Abstract
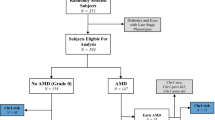
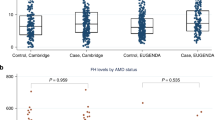
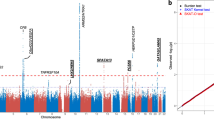
Two common variants in the gene encoding complement factor H (CFH), the Y402H substitution (rs1061170, c.1204C>T)1,2,3,4 and the intronic rs1410996 SNP5,6, explain 17% of age-related macular degeneration (AMD) liability. However, proof for the involvement of CFH, as opposed to a neighboring transcript, and knowledge of the potential mechanism of susceptibility alleles are lacking. Assuming that rare functional variants might provide mechanistic insights, we used genotype data and high-throughput sequencing to discover a rare, high-risk CFH haplotype with a c.3628C>T mutation that resulted in an R1210C substitution. This allele has been implicated previously in atypical hemolytic uremic syndrome, and it abrogates C-terminal ligand binding7,8. Genotyping R1210C in 2,423 AMD cases and 1,122 controls demonstrated high penetrance (present in 40 cases versus 1 control, P = 7.0 × 10−6) and an association with a 6-year-earlier onset of disease (P = 2.3 × 10−6). This result suggests that loss-of-function alleles at CFH are likely to drive AMD risk. This finding represents one of the first instances in which a common complex disease variant has led to the discovery of a rare penetrant mutation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hageman, G.S. et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc. Natl. Acad. Sci. USA 102, 7227–7232 (2005).
Haines, J.L. et al. Complement factor H variant increases the risk of age-related macular degeneration. Science 308, 419–421 (2005).
Klein, R.J. et al. Complement factor H polymorphism in age-related macular degeneration. Science 308, 385–389 (2005).
Edwards, A.O. et al. Complement factor H polymorphism and age-related macular degeneration. Science 308, 421–424 (2005).
Maller, J. et al. Common variation in three genes, including a noncoding variant in CFH, strongly influences risk of age-related macular degeneration. Nat. Genet. 38, 1055–1059 (2006).
Li, M. et al. CFH haplotypes without the Y402H coding variant show strong association with susceptibility to age-related macular degeneration. Nat. Genet. 38, 1049–1054 (2006).
Jozsi, M. et al. Factor H and atypical hemolytic uremic syndrome: mutations in the C-terminus cause structural changes and defective recognition functions. J. Am. Soc. Nephrol. 17, 170–177 (2006).
Manuelian, T. et al. Mutations in factor H reduce binding affinity to C3b and heparin and surface attachment to endothelial cells in hemolytic uremic syndrome. J. Clin. Invest. 111, 1181–1190 (2003).
Friedman, D.S. et al. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 122, 564–572 (2004).
Davila, S. et al. Genome-wide association study identifies variants in the CFH region associated with host susceptibility to meningococcal disease. Nat. Genet. 42, 772–776 (2010).
Jozsi, M. & Zipfel, P.F. Factor H family proteins and human diseases. Trends Immunol. 29, 380–387 (2008).
Boon, C.J. et al. Basal laminar drusen caused by compound heterozygous variants in the CFH gene. Am. J. Hum. Genet. 82, 516–523 (2008).
Devlin, B. & Roeder, K. Genomic control for association studies. Biometrics 55, 997–1004 (1999).
Price, A.L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38, 904–909 (2006).
Sanchez-Corral, P. et al. Structural and functional characterization of factor H mutations associated with atypical hemolytic uremic syndrome. Am. J. Hum. Genet. 71, 1285–1295 (2002).
Martinez-Barricarte, R. et al. The complement factor H R1210C mutation is associated with atypical hemolytic uremic syndrome. J. Am. Soc. Nephrol. 19, 639–646 (2008).
Caprioli, J. et al. The molecular basis of familial hemolytic uremic syndrome: mutation analysis of factor H gene reveals a hot spot in short consensus repeat 20. J. Am. Soc. Nephrol. 12, 297–307 (2001).
Caprioli, J. et al. Complement factor H mutations and gene polymorphisms in haemolytic uraemic syndrome: the C-257T, the A2089G and the G2881T polymorphisms are strongly associated with the disease. Hum. Mol. Genet. 12, 3385–3395 (2003).
Neumann, H.P. et al. Haemolytic uraemic syndrome and mutations of the factor H gene: a registry-based study of German speaking countries. J. Med. Genet. 40, 676–681 (2003).
Servais, A. et al. Primary glomerulonephritis with isolated C3 deposits: a new entity which shares common genetic risk factors with haemolytic uraemic syndrome. J. Med. Genet. 44, 193–199 (2007).
Ferreira, V.P. et al. The binding of factor H to a complex of physiological polyanions and C3b on cells is impaired in atypical hemolytic uremic syndrome. J. Immunol. 182, 7009–7018 (2009).
Weiner, D.E., Tighiouart, H., Reynolds, R. & Seddon, J.M. Kidney function, albuminuria and age-related macular degeneration in NHANES III. Nephrol. Dial. Transplant published online, doi:10.1093/ndt/gfr022 (21 February 2011).
Fagerness, J.A. et al. Variation near complement factor I is associated with risk of advanced AMD. Eur. J. Hum. Genet. 17, 100–104 (2009).
Gold, B. et al. Variation in factor B (BF) and complement component 2 (C2) genes is associated with age-related macular degeneration. Nat. Genet. 38, 458–462 (2006).
Maller, J.B. et al. Variation in complement factor 3 is associated with risk of age-related macular degeneration. Nat. Genet. 39, 1200–1201 (2007).
Noris, M. & Remuzzi, G. Atypical hemolytic-uremic syndrome. N. Engl. J. Med. 361, 1676–1687 (2009).
Seddon, J.M., Sharma, S. & Adelman, R.A. Evaluation of the clinical age-related maculopathy staging system. Ophthalmology 113, 260–266 (2006).
Seddon, J.M., Santangelo, S.L., Book, K., Chong, S. & Cote, J. A genomewide scan for age-related macular degeneration provides evidence for linkage to several chromosomal regions. Am. J. Hum. Genet. 73, 780–790 (2003).
Seddon, J.M., Cote, J., Davis, N. & Rosner, B. Progression of age-related macular degeneration: association with body mass index, waist circumference, and waist-hip ratio. Arch. Ophthalmol. 121, 785–792 (2003).
Seddon, J.M., Cote, J., Page, W.F., Aggen, S.H. & Neale, M.C. The US twin study of age-related macular degeneration: relative roles of genetic and environmental influences. Arch. Ophthalmol. 123, 321–327 (2005).
Seddon, J.M. et al. Dietary fat and risk for advanced age-related macular degeneration. Arch. Ophthalmol. 119, 1191–1199 (2001).
Neale, B.M. et al. Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC). Proc. Natl. Acad. Sci. USA 107, 7395–7400 (2010).
Kathiresan, S. et al. Genome-wide association of early-onset myocardial infarction with single nucleotide polymorphisms and copy number variants. Nat. Genet. 41, 334–341 (2009).
Raychaudhuri, S. et al. Associations of CFHR1–CFHR3 deletion and a CFH SNP to age-related macular degeneration are not independent. Nat. Genet. 42, 553–555 (2010).
Yang, Z. et al. Toll-like receptor 3 and geographic atrophy in age-related macular degeneration. N. Engl. J. Med. 359, 1456–1463 (2008).
Yang, Z. et al. Genetic and functional dissection of HTRA1 and LOC387715 in age-related macular degeneration. PLoS Genet. 6, e1000836 (2010).
Chen, W. et al. Genetic variants near TIMP3 and high-density lipoprotein-associated loci influence susceptibility to age-related macular degeneration. Proc. Natl. Acad. Sci. USA 107, 7401–7406 (2010).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Ewing, B., Hillier, L., Wendl, M.C. & Green, P. Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Res. 8, 175–185 (1998).
Gordon, D., Abajian, C. & Green, P. Consed: a graphical tool for sequence finishing. Genome Res. 8, 195–202 (1998).
Stephens, M., Sloan, J.S., Robertson, P.D., Scheet, P. & Nickerson, D.A. Automating sequence-based detection and genotyping of SNPs from diploid samples. Nat. Genet. 38, 375–381 (2006).
Browning, B.L. & Browning, S.R. A unified approach to genotype imputation and haplotype phase inference for large data sets of trios and unrelated individuals. Am. J. Hum. Genet. 84, 210–223 (2009).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Levey, A.S. et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann. Intern. Med. 130, 461–470 (1999).
Acknowledgements
We appreciate the contribution to the research of J.M.S. of an anonymous donor. This research was supported in part by grants RO1-EY11309 (J.M.S.), K08AR055688-01A1 (S. Raychaudhuri) and U01 MH085520-01 (S. Ripke) from the US National Institutes of Health (NIH), and by the Massachusetts Lions Eye Research Fund, Inc., the American Macular Degeneration Research Fund, the Foundation Fighting Blindness, the Macular Vision Research Foundation, a Research to Prevent Blindness Challenge Grant to the New England Eye Center, Department of Ophthalmology, Tufts University School of Medicine, and the Macular Degeneration Research Fund of the Ophthalmic Epidemiology and Genetics Service, New England Eye Center, Tufts Medical Center, Tufts University School of Medicine. N.K. is a Distinguished Brumley Professor. The MIGen study was funded by grants from the NIH National Heart, Lung, and Blood Institute (NLBI) (R01HL087676) and the NIH National Center for Research Resources (NCRR). We acknowledge informal and helpful discussions from our colleagues B. Neale, R. Plenge, P. de Bakker, D. Reich, E. Stahl, B. Stranger and B. Voight.
Author information
Authors and Affiliations
Contributions
S. Raychaudhuri, J.M.S., N.K. and M.J.D. conceptualized this study, wrote the initial manuscript and interpreted all results. S. Raychaudhuri oversaw the statistical analyses and coordinated collaborative experimental efforts. J.M.S. and M.J.D. oversaw genome-wide genotyping of the Boston-phased data. S. Ripke analyzed the genome-wide Boston-phased genotype data to assess population stratification and recent ancestry. J.M.S., K.C., R.R. and Y.Y. organized the Boston clinical cohort. B.C., P.C. and D.J.Z. organized the Baltimore clinical cohort. P.L.T. and N.K. genotyped the Baltimore samples. A.T. genotyped the Boston samples. O.I., S.G., S.V. and K.M. sequenced the CFH gene.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–3 and 5–8 (PDF 1266 kb)
Supplementary Table 4
Polymorphic variants from high throughput sequencing of CFH in 33 cases and 27 controls (XLSX 96 kb)
Rights and permissions
About this article
Cite this article
Raychaudhuri, S., Iartchouk, O., Chin, K. et al. A rare penetrant mutation in CFH confers high risk of age-related macular degeneration. Nat Genet 43, 1232–1236 (2011). https://doi.org/10.1038/ng.976
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.976
This article is cited by
-
Multi-omics analysis in human retina uncovers ultraconserved cis-regulatory elements at rare eye disease loci
Nature Communications (2024)
-
A novel missense compound heterozygous variant in TLR1 gene is associated with susceptibility to rheumatoid arthritis — structural perspective and functional annotations
Clinical Rheumatology (2023)
-
Complement Mediators in Development to Treat Age-Related Macular Degeneration
Drugs & Aging (2022)
-
Age-related macular degeneration and premorbid allergic diseases: a population-based case–control study
Scientific Reports (2021)
-
Investigational Agents in Development for the Treatment of Geographic Atrophy Secondary to Age-Related Macular Degeneration
BioDrugs (2021)