Abstract
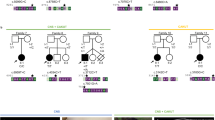
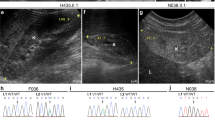
3MC syndrome has been proposed as a unifying term encompassing the overlapping Carnevale, Mingarelli, Malpuech and Michels syndromes. These rare autosomal recessive disorders exhibit a spectrum of developmental features, including characteristic facial dysmorphism, cleft lip and/or palate, craniosynostosis, learning disability and genital, limb and vesicorenal anomalies. Here we studied 11 families with 3MC syndrome and identified two mutated genes, COLEC11 and MASP1, both of which encode proteins in the lectin complement pathway (collectin kidney 1 (CL-K1) and MASP-1 and MASP-3, respectively). CL-K1 is highly expressed in embryonic murine craniofacial cartilage, heart, bronchi, kidney and vertebral bodies. Zebrafish morphants for either gene develop pigmentary defects and severe craniofacial abnormalities. Finally, we show that CL-K1 serves as a guidance cue for neural crest cell migration. Together, these findings demonstrate a role for complement pathway factors in fundamental developmental processes and in the etiology of 3MC syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Change history
09 March 2016
In the version of this article initially published, the authors neglected to acknowledge funding from the National Institute for Health Research Biomedical Research Centre at Great Ormond Street Hospital for Children NHS Foundation Trust and University College London to author Philip L. Beales. The error has been corrected in the HTML and PDF versions of the article.
References
Carnevale, F., Krajewska, G., Fischetto, R., Greco, M.G. & Bonvino, A. Ptosis of eyelids, strabismus, diastasis recti, hip defect, cryptorchidism, and developmental delay in two sibs. Am. J. Med. Genet. 33, 186–189 (1989).
Mingarelli, R., Castriota Scanderbeg, A. & Dallapiccola, B. Two sisters with a syndrome of ocular, skeletal, and abdominal abnormalities (OSA syndrome). J. Med. Genet. 33, 884–886 (1996).
Malpuech, G., Demeocq, F., Palcoux, J.B. & Vanlieferinghen, P. A previously undescribed autosomal recessive multiple congenital anomalies/mental retardation (MCA/MR) syndrome with growth failure, lip/palate cleft(s), and urogenital anomalies. Am. J. Med. Genet. 16, 475–480 (1983).
Michels, V.V., Hittner, H.M. & Beaudet, A.L. A clefting syndrome with ocular anterior chamber defect and lid anomalies. J. Pediatr. 93, 444–446 (1978).
Al Kaissi, A. et al. Asymmetrical skull, ptosis, hypertelorism, high nasal bridge, clefting, umbilical anomalies, and skeletal anomalies in sibs: is Carnevale syndrome a separate entity? Am. J. Med. Genet. A. 143, 349–354 (2007).
Leal, G.F., Silva, E.O., Duarte, A.R. & Campos, J.F. Blepharophimosis, blepharoptosis, defects of the anterior chamber of the eye, caudal appendage, radioulnar synostosis, hearing loss and umbilical anomalies in sibs: 3MC syndrome? Am. J. Med. Genet. A. 146A, 1059–1062 (2008).
Priolo, M. et al. Malpuech syndrome: broadening the clinical spectrum and molecular analysis by array-CGH. Eur. J. Med. Genet. 50, 139–143 (2007).
Keshi, H. et al. Identification and characterization of a novel human collectin CL-K1. Microbiol. Immunol. 50, 1001–1013 (2006).
Møller-Kristensen, M., Thiel, S., Sjoholm, A., Matsushita, M. & Jensenius, J.C. Cooperation between MASP-1 and MASP-2 in the generation of C3 convertase through the MBL pathway. Int. Immunol. 19, 141–149 (2007).
Dahl, M.R. et al. MASP-3 and its association with distinct complexes of the mannan-binding lectin complement activation pathway. Immunity 15, 127–135 (2001).
Leal, G.F. & Baptista, E.V. Three additional cases of the Michels syndrome. Am. J. Med. Genet. A. 143A, 2747–2750 (2007).
Hansen, S. et al. Collectin 11 (CL-11, CL-K1) is a MASP-1/3-associated plasma collectin with microbial-binding activity. J. Immunol. 185, 6096–6104 (2010).
Delalande, J.M. et al. The receptor tyrosine kinase RET regulates hindgut colonization by sacral neural crest cells. Dev. Biol. 313, 279–292 (2008).
Sirmaci, A. et al. MASP1 mutations in patients with facial, umbilical, coccygeal, and auditory findings of Carnevale, Malpuech, OSA, and Michels syndromes. Am. J. Hum. Genet. 87, 679–686 (2010).
Ezekowitz, R.A., Day, L.E. & Herman, G.A. A human mannose-binding protein is an acute-phase reactant that shares sequence homology with other vertebrate lectins. J. Exp. Med. 167, 1034–1046 (1988).
Cortesio, C.L. & Jiang, W. Mannan-binding lectin-associated serine protease 3 cleaves synthetic peptides and insulin-like growth factor-binding protein 5. Arch. Biochem. Biophys. 449, 164–170 (2006).
Degn, S.E., Thiel, S. & Jensenius, J.C. New perspectives on mannan-binding lectin-mediated complement activation. Immunobiology 212, 301–311 (2007).
Degn, S.E. et al. MAp44, a human protein associated with pattern recognition molecules of the complement system and regulating the lectin pathway of complement activation. J. Immunol. 183, 7371–7378 (2009).
Skjoedt, M.O. et al. A novel mannose-binding lectin/ficolin-associated protein is highly expressed in heart and skeletal muscle tissues and inhibits complement activation. J. Biol. Chem. 285, 8234–8243 (2010).
Zundel, S. et al. Characterization of recombinant mannan-binding lectin-associated serine protease (MASP)-3 suggests an activation mechanism different from that of MASP-1 and MASP-2. J. Immunol. 172, 4342–4350 (2004).
Lynch, N.J. et al. Composition of the lectin pathway of complement in Gallus gallus: absence of mannan-binding lectin-associated serine protease-1 in birds. J. Immunol. 174, 4998–5006 (2005).
Theveneau, E. et al. Collective chemotaxis requires contact-dependent cell polarity. Dev. Cell 19, 39–53 (2010).
Westerfield, M. The Zebrafish Book. A Guide for the Laboratory Use of Zebrafish (Danio rerio) (University of Oregon Press, Portland, Oregon, USA, 1995).
Weinberg, E.S. et al. Developmental regulation of zebrafish MyoD in wild-type, no tail and spadetail embryos. Development 122, 271–280 (1996).
Pearson, C.G., Osborn, D.P., Giddings, T.H. Jr., Beales, P.L. & Winey, M. Basal body stability and ciliogenesis requires the conserved component Poc1. J. Cell Biol. 187, 905–920 (2009).
Wiggins, H. & Rappoport, J. An agarose spot assay for chemotactic invasion. Biotechniques 48, 121–124 (2010).
Delalande, J.M. et al. The receptor tyrosine kinase RET regulates hindgut colonization by sacral neural crest cells. Dev. Biol. 313, 279–292 (2008).
Acknowledgements
This work was supported in part by grants from NEWLIFE (P.L.B., A.D.-F. and C.R.), the Wellcome Trust (P.L.B.), Dubai Harvard Foundation for Medical Research (F.S.A.), the University Hospital of Bordeaux (C.R.), the UK Medical Research Council (A.W.) and EU-FP7 (201804-EUCILIA) (V.H.-H., D.J. and D.P.S.O.). This research was supported by the National Institute for Health Research Biomedical Research Centre at Great Ormond Street Hospital for Children NHS Foundation Trust and University College London (P.L.B.). P.L.B. is a Wellcome Trust Senior Research Fellow.
Author information
Authors and Affiliations
Contributions
C.R., A.D.-F. and D.P.S.O. planned and carried out experiments, analyzed data and co-wrote the manuscript. E.C., V.H.-H. and A.W. carried out experiments and analyzed data. H.S., J.K. and D.J. carried out experiments. A.A.K., G.F.L., B.D., F.C. and M.L. clinically ascertained patients and samples. M.B.-G. clinically ascertained patients and samples and planned the study. R.H. clinically ascertained patients and samples, planned the study, analyzed data and edited the manuscript. P.S. provided samples and analyzed data. A.J.B. and H.P. planned and carried out experiments and analyzed data. F.S.A. planned the study, ascertained samples, carried out experiments, analyzed data and edited the manuscript. P.L.B. planned and supervised the study, analyzed data, and co-wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 1 and 2 (PDF 3813 kb)
Rights and permissions
About this article
Cite this article
Rooryck, C., Diaz-Font, A., Osborn, D. et al. Mutations in lectin complement pathway genes COLEC11 and MASP1 cause 3MC syndrome. Nat Genet 43, 197–203 (2011). https://doi.org/10.1038/ng.757
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.757
This article is cited by
-
The C-type lectin COLEC10 is predominantly produced by hepatic stellate cells and involved in the pathogenesis of liver fibrosis
Cell Death & Disease (2023)
-
Effect of anemoside B4 on milk whey in clinical mastitis-affected cows elucidated using tandem mass tag (TMT)-based quantitative proteomics
Scientific Reports (2022)
-
Non-coding de novo mutations in chromatin interactions are implicated in autism spectrum disorder
Molecular Psychiatry (2022)
-
Extensive identification of genes involved in congenital and structural heart disorders and cardiomyopathy
Nature Cardiovascular Research (2022)
-
European Society for Immunodeficiencies (ESID) and European Reference Network on Rare Primary Immunodeficiency, Autoinflammatory and Autoimmune Diseases (ERN RITA) Complement Guideline: Deficiencies, Diagnosis, and Management
Journal of Clinical Immunology (2020)