Abstract
Functional androgen receptor (AR) signaling is necessary for the development of prostate cancer. The therapeutic effect of androgen deprivation therapy for prostate cancer was described over 60 years ago and this treatment remains the mainstay of systemic therapy despite its transient response duration. It has become clear that AR expression and signaling remains intact as the disease evolves from androgen-sensitive cancer to classically (but perhaps inaccurately) termed hormone refractory prostate cancer. Through several genetic and epigenetic adaptations, prostate tumors continue to rely on AR growth signaling and they thus remain targets of 'hormonal' therapy. The development of new strategies and drugs that can abrogate AR signaling will probably result in important clinical benefits. The biology of androgen independence and the development of new approaches targeting AR signaling are reviewed herein.
Key Points
-
More-effective 'hormone therapy' is needed for prostate cancer
-
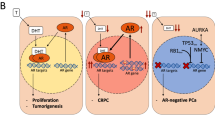
Despite the development of classically termed 'hormone refractory' prostate cancer, androgen receptor signaling remains an active growth-promoting pathway
-
The androgen receptor remains an important therapeutic target in hormone refractory prostate cancer
-
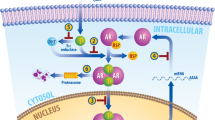
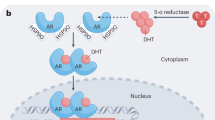
As prostate tumors develop in castrated men, AR signaling is maintained through AR amplification and mutation and the 'intracrine' production of androgen
-
Novel approaches to androgen receptor inhibition are in development and include more-potent antiandrogens, lyase inhibitors, 5α-reductase inhibitors, HSP90 inhibitors, and HDAC inhibitors
-
More-effective 'hormone therapy' might be achieved through combined inhibition of classic androgen receptor signaling and other interrelated pathways such as mTOR, EGFR or MAPK
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Caskey C et al. (1992) Triplet repeat mutations in human disease. Science 256: 784–789
Griffin J (1992) Androgen resistance—the clinical and molecular spectrum. N Engl J Med 326: 611–618
Huggins C and Hodges C (1941) The effect of castration, of oestrogen and of androgen injections on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res 1: 293–297
Evans R (1988) The steroid and thyroid hormone receptor super family. Science 240: 889–895
Pratt W and Toft D (1997) Steroid receptor interactions with heat shock protein and immunophilin charperones. Endocr Rev 18: 306–360
Veldscholte J et al. (1992) Hormone-induced dissociation of the androgen receptor-heat shock protein complex: use of a new monoclonal antibody to distinguish transformed from nontransformed receptors. Biochemistry 31: 7422–7430
Dehm SM and Tindall DJ (2005) Regulation of androgen receptor signaling in prostate cancer. Expert Rev Anticancer Ther 5: 63–74
Shang Y et al. (2002) Formation of the androgen receptor transcription complex. Mol Cell 9: 601–610
Chang CY and McDonnell DP (2005) Androgen receptor-cofactor interactions as targets for new drug discovery. Trends Pharmacol Sci 26: 225–228
Mckenna J et al. (1999) Nuclear receptor coregulators: cellular and molecular biology. Endocr Rev 20: 321–344
Velasco A et al. (2004) Identification and validation of novel androgen-regulated genes in prostate cancer. Endocrinology 145: 3913–3924
Heinein C and Chang C (2002) The roles of androgen receptors and androgen-binding proteins in nongenomic androgen actions. Mol Endocrinol 16: 2181–2187
Ruizeveld de Winter JA et al. (1991) Androgen receptor expression in human tissues: an immunohistochemical study. J Histochem Cytochem 39: 927–936
de Vere White R et al. (1997) Human androgen receptor expression in prostate cancer following androgen ablation. Eur Urol 31: 1–6
Sasaki M et al. (2002) Methylation and inactivation of estrogen, progesterone, and androgen receptors in prostate cancer. J Natl Cancer Inst 94: 384–390
Giovannucci E et al. (1997) The CAG repeat within the androgen receptor gene and its relationship to prostate cancer. Proc Natl Acad Sci USA 94: 3320–3323
Irvine R et al. (1995) The CAG and GGC microsatellites of the androgen receptor gene are in linkage disequilibrium in men with prostate cancer. Cancer Res 55: 1937–1940
Taplin ME and Balk SP (2004) Androgen receptor: a key molecule in the progression of prostate cancer to hormone independence. J Cell Biochem 91: 483–490
Chamberlain N et al. (1994) The length and location of the CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res 22: 3181–3186
Choong C et al. (1996) Reduced androgen receptor gene expression with first exon CAG repeat expansion. Mol Endocrinol 10: 1527–1535
Eisenberger M et al. (1998) Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N Engl J Med 339: 1036–1042
Moul JW et al. (2004) Early versus delayed hormonal therapy for prostate specific antigen only recurrence of prostate cancer after radical prostatectomy. J Urol 171: 1141–1147
Bubendorf L et al. (1999) Survey of gene amplifications during prostate cancer progression by high-throughout fluorescence in situ hybridization on tissue microarrays. Cancer Res 59: 803–806
Edwards J et al. (2003) Androgen receptor gene amplification and protein expression in hormone refractory prostate cancer. Br J Cancer 89: 552–556
Ford OH et al. (2003) Androgen receptor gene amplification and protein expression in recurrent prostate cancer. J Urol 170: 1817–1821
Koivisto P et al. (1997) Androgen receptor gene amplification: a possible molecular mechanism for androgen deprivation therapy failure in prostate cancer. Cancer Res 57: 314–319
Linja M et al. (2001) Amplification and over expression of androgen receptor gene in hormone-refractory prostate cancer. Cancer Res 61: 3550–3555
Visakorpi T et al. (1995) In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat Genet 9: 401–406
Palmberg C et al. (2000) Androgen receptor gene amplificaiton at primary progression predicts response to combined androgen blockade as second line therapy for advanced prostate cancer. J Urol 164: 1992–1995
Veldscholte J et al. (1990) A mutation in the ligand binding domain of the androgen receptor of human LNCaP cells affects steroid binding characteristics and response to anti-androgens. Biochem Biophys Res Commun 173: 534–540
Gottlieb B et al. (2004) The androgen receptor gene mutations database (ARDB): 2004 update. Hum Mutat 23: 527–533
Gelmann E (2002) Molecular biology of the androgen receptor. J Clin Oncol 20: 3001–3015
Taplin ME et al. (1999) Selection for androgen receptor mutations in prostate cancer treated with androgen antagonist. Cancer Res 59: 2511–2515
Fenton M et al. (1997) Functional characterization of mutant androgen receptors from androgen independent prostate cancer. Clin Cancer Res 3: 1383–1388
Joyce R et al. (1998) High dose bicalutamide for androgen independent prostate cancer: Effect of prior hormonal therapy. J Urol 159: 149–153
Taplin ME et al. (2003) Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia group B study 9663. J Clin Oncol 21: 2673–2678
Culig Z et al. (1993) Mutant androgen receptor detected in an advanced-stage prostate carcinoma is activated by adrenal androgen and progesterone. Mol Endocrinol 7: 1541–1550
Culig Z et al. (1996) Activation of two mutant androgen receptors from human prostatic carcinoma by adrenal androgens and metabolic derivatives of testosterone. Cancer Detect Prev 20: 68–75
Zhao X et al. (2000) Glucocorticoids can promote androgen-independent growth of prostate cancer cells through a mutated androgen receptor. Nat Med 6: 703–706
Buchanan G et al. (2001) Mutations at the boundary of the hinge and ligand binding domain of the androgen receptor confer increased transactivation function. Mol Endocrinol 15: 46–56
Ceraline J et al. (2004) Constitutive activation of the androgen receptor by a point mutation in the hinge region: a new mechanism for androgen-independent growth in prostate cancer. Int J Cancer 108: 152–157
Gregory C et al. (2001) A mechanism for androgen receptor-mediated prostate cancer recurrence after androgen deprivation therapy. Cancer Res 61: 4315–4319
Halkidou K et al. (2003) Expression of Tip60, an androgen receptor coactivator, and its role in prostate cancer development. Oncogene 22: 2466–2477
Papaioannou M et al. (2005) Co-activator and co-repressor interplay on the human androgen receptor. Andrologia 37: 211–213
Ueda T et al. (2002) Activation of the androgen receptor N-terminal domain by interleukin-6 via MAPK and STAT3 signal transduction pathways. J Biol Chem 277: 7076–7085
Grossmann M et al. (2001) Androgen receptor signaling in androgen-refractory prostate cancer. J Natl Cancer Inst 93: 1687–1697
Ford C et al. (1998) Molecular basis for interactions of G proteins betagamma subunits with effectors. Science 280: 1271–1274
Kasbohm E et al. (2005) Androgen receptor activation by Gs signaling in prostate cancer cells. J Biol Chem 280: 11583–11589
Waxman J et al. (1983) Treatment with gonadotropin-releasing hormone analogue in advanced prostatic cancer. BMJ 286: 1309–1312
Labrie F et al. (2005) Is dehydroepiandrosterone a hormone? J Endocrinol 187: 169–196
Mohler J et al. (2004) The androgen axis in recurrent prostate cancer. Clin Cancer Res 10: 440–448
Scher H and Sawyers C (2005) Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol 23: 8253–8261
Stanbrough M et al. (2006) Increased expression of genes converting adrenal androgens to testosterone in androgen-independent prostate cancer. Cancer Res 66: 2815–2825
Small EJ and Vogelzang NJ (1997) Second-line hormonal therapy for advanced prostate cancer: a shifting paradigm. J Clin Oncol 15: 382–388
Small EJ et al. (2004) Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol 22: 1025–1033
Bhanalaph T et al. (1974) Current status of bilateral adrenalectomy on advanced prostate carcinoma. Ann Surg 179: 17–23
Attard G et al. (2005) Selective blockade of androgenic steroid synthesis by novel lyase inhibitors as a therapeutic strategy for treating metastatic prostate cancer. BJU Int 96: 1241–1246
Miller W et al. (1997) The regulation of 17, 20 lyase activity. Steroids 62: 133–142
O'Donnell A et al. (2004) Hormonal impact of the 17alpha-hydroxylase/C(17,20)-lyase inhibitor abiraterone acetate (CB7630) in patients with prostate cancer. Br J Cancer 90: 2317–2325
Thompson I et al. (2003) The influence of finasteride on the development of prostate cancer. N Engl J Med 349: 215–224
Andriole G et al. (2004) Effect of the dual 5α-reductase inhibitor dutasteride on markers of tumor regression in prostate cancer. J Urol 172: 915–919
Eisenberger M et al. (2004) Phase I and clinical pharmacology of a type I and II, 5-alpha-reductase inhibitor (LY320236) in prostate cancer: elevation of estradiol as possible mechanism of action. Urology 63: 114–119
Brown T (2004) Nonsteroidal selective androgen receptor modulators (SARMs): designer androgen with flexible structures provide clinical promise. Endocrinology 145: 5420–5428
[No authors listed] (2000) Maximum androgen blockade in advanced prostate cancer: an overview of the randomized trials. Lancet 355: 1491–1498
Samson D et al. (2002) Systemic review and meta-analysis of monotherapy compared with combined androgen blockade for patients with advanced prostate carcinoma. Cancer 95: 361–376
Furr B (1995) Casodex: preclinical studies and controversies. Ann NY Acad Sci 761: 79–96
Bohl C et al. (2005) Structural basis for antagonism and resistance of bicalutamide in prostate cancer. Proc Natl Acad Sci USA 102: 6201–6206
Salvati M et al. (2005) Identification of a novel class of androgen receptor antagonists based on the bicyclic-1H-isoindole-1,3(2H)-dione nucleus. Bioorg Med Chem Lett 15: 389–393
Hodgson M et al. (2005) The androgen receptor recruits nuclear receptor corepressor (N-CoR) in the presence of mifepristone via its N and C termini revealing a novel molecular mechanism for androgen receptor antagonists. J Biol Chem 280: 6511–6519
Hayes J et al. (2006) Efficacy of mifepristone (RU-486) in androgen independent prostate cancer [abstract #235]. Presented at the 2006 Prostate Cancer Symposium: 2006 February 24–26; San Francisco, CA
Chang C et al. (2005) Development of peptide antagonists for the androgen receptor using combinatorial peptide phage display. Mol Endocrinol 19: 2478–2490
Banerji U et al. (2005) Phase I pharmacokinetic and pharmacodynamic study of 17-Allylmino, 17-demethoxygeldannmycin in patients with advanced malignancies. J Clin Oncol 23: 4152–4161
Solit D et al. (2003) Hsp90 as a therapeutic target in prostate cancer. Semin Oncol 30: 709–716
Goetz MP et al. (2005) Phase I trial of 17-allylamino-17-demethoxygeldanamycin in patients with advanced cancer. J Clin Oncol 23: 1078–1087
Grem J et al. (2005) Phase I and pharmacologic study of 17-(allylamino)-17-demethoxygeldanamycin in adult patients with solid tumors. J Clin Oncol 23: 1885–1893
Yu X et al. (2005) Hsp90 inhibitors identified from a library of novobiocin analogues. J Am Chem Soc 127: 1278–1282
Minucci S and Pelicci G (2006) Histone decacetylase inhibitiors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer 6: 38–51
Carducci M et al. (2001) A phase I clinical and pharmacological evaluation of sodium phenylbutyrate on an 120-h infusion schedule. Clin Cancer Res 7: 3047–3055
Kelly W et al. (2003) Phase I clinical trial of histone deactylase inhibitor: suberylanilide hydroxamic acid (SAHA). Clin Cancer Res 9: 3578–3588
Morris M et al. (2002) HER-2 profiling and targeting in prostate carcinoma. Cancer 94: 980–986
Solit D et al. (2004) Phase I pharmacokinetic and pharmacodynamic trial of docetaxel and 17AAG (17-allylamino-17-demethoxygeladanamycin) [abstract #3032]. J Clin Oncol 22
Marquis J et al. (2005) disruption of gene expression and induction of apoptosis in prostate cancer cells by a DNA-damaging agent tethered to an androgen receptor ligand. Chem Biol 12: 779–787
Haag P et al. (2005) Androgen receptor down regulation by small interference RNA induces cell growth inhibition in androgen sensitive as well as in androgen independent prostate cancer cells. J Steroid Biochem Mol Biol 96: 251–258
Papatsoris A et al. (2005) Novel biological agents for the treatment of hormone refractory prostate cancer (HRPC). Curr Med Chem 12: 277–296
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Taplin, ME. Drug Insight: role of the androgen receptor in the development and progression of prostate cancer. Nat Rev Clin Oncol 4, 236–244 (2007). https://doi.org/10.1038/ncponc0765
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc0765
This article is cited by
-
Flare on [18F]PSMA-1007 PET/CT after short-term androgen deprivation therapy and its correlation to FDG uptake: possible marker of tumor aggressiveness in treatment-naïve metastatic prostate cancer patients
European Journal of Nuclear Medicine and Molecular Imaging (2023)
-
Targeting signaling pathways in prostate cancer: mechanisms and clinical trials
Signal Transduction and Targeted Therapy (2022)
-
Nuclear βArrestin1 regulates androgen receptor function in castration resistant prostate cancer
Oncogene (2021)
-
Transcriptional suppression of androgen receptor by 18β-glycyrrhetinic acid in LNCaP human prostate cancer cells
Archives of Pharmacal Research (2020)
-
Design, synthesis, and biological evaluation of 2-(5-methyl-1H-pyrazol-1-yl) acetamide derivatives as androgen receptor antagonists
Medicinal Chemistry Research (2019)