Abstract
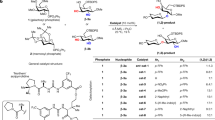
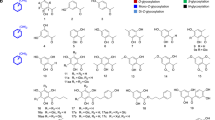
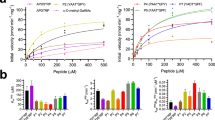
Glycosyltransferases are carbohydrate-active enzymes with essential roles in numerous important biological processes. We have developed a new donor analog for galactosyltransferases that locks a representative target enzyme in a catalytically inactive conformation, thus almost completely abolishing sugar transfer. Results with other galactosyltransferases suggest that this unique mode of glycosyltransferase inhibition may also be generally applicable to other members of this important enzyme family.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Weadge, J.T. & Palcic, M.M. in Wiley Encyclopedia of Chemical Biology Vol. 2 (ed. Begley, T.P.) 198–211 (Wiley, New York, 2009).
Lairson, L.L., Henrissat, B., Davies, G.J. & Withers, S.G. Annu. Rev. Biochem. 77, 521–555 (2008).
Schuman, B., Alfaro, J.A. & Evans, S.V. Top. Curr. Chem. 272, 217–257 (2008).
Breton, C., Snajdrova, L., Jeanneau, C., Koca, J. & Imberty, A. Glycobiology 16, 29R–37R (2006).
Marth, J.D. & Grewal, P.K. Nat. Rev. Immunol. 8, 874–887 (2008).
Rexach, J.E., Clark, P.M. & Hsieh-Wilson, L.C. Nat. Chem. Biol. 4, 97–106 (2008).
Dube, D.H. & Bertozzi, C.R. Nat. Rev. Drug Discov. 4, 477–488 (2005).
Berg, S., Kaur, D., Jackson, M. & Brennan, P.J. Glycobiology 17, 35R–56R (2007).
Qian, X. & Palcic, M.M. in Carbohydrates in Chemistry & Biology (eds. Ernst, B., Hart, G. & Sinaÿ, P.) 293–328 (Wiley-VCH, Weinheim, Germany, 2000).
Qasba, P.K., Ramakrishnan, B. & Boeggeman, E. Trends Biochem. Sci. 30, 53–62 (2005).
Takaya, K. et al. J. Med. Chem. 48, 6054–6065 (2005).
Hennet, T. Cell. Mol. Life Sci. 59, 1081–1095 (2002).
Wagner, G.K., Pesnot, T. & Field, R.A. Nat. Prod. Rep. 26, 1172–1194 (2009).
Gastinel, L.N., Cambillau, C. & Bourne, Y. EMBO J. 18, 3546–3557 (1999).
Alfaro, J.A. et al. J. Biol. Chem. 283, 10097–10108 (2008).
Greco, N.J. & Tor, Y.Z. Nat. Protoc. 2, 305–316 (2007).
Pesnot, T. & Wagner, G.K. Org. Biomol. Chem. 6, 2884–2891 (2008).
Collier, A. & Wagner, G.K. Chem. Commun. (Camb.) 178–180 (2008).
Liu, Y. & Gray, N.S. Nat. Chem. Biol. 2, 358–364 (2006).
Acknowledgements
We thank D.D. Djurhuus, D. Adlercreutz and M.H. Bien for excellent technical assistance during this research. The plasmid for the Neisseria meningitidis α-1,4-GalT was a generous gift from W.W. Wakarchuk (National Research Council of Canada). AA(Gly)B was cloned by H.J. Lee (University of Alberta) and N.O.L. Seto (National Research Council of Canada). We also thank A. Henriksen (Carlsberg Laboratory) for supplying equipment and tools for protein crystallography and for helpful discussions. We are grateful for the help with X-ray data collection provided by the beamline staff at I-911 at MAX-lab (Lund, Sweden) and at X12 at Deutsches Elektronen-Synchrotron (European Molecular Biology Laboratory, Hamburg, Germany). This work was supported by the UK Engineering and Physical Sciences Research Council (First Grant EP/D059186/1 to G.K.W.), the UK Medical Research Council (Discipline Hopping Award G0701861 to G.K.W.), the Leverhulme Trust (Research Fellowship RF/4/RFG/2008/0544 to G.K.W.) and the Danish Agency for Science, Technology and Innovation (grant 272-08-0449 to M.M.P.). We thank the Engineering and Physical Sciences Research Council National Mass Spectrometry Service Centre, Swansea, for the recording of mass spectra.
Author information
Authors and Affiliations
Contributions
T.P. carried out the synthetic work; R.J. carried out the crystallographic analyses; T.P. and R.J. carried out enzymological studies; G.K.W. and M.M.P. designed the study and, together with the other authors, analyzed the data and discussed the results. G.K.W. wrote the paper, with contributions from the other authors. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Schemes 1 and 2, Supplementary Figures 1 and 2 and Supplementary Table 1 (PDF 1945 kb)
Rights and permissions
About this article
Cite this article
Pesnot, T., Jørgensen, R., Palcic, M. et al. Structural and mechanistic basis for a new mode of glycosyltransferase inhibition. Nat Chem Biol 6, 321–323 (2010). https://doi.org/10.1038/nchembio.343
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.343
This article is cited by
-
Plasticity, dynamics, and inhibition of emerging tetracycline resistance enzymes
Nature Chemical Biology (2017)
-
Developing inhibitors of glycan processing enzymes as tools for enabling glycobiology
Nature Chemical Biology (2012)
-
A neutral diphosphate mimic crosslinks the active site of human O-GlcNAc transferase
Nature Chemical Biology (2012)
-
Hijacking a biosynthetic pathway yields a glycosyltransferase inhibitor within cells
Nature Chemical Biology (2011)