Abstract
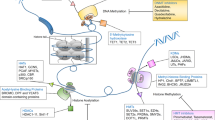
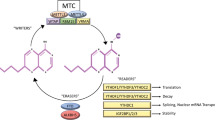
The methyl-CpG-binding domain (MBD) proteins 'read' and interpret the methylation moieties on DNA, and thus are critical mediators of many epigenetic processes. Currently, the MBD family comprises five members; MBD1, MBD2, MBD3, MBD4 and MeCP2. Although not a 'classical' MBD protein, Kaiso also mediates transcriptional repression by using zinc finger domains to bind its targets. Since DNA hypermethylation is a well-recognized mechanism underlying gene silencing events in both tumorigenesis and drug resistance, it is likely that the MBD proteins may be important modulators of tumorigenesis. We review the recent work addressing this possibility, and discuss several of the MBD proteins as potentially excellent novel therapeutic targets.
Key Points
-
Modulation of DNA methylation patterns and the proteins that interpret those patterns modifies the neoplastic process
-
Reduction in levels of DNMT1 reduces tumorigenesis in some tissues, but exacerbates it in other tissues
-
MBD2 deficiency has been shown to suppress intestinal neoplasia in animal models, with apparently little protumorigenic role in other tissues, so identifying it as a promising new therapeutic target
-
Kaiso deficiency has been shown to suppress intestinal neoplasia, identifying it as a promising therapeutic target
-
MBD4 deficiency has multiple effects upon tumorigenesis, but its primary consequence is an increase in mutation rates through failed DNA repair of spontaneous deamination events
-
Although implicated in the neoplastic process, the precise roles of MBD1, MBD3 and MeCP2 remain to be established
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ballestar E and Wolffe AP (2001) Methyl-CpG-binding proteins: targeting specific gene repression. Eur J Biochem 268: 1–6
Svedruzic ZM and Reich NO (2004) The mechanism of target base attack in DNA cytosine carbon 5 methylation. Biochemistry 43: 11460–11473
Urnov FD and Wolffe AP (2001) Chromatin remodeling and transcriptional activation: the cast (in order of appearance). Oncogene 20: 2991–3006
Jones PA and Baylin SB (2002) The fundamental role of epigenetic events in cancer. Nat Rev Genet 3: 415–428
Hendrich B and Tweedie S (2003) The methyl-CpG binding domain and the evolving role of DNA methylation in animals. Trends Genet 19: 269–277
Jones PA and Takai D (2001) The role of DNA methylation in mammalian epigenetics. Science 293: 1068–1070
Strathdee G et al. (1999) A role for methylation of the hMLH1 promoter in loss of hMLH1 expression and drug resistance in ovarian cancer. Oncogene 18: 2335–2341
Plumb JA et al. (2000) Reversal of drug resistance in human tumor xenografts by 2'-deoxy-5-azacytidine-induced demethylation of the hMLH1 gene promoter. Cancer Res 60: 6039–6044
Eads CA et al. (2002) Complete genetic suppression of polyp formation and reduction of CpG-island hypermethylation in Apc(Min/+) Dnmt1-hypomorphic mice. Cancer Res 62: 1296–1269
Lin H et al. (2006) Suppression of intestinal neoplasia by deletion of Dnmt3b. Mol Cell Biol 26: 2976–2983
Kantarjian HM and Issa JP (2005) Decitabine dosing schedules. Semin Hematol 42 (Suppl 2): S17–S22
Kantarjian H et al. (2006) Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer 106: 1794–1803
Li E et al. (1992) Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 69: 915–926
Jackson-Grusby L et al. (2001) Loss of genomic methylation causes p53-dependent apoptosis and epigenetic deregulation. Nat Genet 27: 31–39
Kim M et al. (2004) Dnmt1 deficiency leads to enhanced microsatellite instability in mouse embryonic stem cells. Nucleic Acids Res 32: 5742–5749
Gaudet F et al. (2003) Induction of tumors in mice by genomic hypomethylation. Science 300: 489–492
Yamada Y et al. (2005) Opposing effects of DNA hypomethylation on intestinal and liver carcinogenesis. Proc Natl Acad Sci USA 102: 13580–13585
Meehan RR et al. (1989) Identification of a mammalian protein that binds specifically to DNA containing methylated CpGs. Cell 58: 499–507
Lewis JD et al. (1992) Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69: 905–914
Ng HH et al. (1999) MBD2 is a transcriptional repressor belonging to the MeCP1 histone deacetylase complex. Nat Genet 23: 58–61
Cross SH et al. (1997) A component of the transcriptional repressor MeCP1 shares a motif with DNA methyltransferase and HRX proteins. Nat Genet 16: 256–259
Feng Q et al. (2002) Identification and functional characterization of the p66/p68 components of the MeCP1 complex. Mol Cell Biol 22: 536–546
Feng Q and Zhang Y (2001) The MeCP1 complex represses transcription through preferential binding, remodeling, and deacetylating methylated nucleosomes. Genes Dev 15: 827–832
Hendrich B and Bird A (1998) Identification and characterization of a family of mammalian methyl-CpG binding proteins. Mol Cell Biol 18: 6538–6547
Hendrich B et al. (1999) Genomic structure and chromosomal mapping of the murine and human Mbd1, Mbd2, Mbd3, and Mbd4 genes. Mamm Genome 10: 906–912
Prokhortchouk A et al. (2001) The p120 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev 15: 1613–1618
Nan X et al. (1998) Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393: 386–389
Jones PL et al. (1998) Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nat Genet 19: 187–191
Wade PA et al. (1999) Mi-2 complex couples DNA methylation to chromatin remodelling and histone deacetylation. Nat Genet 23: 62–66
Zhang Y et al. (1999) Analysis of the NuRD subunits reveals a histone deacetylase core complex and a connection with DNA methylation. Genes Dev 13: 1924–1935
Hendrich B et al. (1999) The thymine glycosylase MBD4 can bind to the product of deamination at methylated CpG sites. Nature 401: 301–304
Kondo E et al. (2005) The thymine DNA glycosylase MBD4 represses transcription and is associated with methylated p16(INK4a) and hMLH1 genes. Mol Cell Biol 25: 4388–4396
Fraga MF et al. (2003) The affinity of different MBD proteins for a specific methylated locus depends on their intrinsic binding properties. Nucleic Acids Res 31: 1765–1774
Saito M and Ishikawa F (2002) The mCpG-binding domain of human MBD3 does not bind to mCpG but interacts with NuRD/Mi2 components HDAC1 and MTA2. J Biol Chem 277: 35434–35439
Ballestar E et al. (2003) Methyl-CpG binding proteins identify novel sites of epigenetic inactivation in human cancer. EMBO J 22: 6335–6345
Lopez-Serra L et al. (2006) A profile of methyl-CpG binding domain protein occupancy of hypermethylated promoter CpG Islands of tumor suppressor genes in human cancer. Cancer Res 66: 8342–8346
Fujita N et al. (1999) Methylation-mediated transcriptional silencing in euchromatin by methyl-CpG binding protein MBD1 isoforms. Mol Cell Biol 19: 6415–6426
Villa R et al. (2006) The methyl-CpG binding protein MBD1 is required for PML-RARα function. Proc Natl Acad Sci USA 103: 1400–1405
Watanabe S et al. (2003) Methylated DNA-binding domain 1 and methylpurine-DNA glycosylase link transcriptional repression and DNA repair in chromatin. Proc Natl Acad Sci USA 100: 12859–12864
Bader S et al. (2003) MBD1, MBD2 and CGBP genes at chromosome 18q21 are infrequently mutated in human colon and lung cancers. Oncogene 22: 3506–3510
Zhao X et al. (2003) Mice lacking methyl-CpG binding protein 1 have deficits in adult neurogenesis and hippocampal function. Proc Natl Acad Sci USA 100: 6777–6782
Lewis JD et al. (1992) Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69: 905–914
Feng and Zhang (2001) The MeCP1 complex represses transcription through preferential binding, remodeling, and deacetylating methylated nucleosomes. Genes Dev 15: 827–832
Feng Q et al. (2002) Identification and functional characterization of the p66/p68 components of the MeCP1 complex. Mol Cell Biol 22: 536–546
Sekimata M et al. (2001) Involvement of a novel zinc finger protein, MIZF, in transcriptional repression by interacting with a methyl-CpG-binding protein, MBD2. J Biol Chem 276: 42632–42638
Hendrich B et al. (2001) Closely related proteins MBD2 and MBD3 play distinctive but interacting roles in mouse development. Genes Dev 15: 710–723
Sansom OJ et al. (2003) Deficiency of Mbd2 suppresses intestinal tumorigenesis. Nat Genet 34: 145–147
Suzuki H et al. (2004) Epigenetic inactivation of SFRP genes allows constitutive WNT signaling in colorectal cancer. Nat Genet 36: 417–422
Magdinier F and Wolffe AP (2001) Selective association of the methyl-CpG binding protein MBD2 with the silent p14/p16 locus in human neoplasia. Proc Natl Acad Sci USA 98: 4990–4995
Trinh BN et al. (2002) DNA methyltransferase deficiency modifies cancer susceptibility in mice lacking DNA mismatch repair. Mol Cell Biol 22: 2906–2917
Sansom OJ et al. (2005) MBD2 deficiency does not accelerate p53 mediated lymphomagenesis. Oncogene 24: 2430–2432
Campbell PM et al. (2004) Methylated DNA-binding protein 2 antisense inhibitors suppress tumourigenesis of human cancer cell lines in vitro and in vivo. Carcinogenesis 25: 499–507
Ivanov MA et al. (2003) Enhanced antitumor activity of a combination of MBD2-antisense electrotransfer gene therapy and bleomycin electrochemotherapy. Gene Med 5: 893–899
Zhang Y et al. (1999) Analysis of the NuRD subunits reveals a histone deacetylase core complex and a connection with DNA methylation. Genes Dev 13: 1924–1935
Zhu Y et al. (2004) Genetic and epigenetic analyses of MBD3 in colon and lung cancer. Br J Cancer 90: 1972–1975
Noh EJ et al. (2005) Methyl CpG-binding domain protein 3 mediates cancer-selective cytotoxicity by histone deacetylase inhibitors via differential transcriptional reprogramming in lung cancer cells. Cancer Res 65: 11400–11410
Jiang CL et al. (2002) MBD3L1 and MBD3L2, two new proteins homologous to the methyl-CpG-binding proteins MBD2 and MBD3: characterization of MBD3L1 as a testis-specific transcriptional repressor. Genomics 80: 621–629
Jin SG et al. (2005) MBD3L2 interacts with MBD3 and components of the NuRD complex and can oppose MBD2-MeCP1-mediated methylation silencing. J Biol Chem 280: 12700–12709
Prokhortchouk A (2006) Kaiso-deficient mice show resistance to intestinal cancer. Mol Cell Biol 26: 199–208
Prokhortchouk A et al. (2001) The p120 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev 15: 1613–1618
Daniel JM and Reynolds AB (1999) The catenin p120(ctn) interacts with Kaiso, a novel BTB/POZ domain zinc finger transcription factor. Mol Cell Biol 19: 3614–3623
Ruzov A et al. (2004) Kaiso is a genome-wide repressor of transcription that is essential for amphibian development. Development 131: 6185–6194
Kim SW et al. (2004) Non-canonical Wnt signals are modulated by the Kaiso transcriptional repressor and p120-catenin. Nat Cell Biol 6: 1212–1220
van Roy FM and McCrea PD (2005) A role for Kaiso–p120ctn complexes in cancer? Nat Rev Cancer 5: 956–964
Daniel JM et al. (2002) The p120(ctn)-binding partner Kaiso is a bi-modal DNA-binding protein that recognizes both a sequence-specific consensus and methylated CpG inucleotides. Nucleic Acids Res 30: 2911–2919
Rodova M et al. (2004) Regulation of the rapsyn promoter by kaiso and delta-catenin. Mol Cell Biol 24: 7188–7196
Park JI et al. (2005) Kaiso/p120-catenin and TCF/beta-catenin complexes coordinately regulate canonical Wnt gene targets. Dev Cell 8: 843–854
Filion GJ et al. (2006) A family of human zinc finger proteins that bind methylated DNA and repress transcription. Mol Cell Biol 26: 169–181
Geerdink N et al. (2002) MECP2 mutation in a boy with severe neonatal encephalopathy: clinical, neuropathological and molecular findings. Neuropediatrics 33: 33–36
Villard L et al. (2000) Two affected boys in a Rett syndrome family: clinical and molecular findings. Neurology 55: 1188–1193
Nan X et al. (1997) MeCP2 is a transcriptional repressor with abundant binding sites in genomic chromatin. Cell 88: 471–481
Nan X et al. (1998) Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393: 386–389
Klose RJ et al. (2005) DNA binding selectivity of MeCP2 due to a requirement for A/T sequences adjacent to methyl-CpG. Mol Cell 19: 667–678
Bernard D et al. (2006) The methyl-CpG-binding protein MECP2 is required for prostate cancer cell growth. Oncogene 25: 1358–1366
Muller HM et al. (2003) MeCP2 and MBD2 expression in human neoplastic and non-neoplastic breast tissue and its association with oestrogen receptor status. Br J Cancer 89: 1934–1939
Nakagawachi T et al. (2003) Silencing effect of CpG island hypermethylation and histone modifications on O6-methylguanine-DNA methyltransferase (MGMT) gene expression in human cancer. Oncogene 22: 8835–8844
Bellacosa A (2001) Role of MED1 (MBD4) gene in DNA repair and human cancer. J Cell Physiol 187: 137–144
Kouidou S et al. (2006) Methylation and repeats in silent and nonsense mutations of p53. Mutat Res 599: 167–177
Millar CB et al. (2002) Enhanced CpG mutability and tumorigenesis in MBD4-deficient mice. Science 297: 403–405
Wong E et al. (2002) Mbd4 inactivation increases C-T transition mutations and promotes gastrointestinal tumor formation. Proc Natl Acad Sci USA 99: 14937–14942
Fritzell JA et al. (1997) Role of DNA mismatch repair in the cytotoxicity of ionizing radiation. Cancer Res 57: 5143–5147
Toft NJ et al. (1999) Msh2 status modulates both apoptosis and mutation frequency in the murine small intestine. Proc Natl Acad Sci USA 96: 3911–3915
Meyers M et al. (2001) Role of the hMLH1 DNA mismatch repair protein in fluoropyrimidine-mediated cell death and cell cycle responses. Cancer Res 61: 5193–5201
Sansom OJ et al. (2003) MBD4 deficiency reduces the apoptotic response to DNA damaging agents in the murine small intestine. Oncogene 22: 7130–7136
Cortellino S et al. (2003) The base excision repair enzyme MED1 mediates DNA damage response to antitumor drugs and is associated with mismatch repair system integrity. Proc Natl Acad Sci USA 100: 15071–15076
Parsons BL et al. (2003) MED1: a central molecule for maintenance of genome integrity and response to DNA damage. Proc Natl Acad Sci USA 100: 14601–14602
Sansom OJ et al. (2004) MBD4 deficiency does not increase mutation or accelerate tumourigenesis in mice lacking MMR. Oncogene 23: 5693–5696
Screaton RA et al. (2003) Fas-associated death domain protein interacts with methyl-CpG binding domain protein 4: a potential link between genome surveillance and apoptosis. Proc Natl Acad Sci USA 100: 5211–5216
Bader S et al. (1999) Somatic frameshift mutations in the MBD4 gene of sporadic colon cancers with mismatch repair deficiency. Oncogene 18: 8044–8047
Wade PA (2001) Methyl CpG-binding proteins and transcriptional repression. Bioessays 23: 1131–1137
Le Guezennec X et al. (2006) MBD2/NuRD and MBD3/NuRD, two distinct complexes with different biochemical and functional properties. Mol Cell Biol 26: 843–851
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sansom, O., Maddison, K. & Clarke, A. Mechanisms of Disease: methyl-binding domain proteins as potential therapeutic targets in cancer. Nat Rev Clin Oncol 4, 305–315 (2007). https://doi.org/10.1038/ncponc0812
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc0812
This article is cited by
-
Methyl-CpG-binding domain protein 2 contributes to renal fibrosis through promoting polarized M1 macrophages
Cell Death & Disease (2022)
-
Loss of MBD2 attenuates MLL-AF9-driven leukemogenesis by suppressing the leukemic cell cycle via CDKN1C
Oncogenesis (2021)
-
Expression of DnMTs and MBDs in AlCl3-Induced Neurotoxicity Mouse Model
Biological Trace Element Research (2021)
-
Association between gene promoter methylation of the one-carbon metabolism pathway and serum folate among patients with hyperhomocysteinemia
European Journal of Clinical Nutrition (2020)
-
High Resolution Imaging of DNA Methylation Dynamics using a Zebrafish Reporter
Scientific Reports (2017)