Abstract
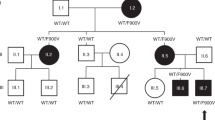
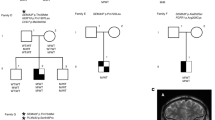
Congenital central hypothyroidism occurs either in isolation or in conjunction with other pituitary hormone deficits. Using exome and candidate gene sequencing, we identified 8 distinct mutations and 2 deletions in IGSF1 in males from 11 unrelated families with central hypothyroidism, testicular enlargement and variably low prolactin concentrations. IGSF1 is a membrane glycoprotein that is highly expressed in the anterior pituitary gland, and the identified mutations impair its trafficking to the cell surface in heterologous cells. Igsf1-deficient male mice show diminished pituitary and serum thyroid-stimulating hormone (TSH) concentrations, reduced pituitary thyrotropin-releasing hormone (TRH) receptor expression, decreased triiodothyronine concentrations and increased body mass. Collectively, our observations delineate a new X-linked disorder in which loss-of-function mutations in IGSF1 cause central hypothyroidism, likely secondary to an associated impairment in pituitary TRH signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Elmlinger, M.W., Kuhnel, W., Lambrecht, H.G. & Ranke, M.B. Reference intervals from birth to adulthood for serum thyroxine (T4), triiodothyronine (T3), free T3, free T4, thyroxine binding globulin (TBG) and thyrotropin (TSH). Clin. Chem. Lab. Med. 39, 973–979 (2001).
Mehta, A. et al. Is the thyrotropin-releasing hormone test necessary in the diagnosis of central hypothyroidism in children. J. Clin. Endocrinol. Metab. 88, 5696–5703 (2003).
van Tijn, D.A., de Vijlder, J.J. & Vulsma, T. Role of the thyrotropin-releasing hormone stimulation test in diagnosis of congenital central hypothyroidism in infants. J. Clin. Endocrinol. Metab. 93, 410–419 (2008).
Faglia, G. et al. Plasma thyrotropin response to thyrotropin-releasing hormone in patients with pituitary and hypothalamic disorders. J. Clin. Endocrinol. Metab. 37, 595–601 (1973).
Crofton, P.M., Tepper, L.A. & Kelnar, C.J. An evaluation of the thyrotrophin-releasing hormone stimulation test in paediatric clinical practice. Horm. Res. 69, 53–59 (2008).
Persani, L. et al. Evidence for the secretion of thyrotropin with enhanced bioactivity in syndromes of thyroid hormone resistance. J. Clin. Endocrinol. Metab. 78, 1034–1039 (1994).
Persani, L., Ferretti, E., Borgato, S., Faglia, G. & Beck-Peccoz, P. Circulating thyrotropin bioactivity in sporadic central hypothyroidism. J. Clin. Endocrinol. Metab. 85, 3631–3635 (2000).
Goede, J. et al. Normative values for testicular volume measured by ultrasonography in a normal population from infancy to adolescence. Horm. Res. Paediatr. 76, 56–64 (2011).
Dattani, M.T., Tziaferi, V. & Hindmarsh, P.C. Evaluation of disordered puberty. in Brook′s Clinical Pediatric Endocrinology (eds. Brook, C.G.D., Clayton, P.E. & Brown, R.S.), 213–238 (Wiley-Blackwell, Oxford, 2009).
Mul, D. et al. Pubertal development in The Netherlands 1965–1997. Pediatr. Res. 50, 479–486 (2001).
Mazzarella, R., Pengue, G., Jones, J., Jones, C. & Schlessinger, D. Cloning and expression of an immunoglobulin superfamily gene (IGSF1) in Xq25. Genomics 48, 157–162 (1998).
Frattini, A., Faranda, S., Redolfi, E., Allavena, P. & Vezzoni, P. Identification and genomic organization of a gene coding for a new member of the cell adhesion molecule family mapping to Xq25. Gene 214, 1–6 (1998).
Robakis, T., Bak, B., Lin, S.H., Bernard, D.J. & Scheiffele, P. An internal signal sequence directs intramembrane proteolysis of a cellular immunoglobulin domain protein. J. Biol. Chem. 283, 36369–36376 (2008).
Su, A.I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl. Acad. Sci. USA 101, 6062–6067 (2004).
Chong, H. et al. Structure and expression of a membrane component of the inhibin receptor system. Endocrinology 141, 2600–2607 (2000).
Bernard, D.J. & Woodruff, T.K. Inhibin binding protein in rats: alternative transcripts and regulation in the pituitary across the estrous cycle. Mol. Endocrinol. 15, 654–667 (2001).
Bernard, D.J., Burns, K.H., Haupt, B., Matzuk, M.M. & Woodruff, T.K. Normal reproductive function in InhBP/p120-deficient mice. Mol. Cell. Biol. 23, 4882–4891 (2003).
Chapman, S.C. & Woodruff, T.K. Modulation of activin signal transduction by inhibin B and inhibin-binding protein (INhBP). Mol. Endocrinol. 15, 668–679 (2001).
Chapman, S.C., Bernard, D.J., Jelen, J. & Woodruff, T.K. Properties of inhibin binding to betaglycan, InhBP/p120 and the activin type II receptors. Mol. Cell. Endocrinol. 196, 79–93 (2002).
Razvi, S. et al. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J. Clin. Endocrinol. Metab. 92, 1715–1723 (2007).
Singh, S. et al. Impact of subclinical thyroid disorders on coronary heart disease, cardiovascular and all-cause mortality: a meta-analysis. Int. J. Cardiol. 125, 41–48 (2008).
Doin, F.C., Rosa-Borges, M., Martins, M.R., Moises, V.A. & Abucham, J. Diagnosis of subclinical central hypothyroidism in patients with hypothalamic-pituitary disease by Doppler echocardiography. Eur. J. Endocrinol. 166, 631–640 (2012).
Fredriks, A.M. et al. Continuing positive secular growth change in The Netherlands 1955–1997. Pediatr. Res. 47, 316–323 (2000).
Freeman, J.V. et al. Cross sectional stature and weight reference curves for the UK, 1990. Arch. Dis. Child. 73, 17–24 (1995).
Cacciari, E. et al. Italian cross-sectional growth charts for height, weight and BMI (2 to 20 yr). J. Endocrinol. Invest. 29, 581–593 (2006).
Cole, T.J. & Roede, M.J. Centiles of body mass index for Dutch children aged 0–20 years in 1980—a baseline to assess recent trends in obesity. Ann. Hum. Biol. 26, 303–308 (1999).
von Schnakenburg, K., Bidlingmaier, F. & Knorr, D. 17-hydroxyprogesterone, androstenedione, and testosterone in normal children and in prepubertal patients with congenital adrenal hyperplasia. Eur. J. Pediatr. 133, 259–267 (1980).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Wu, T.D. & Nacu, S. Fast and SNP-tolerant detection of complex variants and splicing in short reads. Bioinformatics 26, 873–881 (2010).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Bernard, D.J. Both SMAD2 and SMAD3 mediate activin-stimulated expression of the follicle-stimulating hormone β subunit in mouse gonadotrope cells. Mol. Endocrinol. 18, 606–623 (2004).
Lanctôt, C., Gauthier, Y. & Drouin, J. Pituitary homeobox 1 (Ptx1) is differentially expressed during pituitary development. Endocrinology 140, 1416–1422 (1999).
Gaston-Massuet, C. et al. Increased Wingless (Wnt) signaling in pituitary progenitor/stem cells gives rise to pituitary tumors in mice and humans. Proc. Natl. Acad. Sci. USA 108, 11482–11487 (2011).
Siemensma, E.P., de Lind van Wijngaarden, R.F., Otten, B.J., de Jong, F.H. & Hokken-Koelega, A.C. Testicular failure in boys with Prader-Willi syndrome: longitudinal studies of reproductive hormones. J. Clin. Endocrinol. Metab. 97, E452–E459 (2012).
Kevenaar, M.E. et al. Variants in the ACVR1 gene are associated with AMH levels in women with polycystic ovary syndrome. Hum. Reprod. 24, 241–249 (2009).
McGuinness, L. et al. Autosomal dominant growth hormone deficiency disrupts secretory vesicles in vitro and in vivo in transgenic mice. Endocrinology 144, 720–731 (2003).
Pohlenz, J. et al. Improved radioimmunoassay for measurement of mouse thyrotropin in serum: strain differences in thyrotropin concentration and thyrotroph sensitivity to thyroid hormone. Thyroid 9, 1265–1271 (1999).
Weiss, R.E. et al. Mice deficient in the steroid receptor co-activator 1 (SRC-1) are resistant to thyroid hormone. EMBO J. 18, 1900–1904 (1999).
Paxinos, G. & Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates, 2nd edn (Academic Press, San Diego, 2001).
VanGuilder, H.D., Vrana, K.E. & Freeman, W.M. Twenty-five years of quantitative PCR for gene expression analysis. Biotechniques 44, 619–626 (2008).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Acknowledgements
The authors thank X.-H. Liao from the Refetoff laboratory (The University of Chicago, supported by US National Institutes of Health (NIH) grant DK15700) for measuring T4, T3, FT4I and TSH in mice, P. Scheiffele for the antibody to IGSF1 CTD and S. Kimmins (McGill University) and P. Bisschop (Academic Medical Center, University of Amsterdam) for human testis and pituitary RNA, respectively. We also thank H. Bikker, J.C. Moreno, A. Escudero, E. Aten, M. Losekoot, E. Endert, J.W.A. Smit, R.van Rijn and E.L. van Persijn-van Meerten for technical support and advice. We thank F.J. de Jong and Y. de Rijke for measuring serum inhibin B and AMH and for providing age references. We thank S. Tran and X. Zhou for assistance with collection of mouse serum samples. We acknowledge the help of N. Zwaveling, J. Gosen, E.J. Schroor, L.C.G. de Graaff and G. Radetti in providing clinical data. We acknowledge The Eastern Region Sequencing and Informatics Hub (see URLs), who undertook sequencing and preliminary bioinformatics analyses of data from the UK families. We thank the subjects and their families for participating. Our work was supported in part by a grant from the China Scholarship Council (to Y.S.); a National Sciences and Engineering Research Council (NSERC) Doctoral Research Award (to B.B.), NSERC Discovery Grant 341801-07 and a Fonds de la Recherche en Santé du Québec (FRSQ) Chercheur Boursier Senior Award (to D.J.B.); grants from the Wellcome Trust (095564 to N.S. and K.C.; WT077157/Z/05/Z to E.C. and J.K.W.; 084361, 078432 and 086545 to J.P.M.-B.), the National Institutes of Health Research Cambridge Biomedical Research Centre (to N.S. and K.C.) and the UK Medical Research Council (MRC; U117570590 to P.l.T.); a National Health and Medical Research Council of Australia Practitioner Fellowship (to T.M.E.D.); the Dutch Growth Research Foundation (D.G.D.B.); the Young Investigator grant of the Italian Ministry of Health and Istituto Auxologico Italiano IRCCS (GR-2008-1137632 to M.B.); and the Great Ormond Street Children's Hospital Charity (to M.T.D.).
Author information
Authors and Affiliations
Contributions
Y.S., J.T.d.D., M.K., N.S. and K.C. developed the exome sequencing protocol. A.S.P.v.T., W.O., S.G.K., N.R.B., N.M.A.-D., A.M.P., M.H.B., R.C.H., M.T.D., N.S., L.P., I.C., M.B., P.B.-P., H.Z., T.M.E.D., K.C., A.C.S.H.-K., D.G.D.B. and J.M.W. designed the clinical research studies. Y.S., J.F.J.L. and N.S. performed bioinformatics analyses, mutational analysis and genotyping. B.B. generated the vectors expressing mutant IGSF1 and performed all associated biochemical analyses; maintained the mouse colony, collected all mouse tissues and plasma, and analyzed pituitary gene expression; and prepared figures. D.J.B. generated the mouse model, supervised all in vitro and mouse work and participated in data collection and construction of the figures. P.V. and M.G.W. contributed to mouse phenotyping. E.C., J.K.W. and M.G.W. performed mouse T4 measurements. P.l.T. performed measurements of pituitary TSH and prolactin content. S.N.M.G. and J.P.M.-B. carried out the IGSF1 expression studies in mouse and human embryos. C.A.L.R. and C.A.J.B. performed and analyzed the microarray and hybridization experiments. A.S.P.v.T., W.O., W.H.S.-B., T.V., M.J.K., L.P., I.C., M.B., P.B.-P., H.Z., T.M.E.D., A.C.S.H.-K., D.G.D.B., J.J.R., S.G.K., N.R.B., N.M.A.-D., A.M.P., G.C.J.H., E.P.C., M.H.B., R.C.H., A.C.S.H.-K. and M.T.D. contributed to clinical evaluations and the delineation of the subject phenotypes. Y.S., B.B., N.S., A.S.P.v.T., K.C., M.T.D., R.C.H., D.J.B. and J.M.W. prepared the manuscript. D.J.B., J.M.W., K.C. and M.T.D. conceived and supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Tables 1–6 and Supplementary Note (PDF 3269 kb)
Rights and permissions
About this article
Cite this article
Sun, Y., Bak, B., Schoenmakers, N. et al. Loss-of-function mutations in IGSF1 cause an X-linked syndrome of central hypothyroidism and testicular enlargement. Nat Genet 44, 1375–1381 (2012). https://doi.org/10.1038/ng.2453
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2453
This article is cited by
-
Hepatomegaly and fatty liver disease secondary to central hypothyroidism in combination with macrosomia as initial presentation of IGSF1 deficiency syndrome
Hormones (2023)
-
New genetics in congenital hypothyroidism
Endocrine (2021)
-
Acromegaly
Nature Reviews Disease Primers (2019)
-
Congenital hypothyroidism: insights into pathogenesis and treatment
International Journal of Pediatric Endocrinology (2017)
-
Central hypothyroidism — a neglected thyroid disorder
Nature Reviews Endocrinology (2017)