Abstract
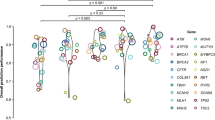
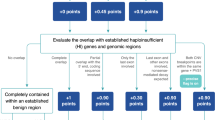
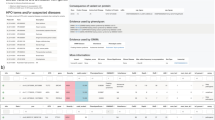
Variant pathogenicity classifiers such as SIFT, PolyPhen-2, CADD, and MetaLR assist in interpretation of the hundreds of rare, missense variants in the typical patient genome by deprioritizing some variants as likely benign. These widely used methods misclassify 26 to 38% of known pathogenic mutations, which could lead to missed diagnoses if the classifiers are trusted as definitive in a clinical setting. We developed M-CAP, a clinical pathogenicity classifier that outperforms existing methods at all thresholds and correctly dismisses 60% of rare, missense variants of uncertain significance in a typical genome at 95% sensitivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Yang, Y. et al. Clinical whole-exome sequencing for the diagnosis of Mendelian disorders. N. Engl. J. Med. 369, 1502–1511 (2013).
Iglesias, A. et al. The usefulness of whole-exome sequencing in routine clinical practice. Genet. Med. 16, 922–931 (2014).
Lee, H. et al. Clinical exome sequencing for genetic identification of rare Mendelian disorders. J. Am. Med. Assoc. 312, 1880–1887 (2014).
Brownstein, C.A. et al. An international effort towards developing standards for best practices in analysis, interpretation and reporting of clinical genome sequencing results in the CLARITY Challenge. Genome Biol. 15, R53 (2014).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Ng, S.B. et al. Targeted capture and massively parallel sequencing of 12 human exomes. Nature 461, 272–276 (2009).
Simpson, M.A. et al. Mutations in NOTCH2 cause Hajdu–Cheney syndrome, a disorder of severe and progressive bone loss. Nat. Genet. 43, 303–305 (2011).
Ng, S.B. et al. Exome sequencing identifies the cause of a mendelian disorder. Nat. Genet. 42, 30–35 (2010).
Taylor, J.C. et al. Factors influencing success of clinical genome sequencing across a broad spectrum of disorders. Nat. Genet. 47, 717–726 (2015).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012).
Rehm, H.L. et al. ACMG clinical laboratory standards for next-generation sequencing. Genet. Med. 15, 733–747 (2013).
Ng, P.C. & Henikoff, S. SIFT: predicting amino acid changes that affect protein function. Nucleic Acids Res. 31, 3812–3814 (2003).
Adzhubei, I.A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).
Kircher, M. et al. A general framework for estimating the relative pathogenicity of human genetic variants. Nat. Genet. 46, 310–315 (2014).
Dong, C. et al. Comparison and integration of deleteriousness prediction methods for nonsynonymous SNVs in whole exome sequencing studies. Hum. Mol. Genet. 24, 2125–2137 (2015).
Hastie, T., Tibshirani, R. & Friedman, J. Elements of Statistical Learning (Springer, 2003).
Fusi, N., Smith, I., Doench, J. & Listgarten, J. In silico predictive modeling of CRISPR/Cas9 guide efficiency. Preprint at bioRxiv http://dx.doi.org/10.1101/021568 (2015).
Ogutu, J.O., Piepho, H.-P. & Schulz-Streeck, T. A comparison of random forests, boosting and support vector machines for genomic selection. BMC Proc. 5 (Suppl. 3), S11 (2011).
Schwarz, J.M., Cooper, D.N., Schuelke, M. & Seelow, D. MutationTaster2: mutation prediction for the deep-sequencing age. Nat. Methods 11, 361–362 (2014).
Reva, B., Antipin, Y. & Sander, C. Predicting the functional impact of protein mutations: application to cancer genomics. Nucleic Acids Res. 39, e118 (2011).
Shihab, H.A. et al. Predicting the functional, molecular, and phenotypic consequences of amino acid substitutions using hidden Markov models. Hum. Mutat. 34, 57–65 (2013).
Chun, S. & Fay, J.C. Identification of deleterious mutations within three human genomes. Genome Res. 19, 1553–1561 (2009).
Petrovski, S., Wang, Q., Heinzen, E.L., Allen, A.S. & Goldstein, D.B. Genic intolerance to functional variation and the interpretation of personal genomes. PLoS Genet. 9, e1003709 (2013).
Pollard, K.S., Hubisz, M.J., Rosenbloom, K.R. & Siepel, A. Detection of nonneutral substitution rates on mammalian phylogenies. Genome Res. 20, 110–121 (2010).
Siepel, A. et al. Evolutionarily conserved elements in vertebrate, insect, worm, and yeast genomes. Genome Res. 15, 1034–1050 (2005).
Henikoff, S. & Henikoff, J.G. Amino acid substitution matrices from protein blocks. Proc. Natl. Acad. Sci. USA 89, 10915–10919 (1992).
Garber, M. et al. Identifying novel constrained elements by exploiting biased substitution patterns. Bioinformatics 25, i54–i62 (2009).
Davydov, E.V. et al. Identifying a high fraction of the human genome to be under selective constraint using GERP++. PLoS Comput. Biol. 6, e1001025 (2010).
Kuhn, R.M., Haussler, D. & Kent, W.J. The UCSC genome browser and associated tools. Brief. Bioinform. 14, 144–161 (2013).
Stenson, P.D. et al. The Human Gene Mutation Database: building a comprehensive mutation repository for clinical and molecular genetics, diagnostic testing and personalized genomic medicine. Hum. Genet. 133, 1–9 (2014).
Ionita-Laza, I., McCallum, K., Xu, B. & Buxbaum, J.D. A spectral approach integrating functional genomic annotations for coding and noncoding variants. Nat. Genet. 48, 214–220 (2016).
UniProt Consortium. UniProt: a hub for protein information. Nucleic Acids Res. 43, D204–D212 (2015).
Yang, Y. et al. Molecular findings among patients referred for clinical whole-exome sequencing. J. Am. Med. Assoc. 312, 1870–1879 (2014).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
Liu, X., Jian, X. & Boerwinkle, E. dbNSFP: a lightweight database of human nonsynonymous SNPs and their functional predictions. Hum. Mutat. 32, 894–899 (2011).
Liu, X., Jian, X. & Boerwinkle, E. dbNSFP v2.0: a database of human non-synonymous SNVs and their functional predictions and annotations. Hum. Mutat. 34, E2393–E2402 (2013).
Pedregosa, F. et al. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows–Wheeler transform. Bioinformatics 26, 589–595 (2010).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Acknowledgements
We thank the members of the Bejerano laboartory, particularly J. Notwell, S. Chinchali, and J. Birgmeier, for technical advice and helpful discussions. P.D.S. and D.N.C. receive financial support from Qiagen through a license agreement with Cardiff University. We thank the PolyPhen-2, CADD, Eigen, FATHMM, MutationTaster, and MetaLR teams for making their training and testing data readily available. This work was funded in part by the Stanford Pediatrics Department, DARPA, a Packard Foundation Fellowship, and a Microsoft Faculty Fellowship to G.B.
Author information
Authors and Affiliations
Contributions
K.A.J., A.M.W., M.J.B., and G.B. designed the study and analyzed results. K.A.J. and M.J.B. implemented the model and performed the experiments. K.A.J., A.M.W., and H.G. wrote software tools that were used for analysis. P.D.S. and D.N.C. curated the HGMD data and provided feedback. J.A.B. provided patient exome cases and feedback. K.A.J., A.M.W., and G.B. wrote the manuscript. All authors reviewed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4, 6, 9 and 10. (PDF 486 kb)
Supplementary Table 5
M-CAP scores for disease-causing mutations found in BRCA1, BRCA2, CFTR and MLL2. (XLSX 43 kb)
Supplementary Table 7
Clinical phenotypes for case study patients. (XLSX 73 kb)
Supplementary Table 8
Rare missense variants in case study patients. (XLSX 150 kb)
Rights and permissions
About this article
Cite this article
Jagadeesh, K., Wenger, A., Berger, M. et al. M-CAP eliminates a majority of variants of uncertain significance in clinical exomes at high sensitivity. Nat Genet 48, 1581–1586 (2016). https://doi.org/10.1038/ng.3703
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3703
This article is cited by
-
A novel missense COL9A3 variant in a pedigree with multiple lumbar disc herniation
Journal of Orthopaedic Surgery and Research (2024)
-
A loss-of-function variant in ZCWPW1 causes human male infertility with sperm head defect and high DNA fragmentation
Reproductive Health (2024)
-
MAGPIE: accurate pathogenic prediction for multiple variant types using machine learning approach
Genome Medicine (2024)
-
Explicable prioritization of genetic variants by integration of rule-based and machine learning algorithms for diagnosis of rare Mendelian disorders
Human Genomics (2024)
-
Genetic architecture and biology of youth-onset type 2 diabetes
Nature Metabolism (2024)