH23 gene and protein. a, Complete amino acid sequence of the longest CDH23 isoform. Both microexons (exon 11 and exon 33) as well as exon 68 are alternatively spliced as shown by cDNA analysis. A putative signal peptide (1-23) and a single transmembrane domain (3067-3086) are marked by a dotted and a solid box, respectively. The 27 cadherin repeats are indicated by gray background. Positions of exons are indicated by arrows, exon numbering is given above the sequence. b, Phylogenetic tree (ClustalX, default parameters) of selected members of the cadherin superfamily based on the sequence of the first cadherin repeat. A calibration bar for the branch lengths is shown in the upper right corner. CDH23 is closely related to two Drosophila melanogaster proteins, CG6445 and CG3389, and to the human flamingo proteins. GenBank accession numbers of selected cadherins: E-cadherin (CAA84586), N-cadherin (AAB22854), VE-cadherin (AAB41796), CDH7 (BAA87415), Dachsous (AAA79329), Cdh3 (AAB50920), PCDH-alpha1 (AAD43699), PCDH-b2 (CAA60685), PCDH1 (AAA36419), PCDH7 (BAA25194), Flamingo1 (AAF61929), Flamingo2 (AAF61930), CDH23 (AF312024), CG6445 (AAF49351), CG3389 (AAF55082), Fat1 (CAA60685), MEGF1 (BAA32458). Ce, Caenorhabditis elegans; Dm, Drosophila; Hs, Homo sapiens; Rn, Rattus norvegicus. (PDF 15 kb)
Rights and permissions
About this article
Cite this article
Bolz, H., von Brederlow, B., Ramírez, A. et al. Mutation of CDH23, encoding a new member of the cadherin gene family, causes Usher syndrome type 1D. Nat Genet 27, 108–112 (2001). https://doi.org/10.1038/83667
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83667
This article is cited by
-
Genetic etiology of hereditary hearing loss in the Gulf Cooperation Council countries
Human Genetics (2022)
-
The genetic and phenotypic landscapes of Usher syndrome: from disease mechanisms to a new classification
Human Genetics (2022)
-
Variants in CDH23 cause a broad spectrum of hearing loss: from non-syndromic to syndromic hearing loss as well as from congenital to age-related hearing loss
Human Genetics (2022)
-
Usher syndrome type IV: clinically and molecularly confirmed by novel ARSG variants
Human Genetics (2022)
-
Mechanisms in cochlear hair cell mechano-electrical transduction for acquisition of sound frequency and intensity
Cellular and Molecular Life Sciences (2021)