Abstract
The development of crop varieties that are better suited to new climatic conditions is vital for future food production1,2. Increases in mean temperature accelerate crop development, resulting in shorter crop durations and reduced time to accumulate biomass and yield3,4. The process of breeding, delivery and adoption (BDA) of new maize varieties can take up to 30 years. Here, we assess for the first time the implications of warming during the BDA process by using five bias-corrected global climate models and four representative concentration pathways with realistic scenarios of maize BDA times in Africa. The results show that the projected difference in temperature between the start and end of the maize BDA cycle results in shorter crop durations that are outside current variability. Both adaptation and mitigation can reduce duration loss. In particular, climate projections have the potential to provide target elevated temperatures for breeding. Whilst options for reducing BDA time are highly context dependent, common threads include improved recording and sharing of data across regions for the whole BDA cycle, streamlining of regulation, and capacity building. Finally, we show that the results have implications for maize across the tropics, where similar shortening of duration is projected.
Similar content being viewed by others
Main
By 2050 the majority of African countries will have significant experience of novel climates1. However, precise information as to when novel climates will occur has not been available until the recent development of techniques to identify the time of emergence of climate change signals5,6. These techniques quantify the signal of a change in climate relative to the background ‘noise’ of current climate variability. Metrics that capture the response of crops to single or multiple aspects of weather or climate (crop–climate indices7) are another tool that has been developed intensively in recent years. Alongside crop yield modelling, these techniques now enable assessments of the projected times at which climate change will alter crop productivity. These alterations are mediated through both crop growth (that is, photosynthesis and biomass accumulation) and development (phenological and morphological responses).
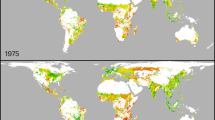
We use seven crop–climate indices (Supplementary Table S2) to identify when heat stress, drought stress and crop duration (that is, time from germination to maturity) become systematically and significantly outside the ranges at present experienced by maize cultivation in sub-Saharan Africa. Crop breeders have long been aware of the need to develop new crop varieties that are suited to future climates, particularly with respect to heat and drought stress8,9. Heat stress impacts are evident in our analysis. However, heat stress indices are not sufficiently constrained at present (that is, uncertainty in their values is too great) for detection of a climate change signal; only the signal in crop duration changes exceeded the noise of climate variability and thus showed a time of emergence within this century (see Methods). The time of emergence of altered crop duration depends on both future emissions and location. For the current emissions trajectory (representative concentration pathways 8.5, RCP8.5) crop duration becomes systematically and significantly shorter than current ranges as early as 2018 in some locations and by 2031 in the majority of maize-growing grid cells (Fig. 1). Crops with these shorter durations will make less use of available rains and solar radiation, implying reduced yields3,4.
The specified year refers to the midpoint of the 20-year period in which the median crop duration falls below the 25th quantile of the baseline period (1995–2014). Grey cells indicate that the crop duration remains within the 25th–75th quantile until at least 2038—the latest possible delivery date for a BDA cycle beginning in 2004 (see Table 1). There are no instances of crop duration exceeding the 75th quantile.
The length of time taken to develop and disseminate maize varieties adapted to novel conditions is dependent on access to appropriate germplasm; phenotyping capacity and precision; choice of selection strategy; suitability, frequency and reliability of conditions for introgression and backcrossing (including the number of growing seasons per year); national level requirements for variety testing and approval; the efficiency of public and private seed systems in making new seed available and accessible; and factors affecting rates of adoption among farmers, such as the effectiveness of extension service provision and consumer acceptance (Table 1).
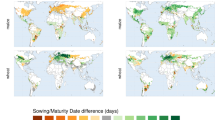
The emergence of new thermal environments (Fig. 1) presents an important challenge. Changes in mean temperature between the start of breeding and the final time of adoption imply that times to crop maturity in farmers’ fields may differ from the values during the breeding process. If duration loss during the BDA cycle brings maturity dates earlier than those observed in the current climate then there will be a mismatch between expected and actual maturity dates. Current estimates of BDA times for African maize suggest that this is commonly the case (Fig. 2). The magnitude of the challenge varies spatially (Supplementary Extended Data Fig. 2), with fewer days of crop duration lost per year in the Sahel and in Mozambique; however, these predominantly dry lowland areas are relatively minor producers of maize across sub-Saharan Africa (see Methods). In the absence of adaptive measures, the duration changes in Fig. 2 imply yield reductions of the order of 2.5–5% for most of Africa for worst-case (that is, longest) BDA scenarios under RCP8.5. A detailed analysis of yield reductions is presented in Supplementary Text S2.
a, Shortest (best-case, solid line) and longest (worst-case, dashed line) BDA times. b, Mean BDA time. Baseline variability in crop duration (25th to 75th quantile for the period 1995 to 2014) across all RCPs is shaded in grey (the baseline variability for each RCP is very similar, see Supplementary Extended Data Fig. 1). The change in the number of days was calculated using 20-year moving medians over the time period 1995–2050.
If there are strong monotonic temperature trends during selection and breeding then the selection process may result in higher thermal time (that is, growing degree day) requirements. In this case the loss of crop duration may not be as great as Fig. 2 suggests, since the analysis for Fig. 2 assumes no temperature trends. However, climate variability makes yield-induced crop duration losses difficult to detect. Further, climate variability, and in particular decadal fluctuations in temperature, make persistent strong linear trends unlikely (ref. 10 and Supplementary Extended Data Fig. 3).
The priorities of public and private sector maize breeding in Africa have traditionally been drought and low nitrogen tolerance, with selected adaptive traits (for example, disease tolerance, stalk strength, grain type) for each target agro-ecological zone. A range of maturity classes are used to match thermal time requirements to environments. Drought escape is commonly targeted by breeding for early maturity, which acts contrary to the requirement for increased thermal time imposed by increases in mean temperature. Further, maize breeding programmes do not lend themselves to selection for higher thermal time requirement because yield is the primary criterion for selection within each maturity class.
Changing the maturity class would perhaps appear to be a simple way of dealing with temperature increases. However, this is challenging, since the new variety will still need to be tailored to context-specific stresses, including: foliar diseases and drought tolerance, which tend to be important for late-maturity varieties; and early vigour and reduced anthesis silking interval, which are more important for early-maturity varieties. Farmer and market preference also plays an important role, for example, white maize kernels in much of eastern and southern Africa.
The three ways to improve the matching of maize varieties to a warmed climate are reducing the BDA time, breeding under elevated temperatures, and climate change mitigation. Options for BDA reduction are highly context dependent: gene bank diversity and available breeding technologies differ across institutions and projects; performance testing for new varieties is subject to country-specific and highly diverse regulatory systems11; adoption rates of improved seed varieties vary significantly across locations and seed systems12,13, and market mechanisms, actors and levels of efficiency are also unequal12.
There are numerous specific opportunities for reducing BDA times (Table 1). High-throughput phenotyping platforms and remote-sensing methods for field phenotyping14 could enhance the utilization of gene bank diversity15. The use of doubled haploid16 and marker assisted selection, and in some cases participatory breeding17, can significantly improve the efficiency of breeding. Improved infrastructure and seed bulking facilities would facilitate more effective and efficient marketing, and there is scope to improve rates of adoption, through enhanced extension services, integrated farmer seed networks, and subsidies on inputs12,18,19.
Whilst appropriate interventions vary, some common themes are present. Improved efficiency, state-of-the-art technologies, and effective marketing all come at a cost and, in many cases, financial and resource capacity is likely to represent a major constraint. The costs associated with the bulking and marketing of new seed varieties acts both as a disincentive for high turnover of new products and limits competition from new seed companies12. However, through coordinated working and partnerships, either in the form of public–private partnerships for technology transfer and development, or the regional sharing of genetic resources and the harmonization of regulations, there is potential for some of these constraints to be overcome. Projects such as the International Maize and Wheat Improvement Centre (CIMMYT) Drought Tolerant Maize for Africa (DTMA) programme, which operates at regional scale through collaboration with National Agricultural Research Stations and private seed sector actors, may represent a model for effective operation20.
Alternative seed system models that involve development and dissemination through informal or farmer-led processes provide further options for adapting to warming. These can address farmer-defined priorities and improve seed access17,21,22. In particular, systems that integrate participatory breeding and/or informal mechanisms of dissemination have been shown to improve the overall efficiency of the BDA process. This is not least because adoption starts earlier in the process (that is, with initial farmer participation)17, farmer preferences are taken into account in seed development, and dissemination is less constrained by formal system inefficiencies19,23. Regulatory structures that allow for flexibility in pursuing alternative pathways of breeding and delivery, a principle that is central to the Food and Agriculture Organization of the United Nations (FAO) Quality Declared Seed scheme for seed testing, for example, may therefore be desirable.
Breeding under elevated temperatures has the potential to reduce the loss of crop duration, independently of BDA times. However, identification of suitable sites where trials can be managed and accessed easily is difficult. CIMMYT has identified heat stress sites in Zimbabwe, Kenya and Ethiopia. Data from these trials is being used to identify donor lines for heat stress that can then be introgressed into pedigree breeding pipelines. Trials can also be conducted in greenhouses which, whilst costly, have the advantage of greater control over temperature. The disadvantage of this technique is that correlations between greenhouse assays and field performance can be poor.
We assessed the potential for climate information to provide target elevated temperatures for breeding. The smallest projected temperature change at the end of the BDA cycle provides a temperature increment for breeding that addresses duration shortening whilst avoiding overcompensation for warming. Such overcompensation would produce an extended duration that may result in crops that mature later than the end of the rainy season. For a specific scenario (see Methods) we calculated the temperature increase required during breeding to match crop thermal time requirements to future temperatures (Fig. 3). For lowland mega-environments a target temperature of +0.5 °C improves the match between crop development rates and temperature. Climate model uncertainty is high; if the two models with the lowest temperature increases were deemed inaccurate, then the target temperature would be +1 °C.
Target temperature increases for breeding maize for Africa at the start of a 2015–2049 (that is, worst case) BDA cycle under RCP 8.5. This increment matches crop thermal time requirements to the temperatures during the time the crop is in use. All five mega-environments and all five climate models are shown. The spread of values comes from the grid cells comprising the mega-environment. Boxes mark median and 25th and 75th quantiles, with whiskers extending to the most extreme data point within 1.5 times the interquartile range. The mega-environments are described in the Methods section. Alt.: altitude.
The third mechanism for avoiding crop duration loss that we investigate is mitigation. For the mean and shortest (that is, best case) BDA times, reducing emission from the current trajectory (RCP 8.5) reduces warming out to 2050, so that crop durations stay within current variability (Fig. 2). Mitigation to RCP 2.6 is notably beneficial. However, for all RCPs, the longest BDA times lead to projected crop durations well outside of current variability. Extending the analysis out to 2100, it is clear that mitigation to RCP2.6 is of significantly more benefit, relative to the other emissions trajectories (Supplementary Extended Data Fig. 4). Here, moving to RCP2.6 is at least as effective as moving from the worst-case to the best-case BDA scenario within a given RCP. In the absence of more precise information on BDA times, it is impossible to know whether or not mitigation alone could avoid duration loss.
Given the uncertainties outlined above, it is likely that a combination of measures to reduce BDA times and mitigate climate change would be needed to ensure that crop durations remain within current interannual variability. To develop specific adaptation plans for breeding, improved recording and use of BDA data is critical. Clear reporting on breeding and delivery time frames, success rates and adoption constraints would enable prioritization of actions that are both appropriate and viable given the capabilities and constraints of specific contexts.
There is also potential for climate information to be targeted at specific breeding efforts, through the identification of target temperatures. Where reliable information on rainfall changes is available these target temperatures could also be used to match crops to the rainy season. Where no such information is available it would be important to assess the risk of drought stress (our analysis suggested no change in drought stress—see Methods).
The crop duration signal detected in this study varies coherently across existing mega-environments in all RCPs and time periods analysed (Supplementary Extended Data Fig. 6), suggesting that the mega-environments are an appropriate tool for targeted climate analyses. Effective use of such analyses would rely on a climate services programme with significant and broad engagement, particularly with breeding programmes and national seed testing bodies.
A further adaptation measure that can be cross-cutting to the measures discussed above is the integration of participatory plant breeding into formalized breeding programmes, such as the barley and wheat programmes of the International Center for Agricultural Research in the Dry Area (ICARDA)2. The evolutionary adaptation of crops, through both natural and farmer-led selection of crop varieties that takes place in open pollinated agricultural systems and the associated dynamic gene bank that exists within farmers’ fields, offer a means to further incremental adaptation that could improve the ability of crops to keep pace with climate change and produce more resilient production systems21,24.
This study has implications beyond Africa, since warming trends across the maize-growing regions of tropics are producing similar trends in accumulated thermal time (Supplementary Extended Data Fig. 7). Whilst the global north shows even greater trends than Africa, interannual variability in these areas causes later emergence of signals. Also, maize photoperiod sensitivity complicates interpretation of the figure in the global north.
More broadly, the shortening of duration in response to temperature is a fundamental process that occurs in other major crops such as rice and wheat4. Hence, the implications of duration loss during BDA cycles need to be assessed for other crop and regions. Finally, it is important to note that duration loss is not the only process that is important under climate change. Heat stress indices need to be better constrained through field experiments to enable detection of climate change signals.
Methods
The description of methods below is divided into six sections: study region and input data; signal-to-noise analysis of crop–climate processes, which led to the choice of focus in the main paper, namely the impact of warming on crop duration; estimation of crop duration loss and yield impacts, which details methods for assessing the impacts of the process identified by the signal-to-noise analysis; definition of breeding, delivery and adoption (BDA) times, covering data gathered as input to the estimation of crop duration loss; changes in growing season precipitation, describing the analysis performed to ensure our results are robust in the face of projected changes in precipitation; and estimation of target temperatures for breeding, an assessment of how breeding programmes could use climate model information to directly inform breeding.
All supplementary figures and tables are contained in the Supplementary Information. A brief description of methods for each of the three main figures is presented at the end of the Supplementary Information.
Study region and input data. Maize breeding programmes across sub-Saharan Africa often involve public and /or private international coordinating partners (such as the International Maize and Wheat Improvement Centre, CIMMYT) and national breeders (for example, National Agricultural Research Stations). Such programmes aim to develop germplasm that is designed for optimal performance within the rainfall and temperature regimes of its target ‘mega-environments’ and exhibits desired traits, such as a range of stress tolerances, and cross-breed this germplasm to develop context-appropriate varieties for marketing and adoption by farmers.
CIMMYT divides the main maize-growing regions into mega-environments depending on their environmental conditions, most importantly temperature and rainfall conditions during the growing season25. In this study, we used CIMMYT’s mega-environments data set for Africa, upscaled to a grid of 1.125° × 1.125° (refs 26,27). We include only grid cells that have a fraction of >0.55 associated with one mega-environment in the study (Supplementary Fig. S1). These include all mega-environments except the highlands, which was not possible to assess due to the coarse resolution of this study. The highest maize producing countries (Supplementary Table S1) largely fall across the central belt, which is characterized by mid- and upper altitudes and relatively wet rainfall regimes.
Input data used in the analyses included the daily climate data used as the basis for computing crop–climate indices; the crop calendar information and soil data used to define cropping seasons; and the yield data used to analyse crop duration impacts on maize yields. Climate data used here are from the Inter-Sectoral Impact Model Intercomparison Project (ISIMIP)28—downloaded from the ISIMIP archive at http://esg.pik-potsdam.de. This data set contains daily bias-corrected minimum and maximum temperature, precipitation and solar radiation for five Global Climate Models of the CMIP5 model ensemble (that is, GFDL-ESM2-M, HadGEM2-ES, IPSL-CM5a-LR, MIROC-ESM-CHEM, NorESM1-M) for the four RCPs (that is, RCP2.6, RCP4.5, RCP6.0 and RCP8.5) at a spatial resolution of 0.5° × 0.5°. Mean daily temperature was calculated as the average of minimum and maximum temperature. Spatially explicit crop calendar data were from the study of ref. 29, whereas soil data were gathered from ref. 30. Crop yield data were gathered from ref. 31, which is a global data set of 1.125° × 1.125° grid spacing constructed on the basis of yield observations at sub-national level, satellite-measured vegetation indices and prescribed growing seasons. The climate, crop calendar and soil data sets were all aggregated to the largest common grid spacing of 1.125° × 1.125° using bilinear interpolation.
Signal-to-noise analysis of crop–climate processes. Crop–climate indices were used to determine the crop–climate process on which the main analysis should be conducted (that is, the impact of warming on crop duration). For a total of nine analysis periods (growing periods), seven crop–climate indices were calculated to assess high temperature stress around anthesis, crop duration loss, drought stress, and lethal temperatures (Supplementary Table S2).
To define the growing periods for the crop–climate indices, we used the crop calendar data set and soil data (described above) together with a simple water balance calculation32. To reflect uncertainty in the definition of growing period, three different start dates were used with three different season lengths (110, 120, 130 days), resulting in nine analysis periods. The first growing period started as soon as the ratio of actual to potential evapotranspiration was greater than 0.35 (from the water balance) and minimum temperature was above 6 °C for five consecutive days between the start and the end of the planting window32 or on the last day of the planting window. The second and third growing periods started 7 and 14 days after the first one, respectively.
To determine the processes through which climate change leads to robust impacts, we performed a signal-to-noise analysis on the seven crop–climate indices for the time period 1951 to 2098. Through uncertainty decomposition we compared the total uncertainty of a crop–climate index (‘noise’) with the change in the crop–climate index (‘signal’) over time. The signal-to-noise analysis was performed as described in ref. 5. We analysed climate model uncertainty from five global climate models (GCMs) in the ISIMIP climate data set (see ‘Study region and input data’, above) and three sources of uncertainty in the crop–climate index calculations: planting date (defined by the start of growing period, see above), baseline crop duration (110, 120 and 130 days) and the choice of threshold when stress is experienced (Supplementary Table S2). A total of 27 estimates of each index were produced. All indices were computed for each GCM and for each of the four representative concentration pathways (RCPs 2.6, 4.5, 6.0 and 8.5), resulting in 135 projections for each RCP and crop–climate index.
The ‘signal’ (s) for a crop–climate index for each projection was defined by fitting three loess fits to the crop–climate index data over time (t) 1951 to 2098. Each of the loess fits was configured differently to quantify uncertainty from the method used to detect the signal. We used the following parameter combinations (α affects the degree of smoothing and degree is the polynomial to be used): α = 0.75 and degree = 1; α = 1 and degree = 1; α = 1 and degree = 2. The residuals from this fit represent the variability (v) for the crop–climate index (equation (1)):

where the subscripts (g) and (c) refer to the GCM and crop–climate index, respectively. The uncertainty in the crop–climate index calculation due to the choice of the GCM is  , and that of the crop–climate index is
, and that of the crop–climate index is  ; where
; where  represents the mean across the crop–climate index calculations for each GCM and
represents the mean across the crop–climate index calculations for each GCM and  represents the mean across the GCMs for each crop–climate index. The variability component of the uncertainty is calculated as a linear trend to σ(vg, c). The ‘noise’ is the total uncertainty, calculated as the sum of the individual uncertainty sources. Changes in crop–climate indices were identified as significant when the signal was larger than twice the noise.
represents the mean across the GCMs for each crop–climate index. The variability component of the uncertainty is calculated as a linear trend to σ(vg, c). The ‘noise’ is the total uncertainty, calculated as the sum of the individual uncertainty sources. Changes in crop–climate indices were identified as significant when the signal was larger than twice the noise.
Supplementary Fig. S2 shows the signal-to-noise analysis for four crop–climate indices corresponding to changes in crop duration, high temperature stress around flowering, drought stress and lethal temperatures (see Supplementary Table S2) grouped per maize mega-environment (see ‘Study region and input data’ and Supplementary Fig. S1). Only the crop duration index showed robust changes under future climates.
The lack of a detected signal for a crop–climate index does not imply that the corresponding stress is not important in determining yield, or that it does not change during the analysis period. For example, high temperature stress around anthesis increases with time (Supplementary Fig. S3), especially for RCP 8.5, which is the current emissions trajectory. A large part of the uncertainty for this index is due to uncertainty in the value of the threshold (Supplementary Fig. S4). If heat stress indices could be better constrained, then detection of a climate change signal becomes possible.
A limitation of the definition of the crop–climate indices is that we examine them in isolation. High temperature stress during anthesis might further increase when coinciding with drought conditions. Depending on water status and vapour pressure deficit (VPD), canopy temperatures, that is, the temperatures experienced by the plant, can differ by about 10 °C relative to air temperatures33,34,35, which is used to calculate the indices. In the dry lowlands and the dry mid-altitudes, drought conditions during the anthesis period occur regularly (Supplementary Fig. S5). It is also in these two mega-environments where heat stress is likely to increase most (Supplementary Fig. S3).
Based on this analysis, we finally calculated the time at which the signal in crop duration is detected (results shown in Fig. 1). This time was computed as the time at which the 20-year median changes in duration fall outside the interquartile range of the baseline period 1995 to 2014. Each data point is the median of 20 years × 3 growing periods × 3 planting dates × 3 sets of cardinal temperatures × 5 GCMs.
Estimation of crop duration loss and yield impacts. To calculate crop duration loss, we first computed total season accumulated thermal time (ATT) using the capped-top function (thermal time accumulation increases linearly from Tb to Topt and stays at Topt for values >Topt) with three combinations of base and optimum temperature, that is, Tb = 7.0 and Topt = 30.0 °C, Tb = 8.0 and Topt = 32.5 °C and Tb = 9.0 and Topt = 35.0 °C (refs 36,37) for each grid cell, analysis period, GCM and RCP. Change in crop duration from the baseline period (1995–2014) was then computed on the basis of ATT. First we calculate the average ATT for the baseline period 1995 to 2014 (ATT_B), separately for each grid cell and three different baseline crop durations 110, 120 and 130 days. The duration loss (DL) is then the difference between the number of days taken to reach ATT_B between the projected and baseline period.
We then estimated the number of days of crop duration lost per year by fitting a linear trend to 20-year moving medians from 1995 to 2050. The resulting trends and correlation coefficients are presented in Supplementary Extended Data Fig. 8. Best, worst and mean cases for BDA times were then used to compute integrated changes in crop duration for the entire BDA period. Resulting reductions in crop duration per BDA cycle are shown in Fig. 2.
To understand possible yield impacts of projected increases in growing degree days and associated reductions in crop duration, three analyses were conducted. Two of these used observed yields from ref. 31 (described in ‘Study region and input data’, above), whereas the third analysis was based on a data set derived from the DSSAT38 model simulations of ref. 39. The latter data set is based on site-specific process-based yield simulations for 140 different cultivars present in the DSSAT maize cultivar database38 in a variety of environments ranging from −5 to −45° in latitude and from 0 to 2,500 m in altitude. These three analyses and their results are described in Supplementary Text S2.
Definition of breeding, delivery and adoption (BDA) times. We define BDA as the time it takes to breed, deliver and adopt new crop varieties (Supplementary Tables S3 and S4). The length of BDA for new maize varieties is context-specific and dependent on access to appropriate germplasm; phenotyping and genomic selection technologies; suitability, frequency and reliability of conditions for introgression and backcrossing (including the number of growing seasons per year); national level requirements for seed testing and approval; the efficiency and capacity of public and private seed systems in making new seed available and accessible; and factors affecting rates of adoption among farmers, such as the effectiveness of extension service provision (Table 1). We characterize a best-case (that is, shortest) and worst-case (that is, longest) scenario for the length of BDA based on estimates of time taken for five main stages—selection, breeding, national testing, seed marketing, and adoption—derived from the literature (Supplementary Table S3).
The best- (that is, shortest), worst- (that is, longest), and mean-case scenarios for BDA times were defined as follows. Results from the Drought Tolerant Maize for Africa (DTMA) project were used to define, for as many countries as available, the length of national seed testing and variety release schemes as well as the time it takes for seed companies to replicate seeds in large enough quantities for marketing11,12,40. The time it takes for farmers to adopt new varieties was defined following refs 41,42,43. The time for parent selection was assumed to be four years (worst case) based on experience of CIMMYT breeding programmes9 or zero years (best case) when parents are from advanced breeding populations. The time taken to develop inbred lines and hybrids was assumed to be nine years (worst case) when conventional breeding methods are utilized and several breeding cycles are required to identify lines of good general combining ability44, or six years (best case) where improved breeding technologies (doubled haploids and marker assisted selection) are used and good general combining ability is inherent in developed lines. In all cases the years for selection and breeding are calculated on the assumption that there are two growing seasons per year. It is recognized, however, that a bimodal rainfall pattern is not commonly experienced across the African continent, and that in many regions the viability of a two-season year depends on varietal maturity classes and/or the existence of controlled breeding facilities. The mean case for selection, breeding and adoption represents the midpoint between the best- and worst-case scenarios and for national testing and markets it uses the average of the mean values from each country for which data is available.
A complete description of stage-specific durations and assumptions for BDA is provided in Supplementary Text S1.
Changes in growing season precipitation. A potential concern for our analysis is that the amount of precipitation is crucial for the length of the growing season for rainfed maize systems. The length of the rainy season determines the maize variety that can be grown—that is, a short-duration variety or a higher-yielding longer-duration variety. If seasonal precipitation changes significantly during the twenty-first century, interactions would arise between precipitation-driven changes in growing season length and the temperature-driven crop duration changes that we project. However, the drought-related index does not show a large signal to noise (DS1, Supplementary Fig. S2), suggesting that this is not the case.
To further examine the importance of precipitation, we calculated the trend in total growing season precipitation (PTOT) for the lowest (RCP 2.6) and the highest RCP (RCP 8.5), and the adjusted R2 for the linear trend during the twenty-first century (Supplementary Fig. S6). The change ranges from −16 to +32 mm per decade for RCP 8.5, with a narrower range for RCP 2.6, even though most areas only experience a change of −8 to +8 mm per decade for both RCPs. Thus, changes in precipitation are low compared to background variability, as low R2 and decadal rates of change demonstrate. This indicates that the potential effects of precipitation changes are not as predictable as changes in mean temperatures, and therefore suggests our analysis is unlikely to be biased by not explicitly including precipitation changes when we project crop duration changes.
Estimation of target temperatures for breeding. The analysis is based on a worst-case (that is, longest) BDA cycle: 34 years total BDA time, of which 13 is used for selection and breeding (Supplementary Table S3). A variety is assumed to remain in use for 13 years after initial adoption, which is commonly the case for maize in Africa. The baseline period for the temperature change calculation is the 13 years of breeding (2015–2027; ‘Breeding period’). The future time slice is the 13 years of field cultivation starting at the end of the BDA cycle (2049–2061; ‘Farmer period’). This analysis captures, on average, temperature change between the Breeding period and the Farmer period—that is, the temperature difference that requires adaptation.
We used RCP8.5 with central values of planting date, baseline crop duration and cardinal temperatures (see ‘Estimation of crop duration loss and yield impacts’, above) to determine the daily meteorological time series for analysis. For each grid cell and each year we calculated the accumulated thermal times for the Breeding and Farmer periods. In a warming scenario this quantity is higher in the Farmer period than the Breeding period. We compared accumulated thermal time in the Breeding period to that of the Farmer period to determine the temperature increments to apply during the Breeding period. Where the median value (across grid cells and years and mega-environments) of accumulated thermal time in the Farmer period was greater than the median in the Breeding period this indicates a potential need for adaptation. However, to avoid overcompensating for warming (and thus overshooting the adaptation target of maintaining crop duration), where the difference between these two periods did not exceed one standard deviation, we assumed that no temperature adjustment was required during breeding.
Where the difference exceeded one standard deviation, daily temperatures were adjusted by the difference in mean growing season temperature across the Farmer and Breeding periods. The analysis was then repeated, and where the test still proved negative the temperatures were further adjusted in increments of 10% of the first adjustment (up or down, as required) until the difference was within one standard deviation. The results of this analysis are given in Fig. 3.
References
Burke, M. B., Lobell, D. B. & Guarino, L. Shifts in African crop climates by 2050, and the implications for crop improvement and genetic resources conservation. Glob. Environ. Change 19, 317–325 (2009).
Ceccarelli, S. et al. Plant breeding and climate changes. J. Agric. Sci. 148, 627–637 (2010).
Bassu, S. et al. How do various maize crop models vary in their responses to climate change factors? Glob. Change Biol. 20, 2301–2320 (2014).
Asseng, S. et al. Rising temperatures reduce global wheat production. Nature Clim. Change 5, 143–147 (2015).
Vermeulen, S. J. et al. Addressing uncertainty in adaptation planning for agriculture. Proc. Natl Acad. Sci. USA 110, 8357–8362 (2013).
Hawkins, E. & Sutton, R. The potential to narrow uncertainty in regional climate predictions. Bull. Am. Meteorol. Soc. 90, 1095–1107 (2009).
Trnka, M. et al. Agroclimatic conditions in Europe under climate change. Glob. Change Biol. 17, 2298–2318 (2011).
Cairns, J. E. et al. Adapting maize production to climate change in sub-Saharan Africa. Food Secur. 5, 345–360 (2013).
Bänziger, M., Setimela, P. S., Hodson, D. & Vivek, B. Breeding for improved abiotic stress tolerance in maize adapted to southern Africa. Agric. Wat. Manage. 80, 212–224 (2006).
Hawkins, E. Our evolving climate: communicating the effects of climate variability. Weather 66, 175–179 (2011).
Setimela, P. S., Badu-Apraku, B. & Mwangi, W. B. Variety Testing and Release Approaches in DTMA Project Countries in Sub-Saharan Africa (CIMMYT, 2009).
Langyintuo, A. S. et al. An Analysis of the Bottlenecks Affecting the Production and Deployment of Maize Seed in Eastern and Southern Africa (CIMMYT, 2008); http://go.nature.com/1XaoFRs
Ceccarelli, S. & Grando, S. Decentralized-participatory plant breeding: an example of demand driven research. Euphytica 155, 349–360 (2007).
Araus, J. L. & Cairns, J. E. Field high-throughput phenotyping: the new crop breeding frontier. Trends Plant Sci. 19, 52–61 (2014).
McCouch, S. et al. Agriculture: feeding the future. Nature 499, 23–24 (2013).
Prigge, V. et al. New insights into the genetics of in vivo induction of maternal haploids, the backbone of doubled haploid technology in maize. Genetics 190, 781–793 (2012).
Ceccarelli, S. Efficiency of plant breeding. Crop Sci. 55, 87–97 (2015).
Bekele, A. & Abebe, Y. Analysis of adoption spell of hybrid maize in the Central Rift Valley, Oromyia National Regional State of Ethiopia: a duration model approach. Sci. Technol. Arts Res. J. 3, 207–213 (2015).
Coomes, O. T. et al. Farmer seed networks make a limited contribution to agriculture? Four common misconceptions. Food Policy 56, 41–50 (2015).
Fisher, M. et al. Drought tolerant maize for farmer adaptation to drought in sub-Saharan Africa: determinants of adoption in eastern and southern Africa. Climatic Change 133, 283–299 (2015).
McGuire, S. & Sperling, L. Making seed systems more resilient to stress. Glob. Environ. Change 23, 644–653 (2013).
Scoones, I. & Thompson, J. The politics of seed in Africa’s green revolution: alternative narratives and competing pathways. IDS Bull. 42, 1–23 (2011).
Louwaars, N. P. & de Boef, W. S. Integrated seed sector development in Africa: a conceptual framework for creating coherence between practices, programs, and policies. J. Crop Improv. 26, 39–59 (2012).
Westengen, O. T. & Brysting, A. K. Crop adaptation to climate change in the semi-arid zone in Tanzania: the role of genetic resources and seed systems. Agric. Food Security 3, 3 (2014).
Setimela, P. et al. Environmental classification of maize-testing sites in the SADC region and its implication for collaborative maize breeding strategies in the subcontinent. Euphytica 145, 123–132 (2005).
Hartkamp, A. D. et al. Maize Production Environments Revisited: A GIS-Based Approach (CIMMYT, 2000); http://repository.cimmyt.org/xmlui/bitstream/handle/10883/1004/70209.pdf?sequence=1
Bellon, M. R. et al. Targeting agricultural research to benefit poor farmers: relating poverty mapping to maize environments in Mexico. Food Policy 30, 476–492 (2005).
Hempel, S., Frieler, K., Warszawski, L., Schewe, J. & Piontek, F. A trend-preserving bias correction—the ISI-MIP approach. Earth Syst. Dynam. Discuss. 4, 49–92 (2013).
Sacks, W. J., Deryng, D., Foley, J. A. & Ramankutty, N. Crop planting dates: an analysis of global patterns. Glob. Ecol. Biogeogr. 19, 607–620 (2010).
Shangguan, W., Dai, Y., Duan, Q., Liu, B. & Yuan, H. A. A global soil data set for earth system modeling. J. Adv. Model. Earth Syst. 6, 249–263 (2014).
Iizumi, T. et al. Historical changes in global yields: major cereal and legume crops from 1982 to 2006. Glob. Ecol. Biogeogr. 23, 346–357 (2014).
Jones, P. G. & Thornton, P. K. Croppers to livestock keepers: livelihood transitions to 2050 in Africa due to climate change. Environ. Sci. Policy 12, 427–437 (2009).
Idso, S. B., Reginato, R. J., Jackson, R. D. & Pinter, P. J. Jr Measuring yield-reducing plant potential depressions in wheat by infrared thermometry. Irrigation Sci. 2, 205–212 (1981).
Ehrler, W. L., Idso, S. B., Jackson, R. D. & Reginato, R. J. Wheat canopy temperature: relation to plant water potential. Agron. J. 70, 251–256 (1978).
Altschuler, M. & Mascarenhas, J. P. Heat shock proteins and the effect of heat shock in plants. Plant Mol. Biol. 1, 103–115 (1982).
Jones, C. A., Kiniry, J. R. & Dyke, P. T. CERES-Maize: A Simulation Model of Maize Growth and Development (Texas A & M Univ. Press, 1986).
Challinor, A. J., Parkes, B. & Ramirez-Villegas, J. Crop yield response to climate change varies with cropping intensity. Glob. Change Biol. 21, 1679–1688 (2015).
Jones, J. W. et al. The DSSAT cropping system model. Eur. J. Agron. 18, 235–265 (2003).
Rosegrant, M. W. et al. Food Security in a World of Natural Resource Scarcity: The Role of Agricultural Technologies (IFPRI, 2014); http://doi.org/bjpv
Tahirou, A., Sanogo, D., Langyintuo, A., Bamire, S. A. & Olanrewaju, A. Assessing the Constraints Affecting Production and Deployment of Maize Seed in DTMA Countries of West Africa (International Institute of Tropical Agriculture, IITA, 2009).
Bekele, A. Analysis of adoption spell of hybrid maize in the Central Rift Valley, Oromyia National Regional State of Ethiopia: a duration model approach. Sci. Technol. Arts Res. J. 3, 207 (2015).
Beyene, A. D. & Kassie, M. Speed of adoption of improved maize varieties in Tanzania: an application of duration analysis. Technol. Forecast. Soc. Change 96, 298–307 (2015).
Morris, M., Dreher, K., Ribaut, J.-M. & Khairallah, M. Money matters (II): costs of maize inbred line conversion schemes at CIMMYT using conventional and marker-assisted selection. Mol. Breeding 11, 235–247 (2003).
MacRobert, J., Setimela, P., Gethi, J. & Regasa, M. W. Maize Hybrid Seed Production Manual (CIMMYT, 2014).
Acknowledgements
This work was supported by the CGIAR Research Program on Climate Change, Agriculture and Food Security (CCAFS). This research was partly funded by the NERC/DFID Future Climate For Africa programme under the AMMA-2050 project, grant number NE/M020126/1. The authors thank E. Hawkins from NCAS—University of Reading for advice on signal-to-noise analysis; J. Cairns, K. Sonder and M. Bänzinger from the International Maize and Wheat Improvement Center (CIMMYT) for providing comments and literature on maize breeding under climate change; A. Jarvis from the International Center for Tropical Agriculture (CIAT) for early discussions and insightful comments on the final draft of the manuscript; and B. Badu-Apraku and D. Fakorede from the International Institute for Tropical Agriculture (IITA) for comments on early results. J.R.-V. thanks C. Grenier from CIAT for literature and discussion on breeding pipelines. We acknowledge funding from the CGIAR Research Program on MAIZE.
Author information
Authors and Affiliations
Contributions
A.J.C. conceived and designed the experiments. A.-K.K. and J.R.-V. performed the experiments. A.-K.K., J.R.-V. and S.W. analysed the data. All authors contributed to writing the manuscript and contributed materials and analysis tools.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4779 kb)
Rights and permissions
About this article
Cite this article
Challinor, A., Koehler, AK., Ramirez-Villegas, J. et al. Current warming will reduce yields unless maize breeding and seed systems adapt immediately. Nature Clim Change 6, 954–958 (2016). https://doi.org/10.1038/nclimate3061
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nclimate3061
This article is cited by
-
Importance of genetic architecture in marker selection decisions for genomic prediction
Theoretical and Applied Genetics (2023)
-
Breeding targets for heat-tolerant rice varieties in Japan in a warming climate
Mitigation and Adaptation Strategies for Global Change (2023)
-
Compound heat and moisture extreme impacts on global crop yields under climate change
Nature Reviews Earth & Environment (2022)
-
Progenitor species hold untapped diversity for potential climate-responsive traits for use in wheat breeding and crop improvement
Heredity (2022)
-
The impact of 1.5 °C and 2.0 °C global warming on global maize production and trade
Scientific Reports (2022)