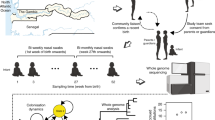
Abstract
Bacterial pathogens evolve during the infection of their human host1,2,3,4,5,6,7,8, but separating adaptive and neutral mutations remains challenging9,10,11. Here we identify bacterial genes under adaptive evolution by tracking recurrent patterns of mutations in the same pathogenic strain during the infection of multiple individuals. We conducted a retrospective study of a Burkholderia dolosa outbreak among subjects with cystic fibrosis, sequencing the genomes of 112 isolates collected from 14 individuals over 16 years. We find that 17 bacterial genes acquired nonsynonymous mutations in multiple individuals, which indicates parallel adaptive evolution. Mutations in these genes affect important pathogenic phenotypes, including antibiotic resistance and bacterial membrane composition and implicate oxygen-dependent regulation as paramount in lung infections. Several genes have not previously been implicated in pathogenesis and may represent new therapeutic targets. The identification of parallel molecular evolution as a pathogen spreads among multiple individuals points to the key selection forces it experiences within human hosts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Suerbaum, S. & Josenhans, C. Helicobacter pylori evolution and phenotypic diversification in a changing host. Nat. Rev. Microbiol. 5, 441–452 (2007).
Smith, E.E. et al. Genetic adaptation by Pseudomonas aeruginosa to the airways of cystic fibrosis patients. Proc. Natl. Acad. Sci. USA 103, 8487–8492 (2006).
Musher, D.M. et al. Emergence of macrolide resistance during treatment of pneumococcal pneumonia. N. Engl. J. Med. 346, 630–631 (2002).
Wong, A. & Kassen, R. Parallel evolution and local differentiation in quinolone resistance in Pseudomonas aeruginosa. Microbiology 157, 937–944 (2011).
Zdziarski, J. et al. Host imprints on bacterial genomes—rapid divergent evolution in individual patients. PLoS Pathog. 6, e1001078 (2010).
Yang, L. et al. Evolutionary dynamics of a bacteria in a human host environment. Proc. Natl. Acad. Sci. USA 108, 7481–7486 (2011).
Kennemann, L. et al. Helicobacter pylori genome evolution during human infection. Proc. Natl. Acad. Sci. USA 108, 5033–5038 (2011).
Mwangi, M.M. et al. Tracking the in vivo evolution of multidrug resistance in Staphylococcus aureus by whole-genome sequencing. Proc. Natl. Acad. Sci. USA 104, 9451–9456 (2007).
Harris, S.R. et al. Evolution of MRSA during hospital transmission and intercontinental spread. Science 327, 469–474 (2010).
Goodarzi, H., Hottes, A.K. & Tavazoie, S. Global discovery of adaptive mutations. Nat. Methods 6, 581–583 (2009).
Pleasance, E.D. et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature 463, 191–196 (2010).
Moxon, E.R., Rainey, P.B., Nowak, M.A. & Lenski, R.E. Adaptive evolution of highly mutable loci in pathogenic bacteria. Curr. Biol. 4, 24–33 (1994).
van der Woude, M.W. & Bäumler, A.J. Phase and antigenic variation in bacteria. Clin. Microbiol. Rev. 17, 581–611 (2004).
Croucher, N.J. et al. Rapid pneumococcal evolution in response to clinical interventions. Science 331, 430–434 (2011).
Holt, K.E. et al. High-throughput sequencing provides insights into genome variation and evolution in Salmonella Typhi. Nat. Genet. 40, 987–993 (2008).
Pallen, M.J. & Wren, B.W. Bacterial pathogenomics. Nature 449, 835–842 (2007).
Elena, S.F. & Lenski, R.E. Evolution experiments with microorganisms: the dynamics and genetic bases of adaptation. Nat. Rev. Genet. 4, 457–469 (2003).
Woods, R. et al. Tests of parallel molecular evolution in long-term experiment with Escherichia coli. Proc. Natl. Acad. Sci. USA 103, 9107–9112 (2006).
Barrick, J.E. et al. Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 461, 1243–1247 (2009).
Lipuma, J.J. The changing microbial epidemiology in cystic fibrosis. Clin. Microbiol. Rev. 23, 299–323 (2010).
Vermis, K. et al. Proposal to accommodate Burkholderia cepacia genomovar VI as Burkholderia dolosa sp. nov. Int. J. Syst. Evol. Microbiol. 54, 689–691 (2004).
Lipuma, J.J. Update on the Burkholderia cepacia complex. Curr. Opin. Pulm. Med. 11, 528–533 (2005).
LiPuma, J.J., Dasen, S.E., Nielson, D.W., Stern, R.C. & Stull, T.L. Person-to-person transmission of Pseudomonas cepacia between patients with cystic fibrosis. Lancet 336, 1094–1096 (1990).
Biddick, R., Spilker, T., Martin, A. & LiPuma, J.J. Evidence of transmission of Burkholderia cepacia, Burkholderia multivorans and Burkholderia dolosa among persons with cystic fibrosis. FEMS Microbiol. Lett. 228, 57–62 (2003).
Kalish, L.A. et al. Impact of Burkholderia dolosa on lung function and survival in cystic fibrosis. Am. J. Respir. Crit. Care Med. 173, 421–425 (2006).
Morelli, G. et al. Microevolution of Helicobacter pylori during prolonged infection of single hosts and within families. PLoS Genet. 6, e1001036 (2010).
Sibley, C.D. et al. A polymicrobial perspective of pulmonary infections exposes an enigmatic pathogen in cystic fibrosis patients. Proc. Natl. Acad. Sci. USA 105, 15070–15075 (2008).
Guss, A.M. et al. Phylogenetic and metabolic diversity of bacteria associated with cystic fibrosis. ISME J. 5, 20–29 (2011).
Mowat, E. et al. Psuedomonas aeruginosa population diversity and turnover in cystic fibrosis infections. Am. J. Respir. Crit. Care Med. 183, 1674–1679 (2011).
Wilder, C.N., Allada, G. & Schuster, M. Instantaneous within-patient diversity of Psuedomonas aeruginosa quorum-sensing populations from cystic fibrosis lung infections. Infect. Immun. 77, 5631–5639 (2009).
Weigel, L.M., Steward, C.D. & Tenover, F.C. gyrA mutations associated with fluoroquinolone resistance in eight species of Enterobacteriaceae. Antimicrob. Agents Chemother. 42, 2661–2667 (1998).
Reyna, F., Huesca, M., Gonzalez, V. & Fuchs, L.Y. Salmonella typhimurium gyrA mutations associated with fluoroquinolone resistance. Antimicrob. Agents Chemother. 39, 1621–1623 (1995).
Silhavy, T.J., Kahne, D. & Walker, S. The bacterial cell envelope. Cold Spring Harb. Perspect. Biol. 2, a000414 (2010).
Vinion-Dubiel, A.D. & Goldberg, J.B. Lipopolysaccharide of Burkholderia cepacia complex. J. Endotoxin Res. 9, 201–213 (2003).
Ortega, X. et al. Reconstitution of O-specific lipopolysaccharide expression in Burkholderia cenocepacia strain J2315, which is associated with transmissible infections in patients with cystic fibrosis. J. Bacteriol. 187, 1324–1333 (2005).
Crosson, S., McGrath, P.T., Stephens, C., McAdams, H.H. & Shapiro, L. Conserved modular design of an oxygen sensory/signaling network with species-specific output. Proc. Natl. Acad. Sci. USA 102, 8018–8023 (2005).
Worlitzsch, D. et al. Effects of reduced mucus oxygen concentration in airway Pseudomonas infections of cystic fibrosis patients. J. Clin. Invest. 109, 317–325 (2002).
Marteyn, B. et al. Modulation of Shigella virulence in response to available oxygen in vivo. Nature 465, 355–358 (2010).
Acknowledgements
We are grateful to M. Caimano, M. Cendron, P. Kokorowski, S. Lory, C. Marx, N. Delany, S. Walker, M. Waldor and R. Ward for insightful discussions and comments, to O. Iartchouk, A. Brown, M. Light and their team at Partners HealthCare Center for Personalized Genetic Medicine (PCPGM) for Illumina sequencing, to J. Deane and L. Williams for technical assistance, to S. Vargas for assistance with IRB protocols and to M. Baym, M. Ernebjerg, A. Palmer, E. Toprak, K. Vetsigian, Z. Yao and all of the Kishony lab members for helpful discussions and general support. J.B.M. was supported by the Foundational Questions in Evolutionary Biology Prize Fellowship and the Systems Biology PhD Program (Harvard Medical School). G.P.P. was supported in part by The Mannion Fund for Research of the Center for the Critically Ill Child of Children's Hospital Boston. J.J.L. was supported by the Cystic Fibrosis Foundation. This work was supported in part by US National Institutes of Health grants (GM080177 to the Systems Biology Department, Harvard Medical School and GM081617 to R.K.), by a grant from the New England Regional Center of Excellence for Biodefense and Emerging Infectious Diseases (NERCE; AI057159 to R.K.) and by a Harvard Catalyst grant (to R.K., A.J.M. and M. Cendron).
Author information
Authors and Affiliations
Contributions
J.-B.M., A.J.M. and R.K. conceived of the study. J.J.L., A.J.M. and G.P.P. collected the clinical samples. T.D.L. and N.L. performed resistance phenotyping. J.B.G., D.R., M.R.D., D.S. and G.P.P. performed LPS phenotyping and complementation. M.A., G.P.-B., A.J.M. and G.P.P. conducted chart review and provided medical information. T.D.L., J.-B.M. and R.K. performed whole-genome sequencing and data analysis. T.D.L., J.-B.M., J.J.L., A.J.M., G.P.P. and R.K. interpreted the results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1, 4 and 5 and Supplementary Note. (PDF 1715 kb)
Supplementary Table 2
Polymorphic loci among 113 B. dolosa isolates (XLSX 217 kb)
Supplementary Table 3
Genes mutated during an epidemic of B. dolosa (XLSX 26 kb)
Rights and permissions
About this article
Cite this article
Lieberman, T., Michel, JB., Aingaran, M. et al. Parallel bacterial evolution within multiple patients identifies candidate pathogenicity genes. Nat Genet 43, 1275–1280 (2011). https://doi.org/10.1038/ng.997
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.997
This article is cited by
-
Localized coevolution between microbial predator and prey alters community-wide gene expression and ecosystem function
The ISME Journal (2023)
-
Mixed strain pathogen populations accelerate the evolution of antibiotic resistance in patients
Nature Communications (2023)
-
Multidrug resistance plasmids underlie clonal expansions and international spread of Salmonella enterica serotype 1,4,[5],12:i:- ST34 in Southeast Asia
Communications Biology (2023)
-
Intermittent antibiotic treatment of bacterial biofilms favors the rapid evolution of resistance
Communications Biology (2023)
-
Quantitative systems-based prediction of antimicrobial resistance evolution
npj Systems Biology and Applications (2023)