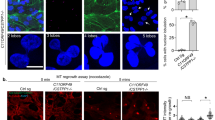
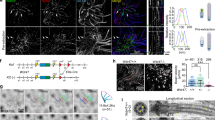
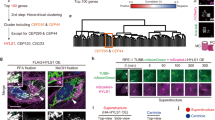
Abstract
Tubulin glutamylation is a post-translational modification that occurs predominantly in the ciliary axoneme and has been suggested to be important for ciliary function1,2. However, its relationship to disorders of the primary cilium, termed ciliopathies, has not been explored. Here we mapped a new locus for Joubert syndrome (JBTS)3, which we have designated as JBTS15, and identified causative mutations in CEP41, which encodes a 41-kDa centrosomal protein4. We show that CEP41 is localized to the basal body and primary cilia, and regulates ciliary entry of TTLL6, an evolutionarily conserved polyglutamylase enzyme5. Depletion of CEP41 causes ciliopathy-related phenotypes in zebrafish and mice and results in glutamylation defects in the ciliary axoneme. Our data identify CEP41 mutations as a cause of JBTS and implicate tubulin post-translational modification in the pathogenesis of human ciliary dysfunction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ikegami, K., Sato, S., Nakamura, K., Ostrowski, L.E. & Setou, M. Tubulin polyglutamylation is essential for airway ciliary function through the regulation of beating asymmetry. Proc. Natl. Acad. Sci. USA 107, 10490–10495 (2010).
Kubo, T., Yanagisawa, H.A., Yagi, T., Hirono, M. & Kamiya, R. Tubulin polyglutamylation regulates axonemal motility by modulating activities of inner-arm dyneins. Curr. Biol. 20, 441–445 (2010).
Parisi, M.A. Clinical and molecular features of Joubert syndrome and related disorders. Am. J. Med. Genet. C. Semin. Med. Genet. 151C, 326–340 (2009).
Andersen, J.S. et al. Proteomic characterization of the human centrosome by protein correlation profiling. Nature 426, 570–574 (2003).
van Dijk, J. et al. A targeted multienzyme mechanism for selective microtubule polyglutamylation. Mol. Cell 26, 437–448 (2007).
Valente, E.M., Brancati, F. & Dallapiccola, B. Genotypes and phenotypes of Joubert syndrome and related disorders. Eur. J. Med. Genet. 51, 1–23 (2008).
Hildebrandt, F. & Zhou, W. Nephronophthisis-associated ciliopathies. J. Am. Soc. Nephrol. 18, 1855–1871 (2007).
Lander, E.S. & Botstein, D. Homozygosity mapping: a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Yamada, T. et al. The gene TSGA14, adjacent to the imprinted gene MEST, escapes genomic imprinting. Gene 288, 57–63 (2002).
Bordo, D. & Bork, P. The rhodanese/Cdc25 phosphatase superfamily. Sequence-structure-function relations. EMBO Rep. 3, 741–746 (2002).
Beales, P.L. et al. IFT80, which encodes a conserved intraflagellar transport protein, is mutated in Jeune asphyxiating thoracic dystrophy. Nat. Genet. 39, 727–729 (2007).
Gorden, N.T. et al. CC2D2A is mutated in Joubert syndrome and interacts with the ciliopathy-associated basal body protein CEP290. Am. J. Hum. Genet. 83, 559–571 (2008).
Colantonio, J.R. et al. The dynein regulatory complex is required for ciliary motility and otolith biogenesis in the inner ear. Nature 457, 205–209 (2009).
Khanna, H. et al. A common allele in RPGRIP1L is a modifier of retinal degeneration in ciliopathies. Nat. Genet. 41, 739–745 (2009).
Essner, J.J., Amack, J.D., Nyholm, M.K., Harris, E.B. & Yost, H.J. Kupffer's vesicle is a ciliated organ of asymmetry in the zebrafish embryo that initiates left-right development of the brain, heart and gut. Development 132, 1247–1260 (2005).
Nonaka, S. et al. Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein. Cell 95, 829–837 (1998).
McGrath, J. & Brueckner, M. Cilia are at the heart of vertebrate left-right asymmetry. Curr. Opin. Genet. Dev. 13, 385–392 (2003).
Amack, J.D. & Yost, H.J. The T box transcription factor no tail in ciliated cells controls zebrafish left-right asymmetry. Curr. Biol. 14, 685–690 (2004).
Ross, A.J. et al. Disruption of Bardet-Biedl syndrome ciliary proteins perturbs planar cell polarity in vertebrates. Nat. Genet. 37, 1135–1140 (2005).
Hoover, A.N. et al. C2cd3 is required for cilia formation and Hedgehog signaling in mouse. Development 135, 4049–4058 (2008).
Caspary, T., Larkins, C.E. & Anderson, K.V. The graded response to Sonic Hedgehog depends on cilia architecture. Dev. Cell 12, 767–778 (2007).
Jurczyk, A. et al. Pericentrin forms a complex with intraflagellar transport proteins and polycystin-2 and is required for primary cilia assembly. J. Cell Biol. 166, 637–643 (2004).
Million, K. et al. Polyglutamylation and polyglycylation of α- and β-tubulins during in vitro ciliated cell differentiation of human respiratory epithelial cells. J. Cell Sci. 112, 4357–4366 (1999).
Suryavanshi, S. et al. Tubulin glutamylation regulates ciliary motility by altering inner dynein arm activity. Curr. Biol. 20, 435–440 (2010).
Vogel, P., Hansen, G., Fontenot, G. & Read, R. Tubulin tyrosine ligase-like 1 deficiency results in chronic rhinosinusitis and abnormal development of spermatid flagella in mice. Vet. Pathol. 47, 703–712 (2010).
Wloga, D. et al. Hyperglutamylation of tubulin can either stabilize or destabilize microtubules in the same cell. Eukaryot. Cell 9, 184–193 (2010).
Becker-Heck, A. et al. The coiled-coil domain containing protein CCDC40 is essential for motile cilia function and left-right axis formation. Nat. Genet. 43, 79–84 (2011).
Zhao, C. & Malicki, J. Genetic defects of pronephric cilia in zebrafish. Mech. Dev. 124, 605–616 (2007).
Pathak, N., Obara, T., Mangos, S., Liu, Y. & Drummond, I.A. The zebrafish fleer gene encodes an essential regulator of cilia tubulin polyglutamylation. Mol. Biol. Cell 18, 4353–4364 (2007).
Pathak, N., Austin, C.A. & Drummond, I.A. Tubulin tyrosine ligase–like genes ttll3 and ttll6 maintain zebrafish cilia structure and motility. J. Biol. Chem. 286, 11685–11695 (2011).
Gaertig, J. & Wloga, D. Ciliary tubulin and its post-translational modifications. Curr. Top. Dev. Biol. 85, 83–113 (2008).
Shida, T., Cueva, J.G., Xu, Z., Goodman, M.B. & Nachury, M.V. The major α-tubulin K40 acetyltransferase αTAT1 promotes rapid ciliogenesis and efficient mechanosensation. Proc. Natl. Acad. Sci. USA 107, 21517–21522 (2010).
Kim, S.K. et al. Planar cell polarity acts through septins to control collective cell movement and ciliogenesis. Science 329, 1337–1340 (2010).
Williams, C.L. et al. MKS and NPHP modules cooperate to establish basal body/transition zone membrane associations and ciliary gate function during ciliogenesis. J. Cell Biol. 192, 1023–1041 (2011).
Rogowski, K. et al. Evolutionary divergence of enzymatic mechanisms for posttranslational polyglycylation. Cell 137, 1076–1087 (2009).
Wloga, D. et al. TTLL3 is a tubulin glycine ligase that regulates the assembly of cilia. Dev. Cell 16, 867–876 (2009).
Bulinski, J.C. Tubulin posttranslational modifications: a Pushmi-Pullyu at work? Dev. Cell 16, 773–774 (2009).
Valente, E.M. et al. Distinguishing the four genetic causes of Jouberts syndrome-related disorders. Ann. Neurol. 57, 513–519 (2005).
Murray, S.S. et al. A highly informative SNP linkage panel for human genetic studies. Nat. Methods 1, 113–117 (2004).
Hoffmann, K. & Lindner, T.H. easyLINKAGE-Plus—automated linkage analyses using large-scale SNP data. Bioinformatics 21, 3565–3567 (2005).
Inglis, P.N., Boroevich, K.A. & Leroux, M.R. Piecing together a ciliome. Trends Genet. 22, 491–500 (2006).
Gherman, A., Davis, E.E. & Katsanis, N. The ciliary proteome database: an integrated community resource for the genetic and functional dissection of cilia. Nat. Genet. 38, 961–962 (2006).
Schnütgen, F. et al. Genomewide production of multipurpose alleles for the functional analysis of the mouse genome. Proc. Natl. Acad. Sci. USA 102, 7221–7226 (2005).
Valente, E.M. et al. Mutations in TMEM216 perturb ciliogenesis and cause Joubert, Meckel and related syndromes. Nat. Genet. 42, 619–625 (2010).
Majumdar, A. & Drummond, I.A. Podocyte differentiation in the absence of endothelial cells as revealed in the zebrafish avascular mutant, cloche. Dev. Genet. 24, 220–229 (1999).
Acknowledgements
We thank the Marshfield Clinic Research Foundation, Center for Inherited Disease Research (supported by the US National Institutes of Health and National Heart, Lung, and Blood Institute) for genotyping support. We thank the International JSRD Study Group, E. Bertini and the French Society of Foetal Pathology for subject referrals, J. Meerloo at the UCSD Microscopy Core (P30NS047101), T. Meerloo, Y. Jones, M. Farquhar and the Department of Cellular and Molecular Medicine (CMM) Electron Microscopy Core Facility at UCSD, S. Wirth and B. Willis for mutant mouse generation, C. Janke (Institute Curie Research Center) for GT335 antibody, TTLLs plasmids and technical advice, I. Drummond and N. Pathak (Massachusetts General Hospital) for ttll6 MO, M. Gorovsky (University of Rochester) for polyE and polyG antibodies, A.T. Look (Dana-Farber Cancer Institute) for the pCS2+ plasmid, S. Audollent for technical help, and B. Sotak, N. Akizu, A. Crawford, V. Cantagrel and E.-J. Choi for stimulating scientific discussion and comments. This work was supported by the US National Institutes of Health (R01NS048453 and R01NS052455 to J.G.G.; R01DK068306 to F.H.; and R01NS064077 to D.A.D.), the American Heart Association (09POST2250641 to J.E.L.), the Italian Ministry of Health (Ricerca Finalizzata Malattie Rare and Ricerca Corrente 2011), the Telethon Foundation Italy (GGP08145) and the Pierfranco and Luisa Mariani Foundation (to E.M.V.), Research was also supported by grants-in-aid from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Japan Society for the Promotion of Science (JSPS), (23117517 and 23570209 to K.I.), the Newlife Charity, the Medical Research Council (G0700073), the Sir Jules Thorn Charitable Trust (09/JTA to C.A.J.), l'Agence National pour la Recherche (ANR) (07-MRAR-Fetalciliopathies to T.A.-B.), Simons Foundation Autism Research Initiative (to J.G.G.) and the Howard Hughes Medical Institute (to F.H. and J.G.G.).
Author information
Authors and Affiliations
Contributions
J.E.L., M.S.Z. and J.G.G. designed the study and experiments with substantial contributions from B.M. S.F.N. helped with fine mapping. J.L.S., S.L.B., J.O., F.B., M.I., A.M.S., T.A.-B., C.V.L., I.A.G., A.C., F.H., C.A.J., D.A.D. and E.M.V. performed genetic screening. J.E.L., J.L.S., J.S., J.O., F.B., M.I., T.A.-B., I.A.G., D.A.D., C.M.L. and J.H.L. performed mutation analysis. M.S.Z., S.E.M., H.R.R., I.R., I.P.C., E.B., C.B. and E.M.V. identified and recruited subjects. K.I. and M.S. shared critical reagents. J.S. helped with genotyping of mutant mice. J.E.L. performed microscopy, biochemical assays and zebrafish and mouse experiments. J.E.L. and J.G.G. interpreted the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Tables 1–4 (PDF 7448 kb)
Supplementary Video 1
A small piece of debris in Kupffer's vesicle of a 12 hpf (6 somite) WT embryo is caught moving ciliary currents. The debris (marked by arrow) follows a counter-clockwise circular path around Kupffer's vesicle. nc, notochord; A, anterior; P, posterior; L, left; R, right directions. (MOV 12423 kb)
Supplementary Video 2
Small pieces of debris in Kupffer's vesicle of a 12 hpf (6 somite) cep41 morphant are caught moving ciliary currents. The debris (within a circle) shows no directional movement, but rather bounces around or stalls. nc, notochord; A, anterior; P, posterior; L, left; R, right directions. (MOV 3588 kb)
Supplementary Video 3
Movement of cilia at the junction area of the pronephric duct and tubule in a 2.5 dpf WT zebrafish embryo. The observed cilium (an arrow) shows rhythmic undulations. Dashed lines demarcate the outline of the pronephric duct lumen and an arrow point out the observed cilium. A, anterior; D, dorsal; P, posterior; V, ventral directions. (MOV 1828 kb)
Supplementary Video 4
Movement of cilia at the junction area of the pronephric duct and tubule in a 2.5 dpf cep41 MO-injected embryo. Motile cilium, observed in the WT embryo, is not found. Dashed lines demarcate the outline of the pronephric duct lumen. A, anterior; D, dorsal; P, posterior; V, ventral directions. (MOV 1699 kb)
Supplementary Video 5
Movement of cilia at the pronephric duct in a 2.5 dpf WT zebrafish embryo. The cilium (an arrow) shows rhythmic undulations. Dashed lines demarcate the outline of the pronephric duct lumen and an arrow point out the observed cilium. A, anterior; D, dorsal; P, posterior; V, ventral directions. (MOV 10860 kb)
Rights and permissions
About this article
Cite this article
Lee, J., Silhavy, J., Zaki, M. et al. CEP41 is mutated in Joubert syndrome and is required for tubulin glutamylation at the cilium. Nat Genet 44, 193–199 (2012). https://doi.org/10.1038/ng.1078
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.1078
This article is cited by
-
Genetic variant of TTLL11 gene and subsequent ciliary defects are associated with idiopathic scoliosis in a 5-generation UK family
Scientific Reports (2021)
-
Distinct regulation of hippocampal neuroplasticity and ciliary genes by corticosteroid receptors
Nature Communications (2021)
-
Co-occurrence of mutations in KIF7 and KIAA0556 in Joubert syndrome with ocular coloboma, pituitary malformation and growth hormone deficiency: a case report and literature review
BMC Pediatrics (2020)
-
Structural basis for polyglutamate chain initiation and elongation by TTLL family enzymes
Nature Structural & Molecular Biology (2020)
-
The tubulin code and its role in controlling microtubule properties and functions
Nature Reviews Molecular Cell Biology (2020)