Abstract
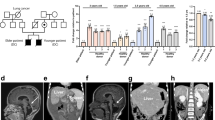
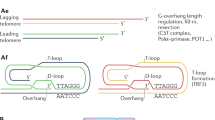
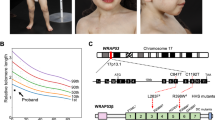
Coats plus is a highly pleiotropic disorder particularly affecting the eye, brain, bone and gastrointestinal tract. Here, we show that Coats plus results from mutations in CTC1, encoding conserved telomere maintenance component 1, a member of the mammalian homolog of the yeast heterotrimeric CST telomeric capping complex. Consistent with the observation of shortened telomeres in an Arabidopsis CTC1 mutant and the phenotypic overlap of Coats plus with the telomeric maintenance disorders comprising dyskeratosis congenita, we observed shortened telomeres in three individuals with Coats plus and an increase in spontaneous γH2AX-positive cells in cell lines derived from two affected individuals. CTC1 is also a subunit of the α-accessory factor (AAF) complex, stimulating the activity of DNA polymerase-α primase, the only enzyme known to initiate DNA replication in eukaryotic cells. Thus, CTC1 may have a function in DNA metabolism that is necessary for but not specific to telomeric integrity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Jain, D. & Cooper, J.P. Telomeric strategies: means to an end. Annu. Rev. Genet. 44, 243–269 (2010).
Savage, S.A. & Bertuch, A.A. The genetics and clinical manifestations of telomere biology disorders. Genet. Med. 12, 753–764 (2010).
Nelson, N.D. & Bertuch, A.A. Dyskeratosis congenita as a disorder of telomere maintenance. Mutat. Res. published online, doi:10.1016/j.mrfmmm.2011.06.008 (2 July 2011).
Tolmie, J.L., Browne, B.H., McGettrick, P.M. & Stephenson, J.B. A familial syndrome with coats' reaction retinal angiomas, hair and nail defects and intracranial calcification. Eye (Lond.) 2, 297–303 (1988).
Crow, Y.J. et al. Coats' plus: a progressive familial syndrome of bilateral Coats' disease, characteristic cerebral calcification, leukoencephalopathy, slow pre- and post-natal linear growth and defects of bone marrow and integument. Neuropediatrics 35, 10–19 (2004).
Nagae-Poetscher, L.M. et al. Leukoencephalopathy, cerebral calcifications, and cysts: new observations. Neurology 62, 1206–1209 (2004).
Linnankivi, T. et al. Cerebroretinal microangiopathy with calcifications and cysts. Neurology 67, 1437–1443 (2006).
Briggs, T.A. et al. Cerebroretinal microangiopathy with calcifications and cysts (CRMCC). Am. J. Med. Genet. A. 146A, 182–190 (2008).
Briggs, T.A. et al. Treatment of gastrointestinal bleeding in a probable case of cerebroretinal microangiopathy with calcifications and cysts. Mol. Syndromol. 1, 159–162 (2011).
Labrune, P. et al. Extensive brain calcifications, leukodystrophy, and formation of parenchymal cysts: a new progressive disorder due to diffuse cerebral microangiopathy. Neurology 46, 1297–1301 (1996).
Linger, B.R. & Price, C.M. Conservation of telomere protein complexes: shuffling through evolution. Crit. Rev. Biochem. Mol. Biol. 44, 434–446 (2009).
Price, C.M. et al. Evolution of CST function in telomere maintenance. Cell Cycle 9, 3157–3165 (2010).
Surovtseva, Y.V. et al. Conserved telomere maintenance component 1 interacts with STN1 and maintains chromosome ends in higher eukaryotes. Mol. Cell 36, 207–218 (2009).
Miyake, Y. et al. RPA-like mammalian Ctc1-Stn1-Ten1 complex binds to single-stranded DNA and protects telomeres independently of the Pot1 pathway. Mol. Cell 36, 193–206 (2009).
Baerlocher, G.M., Vulto, I., de Jong, G. & Lansdorp, P.M. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nat. Protoc. 1, 2365–2376 (2006).
Casteel, D.E. et al. A DNA polymerase-α·primase cofactor with homology to replication protein A-32 regulates DNA replication in mammalian cells. J. Biol. Chem. 284, 5807–5818 (2009).
Goulian, M. & Heard, C.J. The mechanism of action of an accessory protein for DNA polymerase α/primase. J. Biol. Chem. 265, 13231–13239 (1990).
Giraud-Panis, M.J., Teixeira, M.T., Geli, V. & Gilson, E. CST meets shelterin to keep telomeres in check. Mol. Cell 39, 665–676 (2010).
Acknowledgements
We are very grateful to the affected families for their involvement in our research. Y.J.C. acknowledges the Manchester National Institute for Health Research (NIHR) Biomedical Research Centre. This work has received support from the Great Ormond Street Hospital Children's Charity and from the Newlife Foundation. We thank the NHLBI Grand Opportunity (GO) Exome Sequencing Project and acknowledge its ongoing studies that produced and provided exome variant calls for comparison, including the Lung GO Sequencing Project (HL-102923), the Women's Health Initiative (WHI) Sequencing Project (HL-102924), the Broad GO Sequencing Project (HL-102925), the Seattle GO Sequencing Project (HL-102926) and the Heart GO Sequencing Project (HL-103010). We also thank G. Forte for organizing courier delivery of patient materials.
Author information
Authors and Affiliations
Contributions
B.H.A. and J.O. performed exome sequencing. B.H.A., M.S., G.I.R. and E.M.J. were responsible for Sanger sequencing. J. Mayer, S.S.B. and J.E.D. undertook analysis of the exome sequence data. J.E.U. and S.B.D. performed SNP analyses. J.H.L. was responsible for neuroradiological phenotyping. G.M.B. and E.O.L. performed the telomeric length analysis. S.M. and J. Muter assessed γH2AX positivity. S.C.L. analyzed CTC1 structural domains. G.I.R. and G.C.M.B. provided critical input into project development and manuscript preparation. Y.J.C. designed and supervised the project and wrote the manuscript with the support of P.R.K. All other authors identified subjects with Coats plus and/or Labrune syndrome and performed related clinical and laboratory studies. Phenotypic overlap with dyskeratosis congenita was originally appreciated by J.L.T. and J.B.P.S. at the time of the first clinical description of Coats plus.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Tables 1–5 (PDF 1730 kb)
Rights and permissions
About this article
Cite this article
Anderson, B., Kasher, P., Mayer, J. et al. Mutations in CTC1, encoding conserved telomere maintenance component 1, cause Coats plus. Nat Genet 44, 338–342 (2012). https://doi.org/10.1038/ng.1084
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.1084
This article is cited by
-
CST–polymerase α-primase solves a second telomere end-replication problem
Nature (2024)
-
Genetics of human telomere biology disorders
Nature Reviews Genetics (2023)
-
Genetic Counseling and Family Screening Recommendations in Patients with Telomere Biology Disorders
Current Hematologic Malignancy Reports (2023)
-
Cryo-EM structure of the human CST–Polα/primase complex in a recruitment state
Nature Structural & Molecular Biology (2022)
-
Shaping human telomeres: from shelterin and CST complexes to telomeric chromatin organization
Nature Reviews Molecular Cell Biology (2021)