Abstract
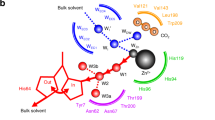
The three dimensional structure of human carbonic anhydrase C has been determined at 2.0 Å resolution. The active site has been identified by the binding of inhibitors and the location of the zinc ion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Meldrum, N. U., and Roughton, F. J. W., J. Physiol., 80, 113 (1933).
Neish, A. C., Biochem. J., 33, 300 (1939).
Maren, T. H., Physiol. Rev., 47, 595 (1967).
Nyman, P.-O., Biochim. Biophys. Acta, 52, 1 (1961).
Furth, A. J., J. Biol. Chem., 243, 4832 (1968).
Khalifah, R. G., J. Biol. Chem., 246, 2561 (1971).
Henderson, L., Henriksson, D., Nyman, P.-O., and Strid, L., Oxygen Affinity of Haemoglobin and Red Cell. Acid-Base Status, Alfred Benzon Symposium IV (Munksgaard, Copenhagen, in the press).
Schneider, F., and Liefländer, M., Z. Physiol. Chem., 334, 279 (1963).
Tashian, R. E., Douglas, D. P., and Yu, Y. L., Biochem. Biophys. Res. Commun., 14, 256 (1964).
Lo, K. W., and Kaiser, E. T., Chem. Commun., 834 (1966).
Verpoorte, J. A., Metha, S., and Edsall, J. T., J. Biol. Chem., 242, 4221 (1967).
Pocker, Y., and Meany, J. E., Biochemistry, 4, 2535 (1965).
Pocker, Y., and Meany, J. E., Biochemistry, 6, 239 (1967).
Derrien, Y., and Laurent, G., Exposés Annuels de Biochimie Médicale, 29, 167 (1969).
Lindskog, S., Henderson, L., Kannan, K. K., Liljas, A., Nyman, P.-O., and Strandberg, B., The Enzymes, third ed. (edit. by Boyer, P.) (Academic Press, New York, in the press).
Lindskog, S., and Malmström, B. G., Biochem. Biophys. Res.Commun., 2, 213 (1960).
Mann, T., and Keilin, D., Nature, 146, 164 (1940).
Kannan, K. K., Liljas, A., Waara, I., Bergstén, P.-C, Lövgren, S., Strandberg, B., Bengtsson, U., Carlbom, U., Fridborg, K., Järup, L., and Petef, M., Cold Spring Harbor Symp. Quant. Biol., 36 (in the press).
Bergstén, P.-C., Waara, I., Lövgren, S., Liljas, A., Kannan, K. K., and Bengtsson, U., Oxygen Affinity of Haemoglobin and Red Cell Acid-Base Status, Alfred Benzon Symposium, IV (Munksgaard, Copenhagen, in the press).
Armstrong, J. McD., Myers, D. V., Verpoorte, J. A., and Edsall, J. T., J. Biol. Chem., 241, 5137 (1966).
Liljas, A., Kannan, K. K., Bergstén, P.-C., Fridborg, K., Järup, L., Lövgren, S., Paradies, H., Strandberg, B., and Waara, L., in CO2: Chemical, Biochemical and Physiological Aspects (edit. by Forster, R. E., Edsall, J. T., Otis, A. B., and Roughton, F. J. W.), 89 (National Aeronautics and Space Administration, Washington DC, 1969).
Tilander, B., Strandberg, B., and Fridborg, K., J. Mol. Biol., 12, 740 (1965).
Fridborg, K., Kannan, K. K., Liljas, A., Lundin, J., Strandberg, B., Strandberg, R., Tilander, B., and Wirén, G., J. Mol. Biol., 25, 505 (1967).
Strandberg, B., Tilander, B., Fridborg, K., Lindskog, S., and Nyman, P. O., J. Mol. Biol., 5, 583 (1962).
Järup, L., Kannan, K. K., Liljas, A., and Strandberg, B., Computer Programs in Biomedicine, 1, 74 (1970).
Dickerson, R. E., Kendrew, J. S., and Strandberg, B. E., Acta Cryst., 14, 1188 (1961).
Muirhead, H., Cox, J. M., Mazzarella, L., and Perutz, M. F., J.Mol. Biol., 28, 117 (1967).
Blow, D. M., and Crick, F. H. C., Acta Cryst., 12, 794 (1959).
Richards, F. M., J. Mol. Biol., 37, 225 (1968).
Ramakrishnan, C., and Nageshwar Prasad, Intern. J. Protein Res., 3, 209 (1971).
Nemethy, G., Phillips, D. C., Leach, S. J., and Sheraga, H. A., Nature, 214, 363 (1967).
Pauling, L., Corey, R. B., and Branson, H. R., Proc. US Nat. Acad. Sci., 37, 205 (1951).
Donohue, J., Proc. US Nat. Acad. Sci., 39, 470 (1953).
Venkatachalam, C. M., Biopolymers, 6, 1425 (1968).
Blake, C. C. F., Mair, G. A., North, A. C. T., Phillips, D. C., and Sarma, V. R., Proc. Roy. Soc., B, 167, 365 (1967).
Birktoft, J. J., Blow, D. M., Henderson, R., and Steitz, T. A., Phil. Trans. Roy. Soc., B, 257, 67 (1970).
Adams, M., Ford, G. C., Koekoek, R., Lentz, P. J., McPherson, A., Rossmann, M. G., Smiley, I. E., Schevitz, R. W., and Wonacott, A. J., Nature, 227, 1098 (1970).
Dickerson, R. E., Takano, T., Eisenberg, D., Kallai, O. B., Samson, L., Cooper, A., and Margoliash, E., J. Biol. Chem., 246, 1511 (1971).
Huggins, M. L., Chem. Revs., 32, 195 (1943).
Pauling, L., and Corey, R. B., Proc. US Nat. Acad. Sci., 37, 729 (1951).
Wyckoff, H. W., Tsernoglou, D., Hanson, A. W., Knox, J. R., Lee, B., and Richards, F. M., J. Biol. Chem., 245, 305 (1970).
Drenth, J., Jansonius, J. N., Koekoek, R., Sluyterman, L. A. A., and Wolthers, B. G., Phil. Trans. Roy. Soc., B, 257, 23 (1970).
Wright, C. S., Alden, R. A., and Kraut, J., Nature, 221, 235 (1969).
Reeke, G. N., Hartsuch, J. A., Ludwig, M. L., Quiocho, F. A., Steitz, T. A., and Lipscomb, W. N., Proc. US Nat. Acad. Sci., 58, 2220 (1967).
Timasheff, S. N., Susi, H., and Stevens, L., J. Biol. Chem., 242, 5467 (1967).
Tashian, R. E., Tanis, R. J., and Ferell, R. E., Oxygen Affinity of Haemoglobin and Red Cell Acid-Base Status, Alfred Benzon Symposium IV (Munksgaard, Copenhagen, in the press).
Beychok, S., Armstrong, J. McD., Lindblow, C., and Edsall, J. T., J. Biol. Chem., 241, 5150 (1966).
Coleman, J. E., J. Biol. Chem., 243, 4574 (1968).
Greenfield, N., and Fasman, G. D., Biochemistry, 8, 4108 (1969).
Coleman, J. E., in Inorganic Biochemistry (edit. by Eichhorn, G. L.) (Elsevier, Amsterdam, in the press).
Rickli, E. E., Ghazanfar, S. A. S., Gibbons, B. H., and Edsall, J. T., J. Biol. Chem., 239, 1065 (1964).
Rosenberg, A., J. Biol. Chem., 241, 5126 (1966).
Riddiford, L. M., J. Biol. Chem., 240, 168 (1965).
Myers, D. V., and Edsall, J. T., Proc. US Nat. Acad. Sci., 53, 169 (1965).
Williams, R. J. P., Biopolymers Symposia, 1, 515 (1964).
Dennard, A. E., and Williams, R. J. P., in Transition Metal Chemistry (edit. by Carlin, R. L.), 2, 115 (1966).
Lindskog, S., and Ehrenberg, A., J. Mol. Biol., 24, 133 (1967).
Coleman, J. E., Oxygen Affinity of Haemogbbin and Red Cell Acid-Base Status, Alfred Benzon Symposium IV (Munksgaard, Copenhagen, in the press).
Lindskog, S., J. Biol. Chem., 238, 945 (1963).
Lindskog, S., and Nyman, P. O., Biochim. Biophys. Acta, 85, 462 (1964).
Barnes, W. H., Proc. Roy. Soc., A, 125, 670 (1929).
Fabry, M. E., Koenig, S. H., and Schillinger, W. E., J. Biol Chem., 245, 4256 (1970).
Coleman, J. E., J. Biol. Chem., 242, 5212 (1967).
Davis, R. P., J. Amer. Chem. Soc., 81, 5674 (1959).
Kernohan, J. C., Biochim. Biophys. Acta, 81, 346 (1964).
Pocker, Y., and Stone, J. T., J. Amer. Chem. Soc., 87, 5497 (1965).
Thorslund, A., and Lindskog, S., Europ. J. Biochem., 3, 117 (1967).
Riepe, M. E., and Wang, J. H., J. Biol. Chem., 243, 2779 (1968).
Ward, R. L., Biochemistry, 8, 1879 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LILJAS, A., KANNAN, K., BERGSTÉN, PC. et al. Crystal Structure of Human Carbonic Anhydrase C. Nature New Biology 235, 131–137 (1972). https://doi.org/10.1038/newbio235131a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/newbio235131a0
This article is cited by
-
Elucidating the role of metal ions in carbonic anhydrase catalysis
Nature Communications (2020)
-
Carbonic anhydrase - a universal enzyme of the carbon-based life
Photosynthetica (2017)
-
Equilibrium studies of copper ion adsorption onto modified kernel of date (Fructus dactylus)
International Journal of Environmental Science and Technology (2015)
-
Thermal decomposition study and biological characterization of zinc(II) 2-chlorobenzoate complexes with bioactive ligands
Journal of Thermal Analysis and Calorimetry (2013)
-
Modification of carbonic anhydrase II with acetaldehyde, the first metabolite of ethanol, leads to decreased enzyme activity
BMC Biochemistry (2008)