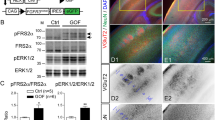
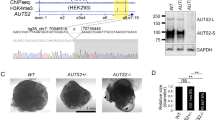
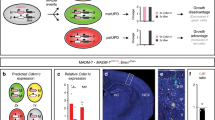
Abstract
The regulated proliferation and differentiation of neural stem cells before the generation and migration of neurons in the cerebral cortex are central aspects of mammalian development. Periventricular neuronal heterotopia, a specific form of mislocalization of cortical neurons, can arise from neuronal progenitors that fail to negotiate aspects of these developmental processes. Here we show that mutations in genes encoding the receptor-ligand cadherin pair DCHS1 and FAT4 lead to a recessive syndrome in humans that includes periventricular neuronal heterotopia. Reducing the expression of Dchs1 or Fat4 within mouse embryonic neuroepithelium increased progenitor cell numbers and reduced their differentiation into neurons, resulting in the heterotopic accumulation of cells below the neuronal layers in the neocortex, reminiscent of the human phenotype. These effects were countered by concurrent knockdown of Yap, a transcriptional effector of the Hippo signaling pathway. These findings implicate Dchs1 and Fat4 upstream of Yap as key regulators of mammalian neurogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Accessions
NCBI Reference Sequence
Protein Data Bank
References
Götz, M. & Huttner, W.B. The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 6, 777–788 (2005).
Sidman, R.L. & Rakic, P. Neuronal migration, with special reference to developing human brain: a review. Brain Res. 62, 1–35 (1973).
Métin, C., Vallee, R.B., Rakic, P. & Bhide, P.G. Modes and mishaps of neuronal migration in the mammalian brain. J. Neurosci. 28, 11746–11752 (2008).
Fox, J.W. et al. Mutations in filamin 1 prevent migration of cerebral cortical neurons in human periventricular heterotopia. Neuron 21, 1315–1325 (1998).
Manzini, M.C. & Walsh, C.A. What disorders of cortical development tell us about the cortex: one plus one does not always make two. Curr. Opin. Genet. Dev. 21, 333–339 (2011).
Sarkisian, M.R., Bartley, C.M. & Rakic, P. Trouble making the first move: interpreting arrested neuronal migration in the cerebral cortex. Trends Neurosci. 31, 54–61 (2008).
Carabalona, A. et al. A glial origin for periventricular nodular heterotopia caused by impaired expression of Filamin-A. Hum. Mol. Genet. 21, 1004–1017 (2012).
Ferland, R.J. et al. Disruption of neural progenitors along the ventricular and subventricular zones in periventricular heterotopia. Hum. Mol. Genet. 18, 497–516 (2009).
Kriegstein, A. & Alvarez-Buylla, A. The glial nature of embryonic and adult neural stem cells. Annu. Rev. Neurosci. 32, 149–184 (2009).
Hansen, D.V., Lui, J.H., Parker, P.R. & Kriegstein, A.R. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature 464, 554–561 (2010).
Fietz, S.A. et al. OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling. Nat. Neurosci. 13, 690–699 (2010).
Chenn, A., Zhang, Y.A., Chang, B.T. & McConnell, S.K. Intrinsic polarity of mammalian neuroepithelial cells. Mol. Cell. Neurosci. 11, 183–193 (1998).
Chenn, A. & McConnell, S.K. Cleavage orientation and the asymmetric inheritance of Notch1 immunoreactivity in mammalian neurogenesis. Cell 82, 631–641 (1995).
Junghans, D. et al. β-catenin–mediated cell-adhesion is vital for embryonic forebrain development. Dev. Dyn. 233, 528–539 (2005).
Kadowaki, M. et al. N-cadherin mediates cortical organization in the mouse brain. Dev. Biol. 304, 22–33 (2007).
Zechner, D. et al. β-catenin signals regulate cell growth and the balance between progenitor cell expansion and differentiation in the nervous system. Dev. Biol. 258, 406–418 (2003).
Machon, O. et al. Role of β-catenin in the developing cortical and hippocampal neuroepithelium. Neuroscience 122, 129–143 (2003).
Cappello, S. et al. The Rho-GTPase cdc42 regulates neural progenitor fate at the apical surface. Nat. Neurosci. 9, 1099–1107 (2006).
Cappello, S. et al. A radial glia-specific role of RhoA in double cortex formation. Neuron 73, 911–924 (2012).
Lien, W.H. et al. αE-catenin controls cerebral cortical size by regulating the hedgehog signaling pathway. Science 311, 1609–1612 (2006).
Kim, S. et al. The apical complex couples cell fate and cell survival to cerebral cortical development. Neuron 66, 69–84 (2010).
Costa, M.R. et al. Par-complex proteins promote proliferative progenitor divisions in the developing mouse cerebral cortex. Development 135, 11–22 (2008).
Bultje, R.S. et al. Mammalian Par3 regulates progenitor cell asymmetric division via notch signaling in the developing neocortex. Neuron 63, 189–202 (2009).
Ishiuchi, T. et al. Mammalian Fat and Dachsous cadherins regulate apical membrane organization in the embryonic cerebral cortex. J. Cell Biol. 185, 959–967 (2009).
van Maldergem, L. et al. Mental retardation with blepharo-naso-facial abnormalities and hand malformations: a new syndrome? Clin. Genet. 41, 22–24 (1992).
Mansour, S. et al. Van Maldergem syndrome: further characterisation and evidence for neuronal migration abnormalities and autosomal recessive inheritance. Eur. J. Hum. Genet. 20, 1024–1031 (2012).
Neuhann, T.M. et al. A further patient with van Maldergem syndrome. Eur. J. Med. Genet. 55, 423–428 (2012).
Tanoue, T. & Takeichi, M. New insights into Fat cadherins. J. Cell Sci. 118, 2347–2353 (2005).
Nagar, B., Overduin, M., Ikura, M. & Rini, J.M. Structural basis of calcium-induced E-cadherin rigidification and dimerization. Nature 380, 360–364 (1996).
Pilichou, K. et al. Mutations in desmoglein-2 gene are associated with arrhythmogenic right ventricular cardiomyopathy. Circulation 113, 1171–1179 (2006).
Dibbens, L.M. et al. X-linked protocadherin 19 mutations cause female-limited epilepsy and cognitive impairment. Nat. Genet. 40, 776–781 (2008).
Saburi, S. et al. Loss of Fat4 disrupts PCP signaling and oriented cell division and leads to cystic kidney disease. Nat. Genet. 40, 1010–1015 (2008).
Mao, Y. et al. Characterization of a Dchs1 mutant mouse reveals requirements for Dchs1-Fat4 signaling during mammalian development. Development 138, 947–957 (2011).
Mahoney, P.A. et al. The fat tumor suppressor gene in Drosophila encodes a novel member of the cadherin gene superfamily. Cell 67, 853–868 (1991).
Clark, H.F. et al. Dachsous encodes a member of the cadherin superfamily that controls imaginal disc morphogenesis in Drosophila. Genes Dev. 9, 1530–1542 (1995).
Katoh, Y. & Katoh, M. Comparative integromics on FAT1, FAT2, FAT3 and FAT4. Int. J. Mol. Med. 18, 523–528 (2006).
Barak, T. et al. Recessive LAMC3 mutations cause malformations of occipital cortical development. Nat. Genet. 43, 590–594 (2011).
Feng, Y. et al. Filamin A (FLNA) is required for cell-cell contact in vascular development and cardiac morphogenesis. Proc. Natl. Acad. Sci. USA 103, 19836–19841 (2006).
Hart, A.W. et al. Cardiac malformations and midline skeletal defects in mice lacking filamin A. Hum. Mol. Genet. 15, 2457–2467 (2006).
Corbo, J.C. et al. Doublecortin is required in mice for lamination of the hippocampus but not the neocortex. J. Neurosci. 22, 7548–7557 (2002).
Bai, J. et al. RNAi reveals doublecortin is required for radial migration in rat neocortex. Nat. Neurosci. 6, 1277–1283 (2003).
Kerjan, G. & Gleeson, J.G. Genetic mechanisms underlying abnormal neuronal migration in classical lissencephaly. Trends Genet. 23, 623–630 (2007).
Ramos, R.L., Bai, J. & LoTurco, J.J. Heterotopia formation in rat but not mouse neocortex after RNA interference knockdown of DCX. Cereb. Cortex 16, 1323–1331 (2006).
Englund, C. et al. Pax6, Tbr2, and Tbr1 are expressed sequentially by radial glia, intermediate progenitor cells, and postmitotic neurons in developing neocortex. J. Neurosci. 25, 247–251 (2005).
Matakatsu, H. & Blair, S.S. Separating the adhesive and signaling functions of the Fat and Dachsous protocadherins. Development 133, 2315–2324 (2006).
Matakatsu, H. & Blair, S.S. Separating planar cell polarity and Hippo pathway activities of the protocadherins Fat and Dachsous. Development 139, 1498–1508 (2012).
Adler, P.N., Charlton, J. & Liu, J. Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype by altering frizzled signaling. Development 125, 959–968 (1998).
Ishikawa, H.O., Takeuchi, H., Haltiwanger, R.S. & Irvine, K.D. Four-jointed is a Golgi kinase that phosphorylates a subset of cadherin domains. Science 321, 401–404 (2008).
Simon, M.A., Xu, A., Ishikawa, H.O. & Irvine, K.D. Modulation of fat:dachsous binding by the cadherin domain kinase four-jointed. Curr. Biol. 20, 811–817 (2010).
Brittle, A.L. et al. Four-jointed modulates growth and planar polarity by reducing the affinity of dachsous for fat. Curr. Biol. 20, 803–810 (2010).
Cho, E. et al. Delineation of a Fat tumor suppressor pathway. Nat. Genet. 38, 1142–1150 (2006).
Dong, J. et al. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell 130, 1120–1133 (2007).
Van Hateren, N.J. et al. FatJ acts via the Hippo mediator Yap1 to restrict the size of neural progenitor cell pools. Development 138, 1893–1902 (2011).
Kawamori, H. et al. Fat/Hippo pathway regulates the progress of neural differentiation signaling in the Drosophila optic lobe. Dev. Growth Differ. 53, 653–667 (2011).
Zhao, B. et al. A coordinated phosphorylation by Lats and CK1 regulates YAP stability through SCF(β-TRCP). Genes Dev. 24, 72–85 (2010).
Li, Y. et al. Genome-wide analysis of N1ICD/RBPJ targets in vivo reveals direct transcriptional regulation of Wnt, SHH, and hippo pathway effectors by Notch1. Stem Cells 30, 741–752 (2012).
Gleeson, J.G. et al. Doublecortin, a brain-specific gene mutated in human X-linked lissencephaly and double cortex syndrome, encodes a putative signaling protein. Cell 92, 63–72 (1998).
Thomas, C. & Strutt, D. The roles of the cadherins Fat and Dachsous in planar polarity specification in Drosophila. Dev. Dyn. 241, 27–39 (2012).
Grusche, F.A., Richardson, H.E. & Harvey, K.F. Upstream regulation of the hippo size control pathway. Curr. Biol. 20, R574–R582 (2010).
Pan, G. et al. Signal transduction by the Fat cytoplasmic domain. Development 140, 831–842 (2013).
Yu, F.X. et al. Regulation of the Hippo-YAP pathway by G-protein–coupled receptor signaling. Cell 150, 780–791 (2012).
Reddy, B.V. & Irvine, K.D. Regulation of Hippo signaling by EGFR-MAPK signaling through Ajuba family proteins. Dev. Cell 24, 459–471 (2013).
Saburi, S., Hester, I., Goodrich, L. & McNeill, H. Functional interactions between Fat family cadherins in tissue morphogenesis and planar polarity. Development 139, 1806–1820 (2012).
Rock, R., Schrauth, S. & Gessler, M. Expression of mouse dchs1, fjx1, and fat-j suggests conservation of the planar cell polarity pathway identified in Drosophila. Dev. Dyn. 234, 747–755 (2005).
Mao, Y., Kucuk, B. & Irvine, K.D. Drosophila lowfat, a novel modulator of Fat signaling. Development 136, 3223–3233 (2009).
Bando, T. et al. Lowfat, a mammalian Lix1 homologue, regulates leg size and growth under the Dachsous/Fat signaling pathway during tissue regeneration. Dev. Dyn. 240, 1440–1453 (2011).
Feng, Y. & Irvine, K.D. Processing and phosphorylation of the Fat receptor. Proc. Natl. Acad. Sci. USA 106, 11989–11994 (2009).
Willecke, M. et al. Boundaries of Dachsous cadherin activity modulate the Hippo signaling pathway to induce cell proliferation. Proc. Natl. Acad. Sci. USA 105, 14897–14902 (2008).
Sopko, R. et al. Phosphorylation of the tumor suppressor fat is regulated by its ligand Dachsous and the kinase discs overgrown. Curr. Biol. 19, 1112–1117 (2009).
Chen, F., Venugopal, V., Murray, B. & Rudenko, G. The structure of neurexin 1α reveals features promoting a role as synaptic organizer. Structure 19, 779–789 (2011).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Li, K. & Stockwell, T.B. VariantClassifier: a hierarchical variant classifier for annotated genomes. BMC Res. Notes 3, 191 (2010).
Saito, T. In vivo electroporation in the embryonic mouse central nervous system. Nat. Protoc. 1, 1552–1558 (2006).
Zhao, B. et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 21, 2747–2761 (2007).
Greer, T. & Dunlap, W.P. Analysis of variance with T measures. Psychol. Methods 2, 200–207 (1997).
Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 6, 65–70 (1979).
Dupont, E. et al. Rapid developmental switch in the mechanisms driving early cortical columnar networks. Nature 439, 79–83 (2006).
Acknowledgements
We thank the families participating in this study for their involvement. This work was supported by funding from Cure Kids New Zealand and the Health Research Council of New Zealand (10/402) (S.P.R.), the Department of Health through the National Institute for Health Research (NIHR) Comprehensive Biomedical Research Centre award to Guy's and St. Thomas' NHS Foundation Trust in partnership with King's College London and King's College Hospital NHS Foundation Trust (M.A.S.), Deutsche Forschungsgemeinschaft:SFB 817, Synergy and the Bundesministerium für Bildung und Forschung (M.G. and S.C.). The human embryonic and fetal material was provided by the joint Medical Research Council (grant G0700089)/Wellcome Trust (grant GR082557) Human Developmental Biology Resource. We thank F. Calzolari for the design of the Yap miRNA, P. Malatesta (Department of Experimental Medicine (DiMES), University of Genoa), K. Guan (Life Sciences Institute, University of Michigan) and S. Piccolo (Department of Molecular Medicine, University of Padua School of Medicine) for sharing plasmids and T. Öztürk and A. Waiser for excellent technical support.
Author information
Authors and Affiliations
Contributions
S.C., M.J.G., S.M., M.G. and S.P.R. conceived and designed the study. S.C., M.J.G., C.B., S.L., M.E., M. Srour, F.F.H., Z.A.J., T.M., N.P., V.M. and M.A.S. performed the experiments and analyzed the data in conjunction with A.J.S.-S., M.A.B., D.M., J.L.M., H.M., M.G. and S.P.R. D.C., T.U., J.T., P.S., N.D.D., L.V.M., T.N., R.N.-E., M. Swinkells, P.T., L.C.W., P.J.G.Z. and S.M. provided reagents, clinical information and analysis of human subjects. S.C., M.J.G., M.G. and S.P.R. wrote the manuscript, which all authors refined and approved.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Tables 1–6 (PDF 5369 kb)
Rights and permissions
About this article
Cite this article
Cappello, S., Gray, M., Badouel, C. et al. Mutations in genes encoding the cadherin receptor-ligand pair DCHS1 and FAT4 disrupt cerebral cortical development. Nat Genet 45, 1300–1308 (2013). https://doi.org/10.1038/ng.2765
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2765
This article is cited by
-
FAT4 overexpression promotes antitumor immunity by regulating the β-catenin/STT3/PD-L1 axis in cervical cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
A Circular RNA Expressed from the FAT3 Locus Regulates Neural Development
Molecular Neurobiology (2023)
-
Neonatal lethality of mouse A/J-7SM consomic strain is caused by an insertion mutation in the Dchs1 gene
Mammalian Genome (2023)
-
Tead transcription factors differentially regulate cortical development
Scientific Reports (2020)
-
Identification of novel loci associated with infant cognitive ability
Molecular Psychiatry (2020)