Abstract
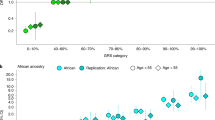
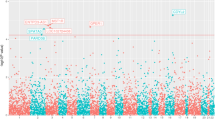
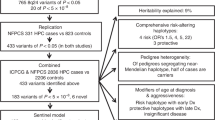
We report targeted sequencing of 63 known prostate cancer risk regions in a multi-ancestry study of 9,237 men and use the data to explore the contribution of low-frequency variation to disease risk. We show that SNPs with minor allele frequencies (MAFs) of 0.1–1% explain a substantial fraction of prostate cancer risk in men of African ancestry. We estimate that these SNPs account for 0.12 (standard error (s.e.) = 0.05) of variance in risk (∼42% of the variance contributed by SNPs with MAF of 0.1–50%). This contribution is much larger than the fraction of neutral variation due to SNPs in this class, implying that natural selection has driven down the frequency of many prostate cancer risk alleles; we estimate the coupling between selection and allelic effects at 0.48 (95% confidence interval [0.19, 0.78]) under the Eyre-Walker model. Our results indicate that rare variants make a disproportionate contribution to genetic risk for prostate cancer and suggest the possibility that rare variants may also have an outsize effect on other common traits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Siegel, R., Ma, J., Zou, Z. & Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 64, 9–29 (2014).
Lin, K., Croswell, J.M., Koenig, H., Lam, C. & Maltz, A. in Prostate-Specific Antigen–Based Screening for Prostate Cancer: An Evidence Update for the U.S. Preventive Services Task Force (Agency for Healthcare Research and Quality, 2011).
Melnikow, J., LeFevre, M., Wilt, T.J. & Moyer, V.A. Counterpoint: randomized trials provide the strongest evidence for clinical guidelines: The US Preventive Services Task Force and Prostate Cancer Screening. Med. Care 51, 301–303 (2013).
Gomella, L.G. et al. Screening for prostate cancer: the current evidence and guidelines controversy. Can. J. Urol. 18, 5875–5883 (2011).
Lichtenstein, P. et al. Environmental and heritable factors in the causation of cancer—analyses of cohorts of twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 343, 78–85 (2000).
Hjelmborg, J.B. et al. The heritability of prostate cancer in the Nordic Twin Study of Cancer. Cancer Epidemiol. Biomarkers Prev. 23, 2303–2310 (2014).
Al Olama, A.A. et al. Multiple loci on 8q24 associated with prostate cancer susceptibility. Nat. Genet. 41, 1058–1060 (2009).
Eeles, R.A. et al. Identification of seven new prostate cancer susceptibility loci through a genome-wide association study. Nat. Genet. 41, 1116–1121 (2009).
Eeles, R.A. et al. Multiple newly identified loci associated with prostate cancer susceptibility. Nat. Genet. 40, 316–321 (2008).
Eeles, R.A. et al. Identification of 23 new prostate cancer susceptibility loci using the iCOGS custom genotyping array. Nat. Genet. 45, 385–391 (2013).
Kote-Jarai, Z. et al. Seven prostate cancer susceptibility loci identified by a multi-stage genome-wide association study. Nat. Genet. 43, 785–791 (2011).
Schumacher, F.R. et al. Genome-wide association study identifies new prostate cancer susceptibility loci. Hum. Mol. Genet. 20, 3867–3875 (2011).
Amundadottir, L.T. et al. A common variant associated with prostate cancer in European and African populations. Nat. Genet. 38, 652–658 (2006).
Gudmundsson, J. et al. Genome-wide association study identifies a second prostate cancer susceptibility variant at 8q24. Nat. Genet. 39, 631–637 (2007).
Gudmundsson, J. et al. Genome-wide association and replication studies identify four variants associated with prostate cancer susceptibility. Nat. Genet. 41, 1122–1126 (2009).
Gudmundsson, J. et al. Common sequence variants on 2p15 and Xp11.22 confer susceptibility to prostate cancer. Nat. Genet. 40, 281–283 (2008).
Gudmundsson, J. et al. Two variants on chromosome 17 confer prostate cancer risk, and the one in TCF2 protects against type 2 diabetes. Nat. Genet. 39, 977–983 (2007).
Sun, J. et al. Evidence for two independent prostate cancer risk-associated loci in the HNF1B gene at 17q12. Nat. Genet. 40, 1153–1155 (2008).
Thomas, G. et al. Multiple loci identified in a genome-wide association study of prostate cancer. Nat. Genet. 40, 310–315 (2008).
Yeager, M. et al. Genome-wide association study of prostate cancer identifies a second risk locus at 8q24. Nat. Genet. 39, 645–649 (2007).
Duggan, D. et al. Two genome-wide association studies of aggressive prostate cancer implicate putative prostate tumor suppressor gene DAB2IP. J. Natl. Cancer Inst. 99, 1836–1844 (2007).
Haiman, C.A. et al. Genome-wide association study of prostate cancer in men of African ancestry identifies a susceptibility locus at 17q21. Nat. Genet. 43, 570–573 (2011).
Takata, R. et al. Genome-wide association study identifies five new susceptibility loci for prostate cancer in the Japanese population. Nat. Genet. 42, 751–754 (2010).
Al Olama, A.A. et al. A meta-analysis of 87,040 individuals identifies 23 new susceptibility loci for prostate cancer. Nat. Genet. 46, 1103–1109 (2014).
Witte, J.S., Visscher, P.M. & Wray, N.R. The contribution of genetic variants to disease depends on the ruler. Nat. Rev. Genet. 15, 765–776 (2014).
Gudmundsson, J. et al. A study based on whole-genome sequencing yields a rare variant at 8q24 associated with prostate cancer. Nat. Genet. 44, 1326–1329 (2012).
Cropp, C.D. et al. 8q24 risk alleles and prostate cancer in African-Barbadian men. Prostate 74, 1579–1588 (2014).
Hazelett, D.J., Coetzee, S.G. & Coetzee, G.A. A rare variant, which destroys a FoxA1 site at 8q24, is associated with prostate cancer risk. Cell Cycle 12, 379–380 (2013).
Haiman, C.A. et al. Multiple regions within 8q24 independently affect risk for prostate cancer. Nat. Genet. 39, 638–644 (2007).
Barrett, J.C., Fry, B., Maller, J. & Daly, M.J. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265 (2005).
Lindblad-Toh, K. et al. A high-resolution map of human evolutionary constraint using 29 mammals. Nature 478, 476–482 (2011).
Gnirke, A. et al. Solution hybrid selection with ultra-long oligonucleotides for massively parallel targeted sequencing. Nat. Biotechnol. 27, 182–189 (2009).
Rohland, N. & Reich, D. Cost-effective, high-throughput DNA sequencing libraries for multiplexed target capture. Genome Res. 22, 939–946 (2012).
Li, Y., Sidore, C., Kang, H.M., Boehnke, M. & Abecasis, G.R. Low-coverage sequencing: implications for design of complex trait association studies. Genome Res. 21, 940–951 (2011).
Lee, S.H. et al. Estimation of SNP heritability from dense genotype data. Am. J. Hum. Genet. 93, 1151–1155 (2013).
Yang, J., Lee, S.H., Goddard, M.E. & Visscher, P.M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Gusev, A. et al. Quantifying missing heritability at known GWAS loci. PLoS Genet. 9, e1003993 (2013).
Lee, S.H., Yang, J., Goddard, M.E., Visscher, P.M. & Wray, N.R. Estimation of pleiotropy between complex diseases using single-nucleotide polymorphism–derived genomic relationships and restricted maximum likelihood. Bioinformatics 28, 2540–2542 (2012).
Eyre-Walker, A. Genetic architecture of a complex trait and its implications for fitness and genome-wide association studies. Proc. Natl. Acad. Sci. USA 107, 1752–1756 (2010).
Kichaev, G. et al. Integrating functional data to prioritize causal variants in statistical fine-mapping studies. PLoS Genet. 10, e1004722 (2014).
Hazelett, D.J. et al. Comprehensive functional annotation of 77 prostate cancer risk loci. PLoS Genet. 10, e1004102 (2014).
Bhatia, G. et al. Haplotypes of common SNPs can explain missing heritability of complex diseases. bioRxiv 10.1101/022418 (12 July 2015).
Huffman, J.E. et al. Rare and low-frequency variants and their association with plasma levels of fibrinogen, FVII, FVIII, and vWF. Blood 126, e19–e29 (2015).
Peloso, G.M. et al. Association of low-frequency and rare coding-sequence variants with blood lipids and coronary heart disease in 56,000 whites and blacks. Am. J. Hum. Genet. 94, 223–232 (2014).
Lange, L.A. et al. Whole-exome sequencing identifies rare and low-frequency coding variants associated with LDL cholesterol. Am. J. Hum. Genet. 94, 233–245 (2014).
Service, S.K. et al. Re-sequencing expands our understanding of the phenotypic impact of variants at GWAS loci. PLoS Genet. 10, e1004147 (2014).
Kolonel, L.N. et al. A multiethnic cohort in Hawaii and Los Angeles: baseline characteristics. Am. J. Epidemiol. 151, 346–357 (2000).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
DePristo, M.A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Browning, B.L. & Browning, S.R. A unified approach to genotype imputation and haplotype-phase inference for large data sets of trios and unrelated individuals. Am. J. Hum. Genet. 84, 210–223 (2009).
Browning, B.L. & Browning, S.R. A fast, powerful method for detecting identity by descent. Am. J. Hum. Genet. 88, 173–182 (2011).
Baran, Y., Quintela, I., Carracedo, Á., Pasaniuc, B. & Halperin, E. Enhanced localization of genetic samples through linkage-disequilibrium correction. Am. J. Hum. Genet. 92, 882–894 (2013).
Chang, C.C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. GigaScience 4, 7 (2015).
Willer, C.J., Li, Y. & Abecasis, G.R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Oehlert, G.W. A note on the delta method. Am. Stat. 46, 27–29 (1992).
Speed, D., Hemani, G., Johnson, M.R. & Balding, D.J. Improved heritability estimation from genome-wide SNPs. Am. J. Hum. Genet. 91, 1011–1021 (2012).
Yang, L. et al. Polygenic transmission and complex neuro developmental network for attention deficit hyperactivity disorder: genome-wide association study of both common and rare variants. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 162B, 419–430 (2013).
Takahata, N. Allelic genealogy and human evolution. Mol. Biol. Evol. 10, 2–22 (1993).
Lohmueller, K.E. The impact of population demography and selection on the genetic architecture of complex traits. PLoS Genet. 10, e1004379 (2014).
Acknowledgements
This work is supported in part by the US National Institutes of Health (R01 CA165862, U19 CA148537, UM1 CA164973, RC2 CA148085, U01 CA1326792, R21 CA182821 and U01 CA188392). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. Many of the risk regions examined were discovered through contributions from: P. Hall (COGS), D.F.E., P. Pharoah, K. Michailidou, M.K. Bolla and Q. Wang (BCAC), A. Berchuck (OCAC), R.A.E., D.F.E., A.A. Al Olama, Z.K.-J. and S. Benlloch (PRACTICAL), G. Chenevix-Trench, A. Antoniou, L. McGuffog, F. Couch and K. Offit (CIMBA), J. Dennis, A.M. Dunning, A. Lee, E. Dicks, C. Luccarini and the staff of the Centre for Genetic Epidemiology Laboratory, J. Benitez, A. Gonzalez-Neira and the staff of the CNIO genotyping unit, J. Simard, D.V.C. Tessier, F. Bacot, D. Vincent, S. LaBoissière, F. Robidoux and the staff of the McGill University and Génome Québec Innovation Centre, S.E. Bojesen, S.F. Nielsen, B.G. Nordestgaard and the staff of the Copenhagen DNA laboratory, and J.M. Cunningham, S.A. Windebank, C.A. Hilker, J. Meyer and the staff of the Mayo Clinic Genotyping Core Facility. Funding for the iCOGS infrastructure came from the European Community's Seventh Framework Programme under grant agreement 223175 (HEALTH-F2-2009-223175) (COGS), Cancer Research UK (C1287/A10118, C1287/A 10710, C12292/A11174, C1281/A12014, C5047/A8384, C5047/A15007, C5047/A10692 and C8197/A16565), the US National Institutes of Health (CA128978) and Post-Cancer GWAS initiative (1U19 CA148537, 1U19 CA148065 and 1U19 CA148112–GAME-ON initiative), the US Department of Defense (W81XWH-10-1-0341), the Canadian Institutes of Health Research (CIHR) for the CIHR Team in Familial Risks of Breast Cancer, the Komen Foundation for the Cure, the Breast Cancer Research Foundation and the Ovarian Cancer Research Fund. D.R. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Consortia
Contributions
N.R., C.A.H. and D.R. defined the regions of interest. N.R., S.M. and D.R. designed the in-solution capture reagent. N.R., A.A. and D.Q. prepared libraries. N.R. performed capture and quality control sequencing. N.R., A.T. and S.M. performed sequence analyses. N.M. performed statistical analyses and simulations. K.A.R., A.T., H.L., A.S., X.S., Z.K.-J., D.F.E., R.A.E., the PRACTICAL consortium, L.L.M., A.L., D.S., S.W., D.V.C. and B.H. generated data and analysis tools. C.A.H., B.P. and D.R. supervised the work. All authors reviewed, revised and wrote feedback for the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
A full list of members and affiliations appears in the Supplementary Note.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–24, Supplementary Tables 2–36, 38 and 43–45, and Supplementary Note. (PDF 3934 kb)
Supplementary Dataset 1
Linkage-disequilibrium maps for the targeted loci (PDF 9178 kb)
Supplementary Table 1
Boundaries and capture information for the targeted loci. (XLSX 75 kb)
Supplementary Table 37
Association meta-analysis summary. (XLSX 48 kb)
Supplementary Table 39
Association analysis summary for the African-ancestry group. (XLSX 42 kb)
Supplementary Table 40
Association analysis summary for the European-ancestry group. (XLSX 40 kb)
Supplementary Table 41
Association analysis summary for the Japanese-ancestry group. (XLSX 42 kb)
Supplementary Table 42
Association analysis summary for the Latino-ancestry group. (XLSX 42 kb)
Rights and permissions
About this article
Cite this article
Mancuso, N., Rohland, N., Rand, K. et al. The contribution of rare variation to prostate cancer heritability. Nat Genet 48, 30–35 (2016). https://doi.org/10.1038/ng.3446
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3446
This article is cited by
-
A method for an unbiased estimate of cross-ancestry genetic correlation using individual-level data
Nature Communications (2023)
-
Genes encoding agrin (AGRN) and neurotrypsin (PRSS12) are associated with muscle mass, strength and plasma C-terminal agrin fragment concentration
GeroScience (2023)
-
Quantifying genetic heterogeneity between continental populations for human height and body mass index
Scientific Reports (2021)
-
Identification of low-frequency variants of UGT1A3 associated with bladder cancer risk by next-generation sequencing
Oncogene (2021)
-
Rare variant analysis in eczema identifies exonic variants in DUSP1, NOTCH4 and SLC9A4
Nature Communications (2021)