Abstract
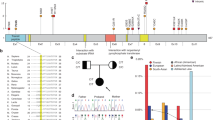
Alternating hemiplegia of childhood (AHC) is a rare, severe neurodevelopmental syndrome characterized by recurrent hemiplegic episodes and distinct neurological manifestations. AHC is usually a sporadic disorder and has unknown etiology. We used exome sequencing of seven patients with AHC and their unaffected parents to identify de novo nonsynonymous mutations in ATP1A3 in all seven individuals. In a subsequent sequence analysis of ATP1A3 in 98 other patients with AHC, we found that ATP1A3 mutations were likely to be responsible for at least 74% of the cases; we also identified one inherited mutation in a case of familial AHC. Notably, most AHC cases are caused by one of seven recurrent ATP1A3 mutations, one of which was observed in 36 patients. Unlike ATP1A3 mutations that cause rapid-onset dystonia-parkinsonism, AHC-causing mutations in this gene caused consistent reductions in ATPase activity without affecting the level of protein expression. This work identifies de novo ATP1A3 mutations as the primary cause of AHC and offers insight into disease pathophysiology by expanding the spectrum of phenotypes associated with mutations in ATP1A3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Verret, S. & Steele, J.C. Alternating hemiplegia in childhood: a report of eight patients with complicated migraine beginning in infancy. Pediatrics 47, 675–680 (1971).
Bourgeois, M., Aicardi, J. & Goutieres, F. Alternating hemiplegia of childhood. J. Pediatr. 122, 673–679 (1993).
Mikati, M.A., Kramer, U., Zupanc, M.L. & Shanahan, R.J. Alternating hemiplegia of childhood: clinical manifestations and long-term outcome. Pediatr. Neurol. 23, 134–141 (2000).
Sweney, M.T. et al. Alternating hemiplegia of childhood: early characteristics and evolution of a neurodevelopmental syndrome. Pediatrics 123, e534–e541 (2009).
Panagiotakaki, E. et al. Evidence of a non-progressive course of alternating hemiplegia of childhood: study of a large cohort of children and adults. Brain 133, 3598–3610 (2010).
Rho, J.M. & Chugani, H.T. Alternating hemiplegia of childhood: insights into its pathophysiology. J. Child Neurol. 13, 39–45 (1998).
Neville, B.G. & Ninan, M. The treatment and management of alternating hemiplegia of childhood. Dev. Med. Child Neurol. 49, 777–780 (2007).
Mikati, M.A. et al. A syndrome of autosomal dominant alternating hemiplegia: clinical presentation mimicking intractable epilepsy; chromosomal studies; and physiologic investigations. Neurology 42, 2251–2257 (1992).
Swoboda, K.J. et al. Alternating hemiplegia of childhood or familial hemiplegic migraine? A novel ATP1A2 mutation. Ann. Neurol. 55, 884–887 (2004).
Bassi, M.T. et al. A novel mutation in the ATP1A2 gene causes alternating hemiplegia of childhood. J. Med. Genet. 41, 621–628 (2004).
Vanmolkot, K.R. et al. Novel mutations in the Na+/K+ ATPase pump gene ATP1A2 associated with familial hemiplegic migraine and benign familial infantile convulsions. Ann. Neurol. 54, 360–366 (2003).
De Fusco, M. et al. Haploinsufficiency of ATP1A2 encoding the Na+/K+ pump α2 subunit associated with familial hemiplegic migraine type 2. Nat. Genet. 33, 192–196 (2003).
Rebhan, M., Chalifa-Caspi, V., Prilusky, J. & Lancet, D. GeneCards: integrating information about genes, proteins and diseases. Trends Genet. 13, 163 (1997).
de Carvalho Aguiar, P. et al. Mutations in the Na+/K+ ATPase α3 gene ATP1A3 are associated with rapid-onset dystonia parkinsonism. Neuron 43, 169–175 (2004).
Anselm, I.A., Sweadner, K.J., Gollamudi, S., Ozelius, L.J. & Darras, B.T. Rapid-onset dystonia-parkinsonism in a child with a novel ATP1A3 gene mutation. Neurology 73, 400–401 (2009).
Svetel, M. et al. Rapid-onset dystonia-parkinsonism: case report. J. Neurol. 257, 472–474 (2010).
Kamm, C. et al. Novel ATP1A3 mutation in a sporadic RDP patient with minimal benefit from deep brain stimulation. Neurology 70, 1501–1503 (2008).
Blanco-Arias, P. et al. A C-terminal mutation of ATP1A3 underscores the crucial role of sodium affinity in the pathophysiology of rapid-onset dystonia-parkinsonism. Hum. Mol. Genet. 18, 2370–2377 (2009).
Ogawa, H., Shinoda, T., Cornelius, F. & Toyoshima, C. Crystal structure of the sodium-potassium pump Na+/K+ ATPase with bound potassium and ouabain. Proc. Natl. Acad. Sci. USA 106, 13742–13747 (2009).
Bellus, G.A. et al. Achondroplasia is defined by recurrent G380R mutations of FGFR3. Am. J. Hum. Genet. 56, 368–373 (1995).
Cooper, D.N. & Youssoufian, H. The CpG dinucleotide and human genetic disease. Hum. Genet. 78, 151–155 (1988).
Clapcote, S.J. et al. Mutation I810N in the α3 isoform of Na+/K+ ATPase causes impairments in the sodium pump and hyperexcitability in the CNS. Proc. Natl. Acad. Sci. USA 106, 14085–14090 (2009).
Stenson, P.D. et al. Human Gene Mutation Database (HGMD): 2003 update. Hum. Mutat. 21, 577–581 (2003).
Jain, E. et al. Infrastructure for the life sciences: design and implementation of the UniProt website. BMC Bioinformatics 10, 136 (2009).
Pruitt, K.D. et al. The consensus coding sequence (CCDS) project: identifying a common protein-coding gene set for the human and mouse genomes. Genome Res. 19, 1316–1323 (2009).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Zhu, M. et al. Inferring copy number variants in high-coverage genomes using ERDS. Am. J. Hum. Genet. (in the press).
Ge, D. et al. SVA: software for annotating and visualizing sequenced human genomes. Bioinformatics 27, 1998–2000 (2011).
Acknowledgements
We are deeply indebted to all the AHC-affected families for their participation in this study. We would like to thank the Alternating Hemiplegia of Childhood Foundation (AHCF) for their efforts in coordinating the collection of US samples and the facilitation of US research collaboration. We also would like to thank D. Poncelin (French Family Foundation) and R. Vavassori (Italian AHC Family Foundation) for facilitating the international collaboration.
We thank the Italian Patient Association for Alternating Hemiplegia (AISEA Onlus) for coordinating and funding the project I.B.AHC Biobank and Clinical Registry for Alternating Hemiplegia. Specifically, we thank M.T. Bassi and E. Tenderini for preparing all of the samples from AHC-affected individuals for analysis. Many thanks also to the Scientific Institute E. Medea, which hosts the I.B.AHC Biobank according to the I.B.AHC protocol.
We also thank the ENRAH for the SMEs Consortium, the ENRAH validation committee and all collaborating physicians for the data collection5. We are also grateful to the DNA and cell bank of Genethon for the processing of French blood samples.
The following individuals contributed next-generation sequenced control samples to this study: D. Attix, E. Behr, R. Brown, J. Burke, D. Daskalakis, V. Dixon, Z. Farfel, R. Gbadegesin, A. Holden, E. Holtzman, J. Hoover-Fong, C. Hulette, S. Kerns, D. Lancet, D. Levy, N. Liang, W. Lowe, P. Lugar, D. Marchuk, J. McEvoy, J. Milner, H. Oster, R. Ottman, S. Palmer, E. Pras, V. Shashi, N. Sobriera, D. Valle, K. Welsh-Bohmer and M. Winn, as well as the MURDOCK study community registry. Funding for the collection and sequencing of control samples was provided by the Center for Human Genome Variation, the Center for HIV/AIDS Vaccine Immunology and the Joseph and Kathleen Bryan Alzheimer's Disease Research Center under grants from the National Institutes of Health (NIH), including the National Institute of Allergy and Infectious Disease (UO1AIO67854); the National Institute on Aging (P30 AG028377); the National Institute of Neurological Disorders and Stroke (RC2NS070344 and 1RC2NS070342-01) and the National Institute of Mental Health (RC2MH089915).
This study was funded in part by grants from the AHCF (to K.J.S., S.P.R. and T.M.N.); the ENRAH for SMEs Consortium under the European Commission Sixth Framework Programme; the Institut National de la Santé et de la Recherche Médicale (to S.N. and B.F.); the Centre National de la Recherche Scientifique (to S.N. and B.F.); the University Pierre and Marie Curie (to S.N. and B.F.); the Association Française Contre les Myopathies (to S.N. and B.F.); the Association Française de l'Hémiplégie Alternante (to S.N., A.M.J.M.v.d.M. and B.d.V.); AISEA Onlus (to F.G. and G.N.); the Center for Human Genome Variation; the Wellcome Trust (084730 to S.M.S.); the National Center for Research Resources (UL1RR025764 to the University of Utah Center for Clinical and Translational Sciences; K.J.S.); the NIH (1T32HL105321-01 to C.H.); the University of Luxembourg Institute for Systems Biology Program (to C.H.) and the Center for Medical Systems Biology established in The Netherlands Genomics Initiative and The Netherlands Organisation for Scientific Research (project 050-060-409 to A.M.J.M.v.d.M. and M.D.F.). S.N. is a recipient of a Contrat d'Interface from Assistance Publique-Hôpitaux de Paris.
Author information
Authors and Affiliations
Consortia
Contributions
E.L.H., Y.H., S.M.S., M.A.M. and D.B.G. conceived and designed the study. Genetic data were generated and analyzed by E.L.H., K.J.S., Y.H., F.G., S.N., B.d.V., F.D.T., S.F., E.A., L.D.P., C.H., L.B.J., K.V.S., C.E.G., L.L., G.N., A.A. A.M.J.M.v.d.M and D.B.G. DNA samples and phenotypic information for AHC patients were collected, compiled and analyzed by K.J.S., F.G., S.N., N.M.W., B.d.V., F.D.T., B.F., S.H., E.P., M.T.S., T.M.N., L.V., S.P.R., K.J.M., K.S., L.J.P., J.H., M.D.F., A.M.B., G.K.H., C.M.W., D.W., B.J.L., P.U., M.D.K., I.E.S., G.N., A.A., S.M.S., M.A.M., the European AHC Genetics Consortium, the I.B.AHC Consortium and the ENRAH for SMEs Consortium. E.L.H., A.M.J.M.v.d.M., S.M.S., M.A.M. and D.B.G. wrote the paper. All authors reviewed the compiled manuscript.
Corresponding authors
Ethics declarations
Competing interests
D.B.G., E.L.H., K.V.S., M.A.M. and Duke University are named on a patent application filed by Duke University on the basis of this work.
Additional information
A full list of members and affiliations appears at the end of this paper.
A full list of members and affiliations appears at the end of this paper.
A full list of members and affiliations appears at the end of this paper.
Supplementary information
Rights and permissions
About this article
Cite this article
Heinzen, E., Swoboda, K., Hitomi, Y. et al. De novo mutations in ATP1A3 cause alternating hemiplegia of childhood. Nat Genet 44, 1030–1034 (2012). https://doi.org/10.1038/ng.2358
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2358
This article is cited by
-
Alternating hemiplegia of childhood misdiagnosed as hysteria: a case report
Acta Epileptologica (2024)
-
Childhood-related neural genotype–phenotype in ATP1A3 mutations: comprehensive analysis
Genes & Genomics (2024)
-
Exome sequencing of ATP1A3-negative cases of alternating hemiplegia of childhood reveals SCN2A as a novel causative gene
European Journal of Human Genetics (2024)
-
The CLDN5 gene at the blood-brain barrier in health and disease
Fluids and Barriers of the CNS (2023)
-
Advances for pediatricians in 2022: allergy, anesthesiology, cardiology, dermatology, endocrinology, gastroenterology, genetics, global health, infectious diseases, metabolism, neonatology, neurology, oncology, pulmonology
Italian Journal of Pediatrics (2023)