Abstract
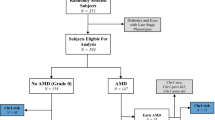
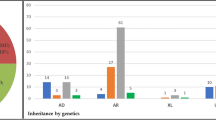
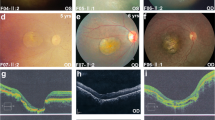
Age-related macular degeneration (AMD) is a prevalent multifactorial disorder of the central retina1,2,3. Genetic variants at two chromosomal loci, 1q31 and 10q26, confer major disease risks, together accounting for more than 50% of AMD pathology4,5,6,7,8,9. Signals at 10q26 center over two nearby genes, ARMS2 (age-related maculopathy susceptibility 2, also known as LOC387715)8,9 and HTRA1 (high-temperature requirement factor A1)10,11, suggesting two equally probable candidates. Here we show that a deletion-insertion polymorphism in ARMS2 (NM_001099667.1:c.*372_815del443ins54) is strongly associated with AMD, directly affecting the transcript by removing the polyadenylation signal and inserting a 54-bp element known to mediate rapid mRNA turnover. As a consequence, expression of ARMS2 in homozygous carriers of the indel variant is not detectable. Confirming previous findings12, we demonstrate a mitochondrial association of the normal protein and further define its retinal localization to the ellipsoid region of the photoreceptors. Our data suggest that ARMS2 has a key role in AMD, possibly through mitochondria-related pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Mitchell, P., Smith, W., Attebo, K. & Wang, J.J. Prevalence of age-related maculopathy in Australia. The Blue Mountains Eye Study. Ophthalmology 102, 1450–1460 (1995).
Vingerling, J.R. et al. The prevalence of age-related maculopathy in the Rotterdam Study. Ophthalmology 102, 205–210 (1995).
Seddon, J.M., Ajani, U.A. & Mitchell, B.D. Familial aggregation of age-related maculopathy. Am. J. Ophthalmol. 123, 199–206 (1997).
Klein, R.J. et al. Complement factor H polymorphism in age-related macular degeneration. Science 308, 385–389 (2005).
Edwards, A.O. et al. Complement factor H polymorphism and age-related macular degeneration. Science 308, 421–424 (2005).
Haines, J.L. et al. Complement factor H variant increases the risk of age-related macular degeneration. Science 308, 419–421 (2005).
Hageman, G.S. et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc. Natl. Acad. Sci. USA 102, 7227–7232 (2005).
Jakobsdottir, J. et al. Susceptibility genes for age-related maculopathy on chromosome 10q26. Am. J. Hum. Genet. 77, 389–407 (2005).
Rivera, A. et al. Hypothetical LOC387715 is a second major susceptibility gene for age-related macular degeneration, contributing independently of complement factor H to disease risk. Hum. Mol. Genet. 14, 3227–3236 (2005).
Dewan, A. et al. HTRA1 promoter polymorphism in wet age-related macular degeneration. Science 314, 989–992 (2006).
Yang, Z. et al. A variant of the HTRA1 gene increases susceptibility to age-related macular degeneration. Science 314, 992–993 (2006).
Kanda, A. et al. A variant of mitochondrial protein LOC387715/ARMS2, not HTRA1, is strongly associated with age-related macular degeneration. Proc. Natl. Acad. Sci. USA 104, 16227–16232 (2007).
Grau, S. et al. The role of human HtrA1 in arthritic disease. J. Biol. Chem. 281, 6124–6129 (2006).
Khabar, K.S. The AU-rich transcriptome: more than interferons and cytokines, and its role in disease. J. Interferon Cytokine Res. 25, 1–10 (2005).
Barreau, C., Paillard, L. & Osborne, H.B. AU-rich elements and associated factors: are there unifying principles? Nucleic Acids Res. 33, 7138–7150 (2005).
Garneau, N.L., Wilusz, J. & Wilusz, C.J. The highways and byways of mRNA decay. Nat. Rev. Mol. Cell Biol. 8, 113–126 (2007).
Molday, L.L., Hicks, D., Sauer, C.G., Weber, B.H. & Molday, R.S. Expression of X-linked retinoschisis protein RS1 in photoreceptor and bipolar cells. Invest. Ophthalmol. Vis. Sci. 42, 816–825 (2001).
Zeviani, M. & Carelli, V. Mitochondrial disorders. Curr. Opin. Neurol. 20, 564–571 (2007).
Zarbin, M.A. Current concepts in the pathogenesis of age-related macular degeneration. Arch. Ophthalmol. 122, 598–614 (2004).
Lin, M.T. & Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443, 787–795 (2006).
Gabriel, S.B. et al. The structure of haplotype blocks in the human genome. Science 296, 2225–2229 (2002).
Barrett, J.C., Fry, B., Maller, J. & Daly, M.J. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265 (2005).
Dudbridge, F. Pedigree disequilibrium tests for multilocus haplotypes. Genet. Epidemiol. 25, 115–121 (2003).
Dudbridge, F. UNPHASED user guide. Technical Report 5. (MRC Biostatistics Unit, Cambridge, 2006).
Fisher, S.A. et al. Case-control genetic association study of fibulin-6 (FBLN6 or HMCN1) variants in age-related macular degeneration (AMD). Hum. Mutat. 28, 406–413 (2007).
Geysen, H.M., Rodda, S.J., Mason, T.J., Tribbick, G. & Schoofs, P.G. Strategies for epitope analysis using peptide synthesis. J. Immunol. Methods 102, 259–274 (1987).
Fernandez-Vizarra, E., Lopez-Perez, M.J. & Enriquez, J.A. Isolation of biogenetically competent mitochondria from mammalian tissues and cultured cells. Methods 26, 292–297 (2002).
Wu, W.W. & Molday, R.S. Defective discoidin domain structure, subunit assembly, and endoplasmic reticulum processing of retinoschisin are primary mechanisms responsible for X-linked retinoschisis. J. Biol. Chem. 278, 28139–28146 (2003).
Acknowledgements
We are grateful to the individuals with AMD and the control subjects for their participation in this study; G. Huber for help with placental samples; P. Lichtner and T. Meitinger for SNP genotyping, R.S. Molday (Department of Biochemistry and Molecular Biology, University of British Columbia, Vancouver, Canada) for kindly sharing monoclonal antibodies RS1-3R10 and Rho-1D4, F.R. Rauscher (Institute of Human Genetics, University of Regensburg, Germany) for providing ARMS2 cDNA clones of isoforms 1 and 2, and D. Wagner for technical assistance. This work was supported in part by grants to B.H.F.W. from the German Research Foundation (DFG), the Ruth and Milton Steinbach Foundation New York (B.H.F.W.) and the Alcon Research Institute.
Author information
Authors and Affiliations
Contributions
L.G.F., T.L., A.J. and A.R. carried out all experimental studies. Specifically, L.G.F. participated in conception and design of the association study, was involved in all aspects of genotyping and data analysis and drafted the manuscript. T.L. made major contributions to the generation of ARMS2 antibodies and was responsible for all aspects of the protein work. A.J. carried out the immunohistochemistry studies. A.R. was involved in data acquisition and the characterization of ARMS2 isoforms and carried out the RNA work. S.A.F. performed the statistical analyses and critically revised the manuscript. C.N.K. recruited the individuals with AMD and the controls and collected the peripheral blood samples. B.H.F.W. conceived of the study, participated in its design and coordination and finalized the manuscript. All authors read and approved the final text.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2 and Supplementary Tables 1–5 (PDF 3198 kb)
Rights and permissions
About this article
Cite this article
Fritsche, L., Loenhardt, T., Janssen, A. et al. Age-related macular degeneration is associated with an unstable ARMS2 (LOC387715) mRNA. Nat Genet 40, 892–896 (2008). https://doi.org/10.1038/ng.170
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.170
This article is cited by
-
The role of mitochondrial genes on nuclear gene expression in neovascular age related macular degeneration: analysis of nuclear VEGF gene expression after ranibizumab treatment in cytoplasmic hybrid retinal pigment epithelial cell lines correlated with clinical evolution
International Journal of Retina and Vitreous (2023)
-
Retinal drusen counts are increased in inflammatory bowel disease, and with longer disease duration, more complications and associated IgA glomerulonephritis
Scientific Reports (2022)
-
Association of the HtrA1 rs11200638 Polymorphism with Neovascular Age-Related Macular Degeneration in Indonesia
Ophthalmology and Therapy (2022)
-
Macular thickness varies with age-related macular degeneration genetic risk variants in the UK Biobank cohort
Scientific Reports (2021)
-
HTRA1 rs11200638 variant and AMD risk from a comprehensive analysis about 15,316 subjects
BMC Medical Genetics (2020)