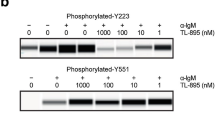
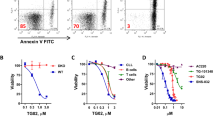
Abstract
Chronic myelogenous leukemia (CML) is a myeloproliferative disorder characterized at the molecular level by the expression of Bcr-abl, a 210-kDa fusion protein with deregulated tyrosine kinase activity. Encouraged by the clinical validation of Bcr-abl as the target for the treatment of CML by imatinib, we sought to identify pharmacological agents that could target this kinase by a distinct mechanism. We report the discovery of a new class of Bcr-abl inhibitors using an unbiased differential cytotoxicity screen of a combinatorial kinase-directed heterocycle library. Compounds in this class (exemplified by GNF-2) show exclusive antiproliferative activity toward Bcr-abl–transformed cells, with potencies similar to imatinib, while showing no inhibition of the kinase activity of full-length or catalytic domain of c-abl. We propose that this new class of compounds inhibits Bcr-abl kinase activity through an allosteric non-ATP competitive mechanism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Schindler, T. et al. Structural mechanism for STI-571 inhibition of Abelson tyrosine kinase. Science 289, 1938–1942 (2000).
Gorre, M.E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293, 876–880 (2001).
von Bubnoff, N., Schneller, F., Peschel, C. & Duyster, J. BCR-ABL gene mutations in relation to clinical resistance of Philadelphia-chromosome-positive leukaemia to STI571: a prospective study. Lancet 359, 487–491 (2002).
Cowan-Jacob, S.W. et al. Imatinib (STI571) resistance in chronic myelogenous leukemia: molecular basis of the underlying mechanisms and potential strategies for treatment. Mini Rev. Med. Chem. 4, 285–299 (2004).
Weisberg, E. et al. Characterization of AMN107, a selective inhibitor of native and mutant Bcr-Abl. Cancer Cell 7, 129–141 (2005).
Shah, N.P. et al. Overriding imatinib resistance with a novel ABL kinase inhibitor. Science 305, 399–401 (2004).
Superti-Furga, G. & Courtneidge, S.A. Structure-function relationships in Src family and related protein tyrosine kinases. Bioessays 17, 321–330 (1995).
Nagar, B. et al. Structural basis for the autoinhibition of c-Abl tyrosine kinase. Cell 112, 859–871 (2003).
Barker, A.J. et al. Studies leading to the identification of ZD1839 (IRESSA): an orally active, selective epidermal growth factor receptor tyrosine kinase inhibitor targeted to the treatment of cancer. Bioorg. Med. Chem. Lett. 11, 1911–1914 (2001).
Meijer, L. et al. Biochemical and cellular effects of roscovitine, a potent and selective inhibitor of the cyclin-dependent kinases cdc2, cdk2 and cdk5. Eur. J. Biochem. 243, 527–536 (1997).
Buchdunger, E. et al. Inhibition of the Abl protein-tyrosine kinase in vitro and in vivo by a 2-phenylaminopyrimidine derivative. Cancer Res. 56, 100–104 (1996).
Regan, J. et al. Pyrazole urea-based inhibitors of p38 MAP kinase: from lead compound to clinical candidate. J. Med. Chem. 45, 2994–3008 (2002).
Lyons, J.F., Wilhelm, S., Hibner, B. & Bollag, G. Discovery of a novel Raf kinase inhibitor. Endocr. Relat. Cancer 8, 219–225 (2001).
Lowinger, T.B., Riedl, B., Dumas, J. & Smith, R.A. Design and discovery of small molecules targeting raf-1 kinase. Curr. Pharm. Des. 8, 2269–2278 (2002).
Ohren, J.F. et al. Structures of human MAP kinase kinase 1 (MEK1) and MEK2 describe novel noncompetitive kinase inhibition. Nat. Struct. Mol. Biol. 11, 1192–1197 (2004).
Barnett, S.F. et al. Identification and characterization of pleckstrin-homology-domain-dependent and isoenzyme-specific Akt inhibitors. Biochem. J. 385, 399–408 (2005).
Grimsby, J. et al. Allosteric activators of glucokinase: potential role in diabetes therapy. Science 301, 370–373 (2003).
Ding, S., Gray, N.S., Wu, X., Ding, Q. & Schultz, P.G. A combinatorial scaffold approach toward kinase-directed heterocycle libraries. J. Am. Chem. Soc. 124, 1594–1596 (2002).
Goldman, J.M. & Melo, J.V. Chronic myeloid leukemia–advances in biology and new approaches to treatment. N. Engl. J. Med. 349, 1451–1464 (2003).
Druker, B.J. et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat. Med. 2, 561–566 (1996).
Carroll, M. et al. CGP 57148, a tyrosine kinase inhibitor, inhibits the growth of cells expressing BCR-ABL, TEL-ABL, and TEL-PDGFR fusion proteins. Blood 90, 4947–4952 (1997).
Dorsey, J.F. et al. Interleukin-3 protects Bcr-Abl-transformed hematopoietic progenitor cells from apoptosis induced by Bcr-Abl tyrosine kinase inhibitors. Leukemia 16, 1589–1595 (2002).
Hantschel, O. et al. A myristoyl/phosphotyrosine switch regulates c-Abl. Cell 112, 845–857 (2003).
Chou, T.C. & Talalay, P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul. 22, 27–55 (1984).
von Bubnoff, N. et al. Inhibition of wild-type and mutant Bcr-Abl by pyrido-pyrimidine-type small molecule kinase inhibitors. Cancer Res. 63, 6395–6404 (2003).
Burke, J.R. et al. BMS-345541 is a highly selective inhibitor of IκB kinase that binds at an allosteric site of the enzyme and blocks NF-κB-dependent transcription in mice. J. Biol. Chem. 278, 1450–1456 (2003).
Nardi, V., Azam, M. & Daley, G.Q. Mechanisms and implications of imatinib resistance mutations in BCR-ABL. Curr. Opin. Hematol. 11, 35–43 (2004).
Huse, M. & Kuriyan, J. The conformational plasticity of protein kinases. Cell 109, 275–282 (2002).
Capdeville, R., Buchdunger, E., Zimmermann, J. & Matter, A. Glivec (STI571, imatinib), a rationally developed, targeted anticancer drug. Nat. Rev. Drug Discov. 1, 493–502 (2002).
Klejman, A., Rushen, L., Morrione, A., Slupianek, A. & Skorski, T. Phosphatidylinositol-3 kinase inhibitors enhance the anti-leukemia effect of STI571. Oncogene 21, 5868–5876 (2002).
Sun, X., Layton, J.E., Elefanty, A. & Lieschke, G.J. Comparison of effects of the tyrosine kinase inhibitors AG957, AG490, and STI571 on BCR-ABL–expressing cells, demonstrating synergy between AG490 and STI571. Blood 97, 2008–2015 (2001).
Topaly, J., Zeller, W.J. & Fruehauf, S. Synergistic activity of the new ABL-specific tyrosine kinase inhibitor STI571 and chemotherapeutic drugs on BCR-ABL-positive chronic myelogenous leukemia cells. Leukemia 15, 342–347 (2001).
Gumireddy, K. et al. A non-ATP-competitive inhibitor of BCR-ABL overrides imatinib resistance. Proc. Natl. Acad. Sci. USA 102, 1992–1997 (2005).
Branford, S. et al. Detection of BCR-ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate-binding loop (P-loop) are associated with a poor prognosis. Blood 102, 276–283 (2003).
Danhauser-Riedl, S., Warmuth, M., Druker, B.J., Emmerich, B. & Hallek, M. Activation of Src kinases p53/56lyn and p59hck by p210bcr/abl in myeloid cells. Cancer Res. 56, 3589–3596 (1996).
Acknowledgements
We thank J.D. Griffin, G. Gilliland, R. Salgia and J. Duyster for kindly providing us with cell lines, and C. Trussell, D. Kemp, M. Warmuth and S. Kim for their help and valuable discussions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Inhibition of cell proliferation. (PDF 19 kb)
Supplementary Table 2
Inhibition of in vitro kinase activity. (PDF 68 kb)
Rights and permissions
About this article
Cite this article
Adrián, F., Ding, Q., Sim, T. et al. Allosteric inhibitors of Bcr-abl–dependent cell proliferation. Nat Chem Biol 2, 95–102 (2006). https://doi.org/10.1038/nchembio760
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio760
This article is cited by
-
The importance of personalized medicine in chronic myeloid leukemia management: a narrative review
Egyptian Journal of Medical Human Genetics (2023)
-
Asciminib as a third line option in chronic myeloid leukemia
International Journal of Hematology (2023)
-
Selective inhibitors of JAK1 targeting an isoform-restricted allosteric cysteine
Nature Chemical Biology (2022)
-
Therapy Resistance and Disease Progression in CML: Mechanistic Links and Therapeutic Strategies
Current Hematologic Malignancy Reports (2022)
-
Druggable exosites of the human kino-pocketome
Journal of Computer-Aided Molecular Design (2020)