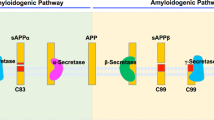
Abstract
Alzheimer's disease (AD) is the most common cause of age-related dementia. Pathologically, AD is characterized by the deposition in the brain of amyloid-β peptides derived from proteolysis of amyloid precursor protein (APP) by β-site APP cleaving enzyme 1 (BACE1) and γ-secretase. A growing body of evidence implicates cholesterol and cholesterol-rich membrane microdomains in amyloidogenic processing of APP. Here, we review recent findings regarding the association of BACE1, γ-secretase and APP in lipid rafts, and discuss potential therapeutic strategies for AD that are based on knowledge gleaned from the membrane environment that fosters APP processing.
Key Points
-
Cholesterol has been an important focus of research into Alzheimer's disease, but controversy has emerged regarding the therapeutic value of lowering cholesterol as a protective measure against this disease
-
Certain aspects of cholesterol metabolism regulate amyloid-β production through direct modulation of amyloid precursor protein (APP) processing by secretases
-
β-Site APP cleaving enzyme 1, γ-secretase and APP C-terminal fragments are localized in lipid rafts, which are membrane microdomains rich in cholesterol and sphingolipids
-
Amyloidogenic processing of APP occurs in lipid raft domains, and experimental manipulations that disturb the integrity of lipid rafts affect amyloid-β production
-
Compartmentalization of APP C-terminal fragments and secretases offers unique opportunities to selectively target APP processing
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Alzheimer A (1907) On a peculiar disease of the cerebral cortex [German]. Allgemeine Zeitschrift für Psychiatrie und Psychisch-Gerichtliche Medizin 64: 146–148
Vassar R (2004) BACE1: the β-secretase enzyme in Alzheimer's disease. J Mol Neurosci 23: 105–114
Allinson TM et al. (2003) ADAMs family members as amyloid precursor protein alpha-secretases. J Neurosci Res 74: 342–352
Iwatsubo T (2004) The γ-secretase complex: machinery for intramembrane proteolysis. Curr Opin Neurobiol 14: 379–383
Kitazume S et al. (2001) Alzheimer's β-secretase, β-site amyloid precursor protein-cleaving enzyme, is responsible for cleavage secretion of a Golgi-resident sialyltransferase. Proc Natl Acad Sci USA 98: 13554–13559
Li Q et al. (2004) Cleavage of amyloid-β precursor protein and amyloid-β precursor-like protein by BACE 1. J Biol Chem 279: 10542–10550
Lichtenthaler SF et al. (2003) The cell adhesion protein P-selectin glycoprotein ligand-1 is a substrate for the aspartyl protease BACE1. J Biol Chem 278: 48713–48719
von Arnim CA et al. (2005) The low density lipoprotein receptor-related protein (LRP) is a novel β-secretase (BACE1) substrate. J Biol Chem 280: 17777–17785
Wong HK et al. (2005) β Subunits of voltage-gated sodium channels are novel substrates of β-site amyloid precursor protein-cleaving enzyme (BACE1) and γ-secretase. J Biol Chem 280: 23009–23017
Hu X et al. (2006) Bace1 modulates myelination in the central and peripheral nervous system. Nat Neurosci 9: 1520–1525
Willem M et al. (2006) Control of peripheral nerve myelination by the β-secretase BACE1. Science 314: 664–666
Lazarov O et al. (2002) Evidence that synaptically released β-amyloid accumulates as extracellular deposits in the hippocampus of transgenic mice. J Neurosci 22: 9785–9793
Koo EH et al. (1990) Precursor of amyloid protein in Alzheimer disease undergoes fast anterograde axonal transport. Proc Natl Acad Sci USA 87: 1561–1565
Sisodia SS (1992) β-amyloid precursor protein cleavage by a membrane-bound protease. Proc Natl Acad Sci USA 89: 6075–6079
Huse JT et al. (2000) Maturation and endosomal targeting of β-site amyloid precursor protein-cleaving enzyme: the Alzheimer's disease β-secretase. J Biol Chem 275: 33729–33737
Koo EH et al. (1994) Evidence that production and release of amyloid β-protein involves the endocytic pathway. J Biol Chem 269: 17386–17389
Thinakaran G et al. (1996) Metabolism of the “Swedish” amyloid precursor protein variant in neuro2a (N2a) cells. Evidence that cleavage at the “β-secretase” site occurs in the Golgi apparatus. J Biol Chem 271: 9390–9397
Vetrivel KS et al. (2004) Association of γ-secretase with lipid rafts in post-Golgi and endosome membranes. J Biol Chem 279: 44945–44954
Simons K et al. (2000) Lipid rafts and signal transduction. Nat Rev Mol Cell Biol 1: 31–39
Brown DA et al. (1998) Functions of lipid rafts in biological membranes. Annu Rev Cell Dev Biol 14: 111–136
Lichtenberg D et al. (2005) Detergent-resistant membranes should not be identified with membrane rafts. Trends Biochem Sci 30: 430–436
Pike LJ (2006) Rafts defined: a report on the Keystone Symposium on Lipid Rafts and Cell Function. J Lipid Res 47: 1597–1598
Hancock JF (2006) Lipid rafts: contentious only from simplistic standpoints. Nat Rev Mol Cell Biol 7: 456–462
Benjannet S et al. (2001) Post-translational processing of beta-secretase (β-amyloid-converting enzyme) and its ectodomain shedding. The pro- and transmembrane/cytosolic domains affect its cellular activity and amyloid-β production. J Biol Chem 276: 10879–10887
Riddell DR et al. (2001) Compartmentalization of β-secretase (Asp2) into low-buoyant density, noncaveolar lipid rafts. Curr Biol 11: 1288–1293
Cordy JM et al. (2003) Exclusively targeting β-secretase to lipid rafts by GPI-anchor addition up-regulates β-site processing of the amyloid precursor protein. Proc Natl Acad Sci USA 100: 11735–11740
Hattori C et al. (2006) BACE1 interacts with lipid raft proteins. J Neurosci Res 84: 912–917
Ehehalt R et al. (2003) Amyloidogenic processing of the Alzheimer β-amyloid precursor protein depends on lipid rafts. J Cell Biol 160: 113–123
Abad-Rodriguez J et al. (2004) Neuronal membrane cholesterol loss enhances amyloid peptide generation. J Cell Biol 167: 953–960
Vetrivel KS et al. (2005) Spatial segregation of γ-secretase and substrates in distinct membrane domains. J Biol Chem 280: 25892–25900
Escriba PV (2006) Membrane-lipid therapy: a new approach in molecular medicine. Trends Mol Med 12: 34–43
Kuo YM et al. (1998) Elevated low-density lipoprotein in Alzheimer's disease correlates with brain Aβ 1–42 levels. Biochem Biophys Res Commun 252: 711–715
Kivipelto M et al. (2001) Midlife vascular risk factors and Alzheimer's disease in later life: longitudinal, population based study. BMJ 322: 1447–1451
Jick H et al. (2000) Statins and the risk of dementia. Lancet 356: 1627–1631
Wolozin B et al. (2000) Decreased prevalence of Alzheimer disease associated with 3-hydroxy-3-methyglutaryl coenzyme A reductase inhibitors. Arch Neurol 57: 1439–1443
Sparks DL et al. (1994) Induction of Alzheimer-like β-amyloid immunoreactivity in the brains of rabbits with dietary cholesterol. Exp Neurol 126: 88–94
Simons M et al. (1998) Cholesterol depletion inhibits the generation of β-amyloid in hippocampal neurons. Proc Natl Acad Sci USA 95: 6460–6464
Fassbender K et al. (2001) Simvastatin strongly reduces levels of Alzheimer's disease β-amyloid peptides Aβ42 and Aβ40 in vitro and in vivo. Proc Natl Acad Sci USA 98: 5856–5861
Refolo LM et al. (2000) Hypercholesterolemia accelerates the Alzheimer's amyloid pathology in a transgenic mouse model. Neurobiol Dis 7: 321–331
Refolo LM et al. (2001) A cholesterol-lowering drug reduces β-amyloid pathology in a transgenic mouse model of Alzheimer's disease. Neurobiol Dis 8: 890–899
Miida T et al. (2005) Can statin therapy really reduce the risk of Alzheimer's disease and slow its progression? Curr Opin Lipidol 16: 619–623
Hoyer S et al. (2007) Alzheimer disease—no target for statin treatment: a mini review. Neurochem Res 32: 695–706
Canevari L et al. (2007) Alzheimer's disease and cholesterol: the fat connection. Neurochem Res 32: 739–750
Wolozin B et al. (2006) Re-assessing the relationship between cholesterol, statins and Alzheimer's disease. Acta Neurol Scand Suppl 185: 63–70
Kojro E et al. (2001) Low cholesterol stimulates the nonamyloidogenic pathway by its effect on the α-secretase ADAM 10. Proc Natl Acad Sci USA 98: 5815–5820
Hansen GH et al. (2000) Cholesterol depletion of enterocytes: effect on the Golgi complex and apical membrane trafficking. J Biol Chem 275: 5136–5142
Hao M et al. (2004) Effects of cholesterol depletion and increased lipid unsaturation on the properties of endocytic membranes. J Biol Chem 279: 14171–14178
Kirsch C et al. (2003) Statin effects on cholesterol micro-domains in brain plasma membranes. Biochem Pharmacol 65: 843–856
Runz H et al. (2002) Inhibition of intracellular cholesterol transport alters presenilin localization and amyloid precursor protein processing in neuronal cells. J Neurosci 22: 1679–1689
Sun Y et al. (2003) Expression of liver X receptor target genes decreases cellular amyloid β peptide secretion. J Biol Chem 278: 27688–27694
Puglielli L et al. (2001) Acyl-coenzyme A: cholesterol acyltransferase modulates the generation of the amyloid β-peptide. Nat Cell Biol 3: 905–912
Cole SL et al. (2005) Statins cause intracellular accumulation of amyloid precursor protein, β-secretase-cleaved fragments, and amyloid β-peptide via an isoprenoid-dependent mechanism. J Biol Chem 280: 18755–18770
Sawamura N et al. (2004) Modulation of amyloid precursor protein cleavage by cellular sphingolipids. J Biol Chem 279: 11984–11991
Grimm MO et al. (2005) Regulation of cholesterol and sphingomyelin metabolism by amyloid-β and presenilin. Nat Cell Biol 7: 1118–1123
Smotrys JE et al. (2004) Palmitoylation of intracellular signaling proteins: regulation and function. Annu Rev Biochem 73: 559–587
Scheiffele P et al. (1997) Interaction of influenza virus haemagglutinin with sphingolipid-cholesterol membrane domains via its transmembrane domain. EMBO J 16: 5501–5508
Bruckner K et al. (1999) EphrinB ligands recruit GRIP family PDZ adaptor proteins into raft membrane microdomains. Neuron 22: 511–524
Sidera C et al. (2005) Post-translational processing of beta-secretase in Alzheimer's disease. Proteomics 5: 1533–1543
Crameri A et al. (2006) The role of seladin-1/DHCR24 in cholesterol biosynthesis, APP processing and Aβ generation in vivo. EMBO J 25: 432–443
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Cheng, H., Vetrivel, K., Gong, P. et al. Mechanisms of Disease: new therapeutic strategies for Alzheimer's disease—targeting APP processing in lipid rafts. Nat Rev Neurol 3, 374–382 (2007). https://doi.org/10.1038/ncpneuro0549
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0549
This article is cited by
-
γ-Secretase in Alzheimer’s disease
Experimental & Molecular Medicine (2022)
-
Donepezil effects on cholesterol and oxysterol plasma levels of Alzheimer’s disease patients
European Archives of Psychiatry and Clinical Neuroscience (2018)
-
Membrane Aging as the Real Culprit of Alzheimer’s Disease: Modification of a Hypothesis
Neuroscience Bulletin (2018)
-
Alzheimer disease: modeling an Aβ-centered biological network
Molecular Psychiatry (2016)
-
Cellular Membrane Fluidity in Amyloid Precursor Protein Processing
Molecular Neurobiology (2014)