Abstract
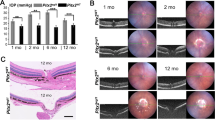
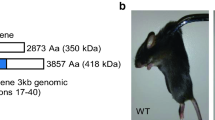
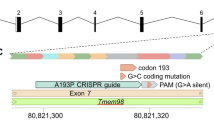
Angle-closure glaucoma (ACG) is a subset of glaucoma affecting 16 million people1,2,3. Although 4 million people are bilaterally blind from ACG4,5, the causative molecular mechanisms of ACG remain to be defined. High intraocular pressure induces glaucoma in ACG. High intraocular pressure traditionally was suggested to result from the iris blocking or closing the angle of the eye, thereby limiting aqueous humor drainage. Eyes from individuals with ACG often have a modestly decreased axial length, shallow anterior chamber and relatively large lens, features that predispose to angle closure6. Here we show that genetic alteration of a previously unidentified serine protease (PRSS56) alters axial length and causes a mouse phenotype resembling ACG. Mutations affecting this protease also cause a severe decrease of axial length in individuals with posterior microphthalmia. Together, these data suggest that alterations of this serine protease may contribute to a spectrum of human ocular conditions including reduced ocular size and ACG.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Quigley, H.A. Number of people with glaucoma worldwide. Br. J. Ophthalmol. 80, 389–393 (1996).
Ritch, R. & Lowe, R.F. Angle-closure glaucoma: mechanisms and epidemiology. in The Glaucomas (eds. Ritch, R., Shields, M.B. & Krupin, T.) 801–819 (Mosby-Year Book, Inc., St. Louis, Missouri, 1996).
Shields, M.B. Pupillary-block glaucomas. in Textbook of Glaucoma (ed. Cooke, D.B.) 177–194 (Williams & Wilkens, Baltimore, Maryland, USA, 1996).
Seah, S.K. et al. Incidence of acute primary angle-closure glaucoma in Singapore. An island-wide survey. Arch. Ophthalmol. 115, 1436–1440 (1997).
Foster, P.J. & Johnson, G.J. Glaucoma in China: how big is the problem? Br. J. Ophthalmol. 85, 1277–1282 (2001).
Lowe, R.F. Aetiology of the anatomical basis for primary angle-closure glaucoma. Biometrical comparisons between normal eyes and eyes with primary angle-closure glaucoma. Br. J. Ophthalmol. 54, 161–169 (1970).
Quigley, H.A. Angle-closure glaucoma-simpler answers to complex mechanisms: LXVI Edward Jackson Memorial Lecture. Am. J. Ophthalmol. 148, 657–669.e1 (2009).
Sakai, H. et al. Uveal effusion in primary angle-closure glaucoma. Ophthalmology 112, 413–419 (2005).
Hrabé de Angelis, M.H. et al. Genome-wide, large-scale production of mutant mice by ENU mutagenesis. Nat. Genet. 25, 444–447 (2000).
Elagouz, M., Stanescu-Segall, D. & Jackson, T.L. Uveal effusion syndrome. Surv. Ophthalmol. 55, 134–145 (2010).
Kumar, R.S. et al. Confirmation of the presence of uveal effusion in Asian eyes with primary angle closure glaucoma: an ultrasound biomicroscopy study. Arch. Ophthalmol. 126, 1647–1651 (2008).
Quigley, H.A. What's the choroid got to do with angle closure? Arch. Ophthalmol. 127, 693–694 (2009).
Ritch, R. Angle-closure glaucoma: clinical types. in The Glaucomas, Clinical Science, Vol. 2 (ed. Ritch, R., Shields, M.B., Krupin, T.) 821–840 (Mosby Year Book, St. Louis, Missouri, USA, 1996).
Ross, J., Jiang, H., Kanost, M.R. & Wang, Y. Serine proteases and their homologs in the Drosophila melanogaster genome: an initial analysis of sequence conservation and phylogenetic relationships. Gene 304, 117–131 (2003).
Rymer, J. & Wildsoet, C.F. The role of the retinal pigment epithelium in eye growth regulation and myopia: a review. Vis. Neurosci. 22, 251–261 (2005).
Mott, J.D. & Werb, Z. Regulation of matrix biology by matrix metalloproteinases. Curr. Opin. Cell Biol. 16, 558–564 (2004).
Hausman, R.E. Ocular extracellular matrices in development. Prog. Retin. Eye Res. 26, 162–188 (2007).
Tektas, O.Y. & Lutjen-Drecoll, E. Structural changes of the trabecular meshwork in different kinds of glaucoma. Exp. Eye Res. 88, 769–775 (2009).
Keller, K.E., Aga, M., Bradley, J.M., Kelley, M.J. & Acott, T.S. Extracellular matrix turnover and outflow resistance. Exp. Eye Res. 88, 676–682 (2009).
Fleenor, D.L. et al. TGFβ2-induced changes in human trabecular meshwork: implications for intraocular pressure. Invest. Ophthalmol. Vis. Sci. 47, 226–234 (2006).
Jackson, T.L. et al. Scleral hydraulic conductivity and macromolecular diffusion in patients with uveal effusion syndrome. Invest. Ophthalmol. Vis. Sci. 49, 5033–5040 (2008).
Hmani-Aifa, M. et al. A genome-wide linkage scan in Tunisian families identifies a novel locus for non-syndromic posterior microphthalmia to chromosome 2q37.1. Hum. Genet. 126, 575–587 (2009).
Mutti, D.O. et al. Axial growth and changes in lenticular and corneal power during emmetropization in infants. Invest. Ophthalmol. Vis. Sci. 46, 3074–3080 (2005).
John, S.W.M., Hagaman, J.R., MacTaggart, T.E., Peng, L. & Smithes, O. Intraocular pressure in inbred mouse strains. Invest. Ophthalmol. Vis. Sci. 38, 249–253 (1997).
Aihara, M., Lindsey, J.D. & Weinreb, R.N. Aqueous humor dynamics in mouse. Invest. Ophthalmol. Vis. Sci. 44, 5168–5173 (2003).
Anderson, M.G. et al. Mutations in genes encoding melanosomal proteins cause pigmentary glaucoma in DBA/2J mice. Nat. Genet. 30, 81–85 (2002).
Smith, R.S., Sundberg, J.P. & John, S.W.M. The anterior segment. in Systematic Evaluation of the Mouse Eye: Anatomy, Pathology and Biomethods (eds. Smith, R.S., John, S.W.M., Nishina, P.M. & Sundberg, J.P.) 366 (CRC Press, Boca Raton, Florida, USA, 2002).
Howell, G.R. et al. Axons of retinal ganglion cells are insulted in the optic nerve early in DBA/2J glaucoma. J. Cell Biol. 179, 1523–1537 (2007).
Smith, R.S., Korb, D. & John, S.W.M. A goniolens for clinical monitoring of the mouse iridocorneal angle and optic nerve. Mol. Vis. 8, 26–31 (2002).
Akilesh, S., Shaffer, D.J. & Roopenian, D. Customized molecular phenotyping by quantitative gene expression and pattern recognition analysis. Genome Res. 13, 1719–1727 (2003).
Acknowledgements
The authors thank A. Bell, S. Kneeland, H. McLaughlin, A. Gillette, M. Ryan, Annette Molbaek, Åsa Schipper and The Jackson Laboratory's Fine Mapping Service and Allele Typing and Sequencing services for technical assistance. We thank P. Nishina for generous use of her OCT and J. Vance of Bioptigen for his expert technical assistance, G. Cox, P. Nishina and M. de Vries for comments on the manuscript and J. Hammer for help with graphics. The National Eye Institute grant EY11721, Barbara and Joseph Cohen Foundation, Ministère de l'Enseignement Supérieur de la Recherche Scientifique et de la Technologie, Tunisia, the Middle East and North Africa region (MENA) (Swedish research links programme supported by the Swedish Research Council (VR)/the Swedish International Development Cooperation Agency (SIDA)) and the Project 2006 planning grant # 348-2005-6336 supported this work. S.W.M.J. is an investigator of the Howard Hughes Medical Institute. We thank the family members for their valuable participation and cooperation.
Author information
Authors and Affiliations
Contributions
K.S.N. conceived and executed experiments and participated in study design, data interpretation and manuscript preparation. Z.A. participated in characterization of the protease and maintenance of the mouse strains, genotyping and phenotyping. A.L.K. participated in fine mapping, sequencing of candidate genes, maintenance of mouse strains and histological assessment. I.M.C. contributed to mutant identification, gene mapping and phenotyping. D.G.M. performed physiological experiments. I.S. participated in cell-biology–based experiments. G.R.H. participated in overall design of gene mapping and histological assessment. R.S.S. contributed to histological assessment and interpretation. S.W.M.J. conceived the study, oversaw all aspects of the study and participated in design, conducting experiments, data interpretation and manuscript preparation.
M.H.-A. contributed to experimental design for human families and conducted experiments and participated in data analysis and manuscript preparation. S.B.S. participated in sequencing of human candidate genes. W.B. is an ophthalmologist and performed clinical evaluation of the patients. Z.B. is also an ophthalmologist and performed clinical evaluation of the patients. B.H. participated in human gene sequencing. P.S. provided advice and help with sequencing of some human candidate genes. H.A. oversaw the human study and participated in data analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Figures 1–6 and Supplementary Note. (PDF 2984 kb)
Rights and permissions
About this article
Cite this article
Nair, K., Hmani-Aifa, M., Ali, Z. et al. Alteration of the serine protease PRSS56 causes angle-closure glaucoma in mice and posterior microphthalmia in humans and mice. Nat Genet 43, 579–584 (2011). https://doi.org/10.1038/ng.813
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.813
This article is cited by
-
Serine protease PRSS56, a novel cancer-testis antigen activated by DNA hypomethylation, promotes colorectal and gastric cancer progression via PI3K/AKT axis
Cell & Bioscience (2023)
-
Phenotypic consequences of a nanophthalmos-associated TMEM98 variant in human and mouse
Scientific Reports (2023)
-
Genetic analysis assists diagnosis of clinical systemic disease in children with excessive hyperopia
BMC Pediatrics (2022)
-
The majority of autosomal recessive nanophthalmos and posterior microphthalmia can be attributed to biallelic sequence and structural variants in MFRP and PRSS56
Scientific Reports (2020)
-
Novel TMEM98, MFRP, PRSS56 variants in a large United States high hyperopia and nanophthalmos cohort
Scientific Reports (2020)