Abstract
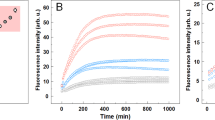
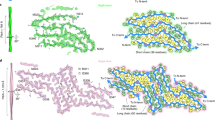
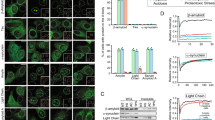
The aberrant assembly of peptides and proteins into fibrillar aggregates proceeds through oligomeric intermediates that are thought to be the primary pathogenic species in many protein deposition diseases. We describe two types of oligomers formed by the HypF-N protein that are morphologically and tinctorially similar, as detected with atomic force microscopy and thioflavin T assays, though one is benign when added to cell cultures whereas the other is toxic. Structural investigation at a residue-specific level using site-directed labeling with pyrene indicated differences in the packing of the hydrophobic interactions between adjacent protein molecules in the oligomers. The lower degree of hydrophobic packing was found to correlate with a higher ability to penetrate the cell membrane and cause an influx of Ca2+ ions. Our findings suggest that structural flexibility and hydrophobic exposure are primary determinants of the ability of oligomeric assemblies to cause cellular dysfunction and its consequences, such as neurodegeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Chiti, F. & Dobson, C.M. Protein misfolding, functional amyloid, and human disease. Annu. Rev. Biochem. 75, 333–366 (2006).
Janson, J., Ashley, R.H., Harrison, D., McIntyre, S. & Butler, P.C. The mechanism of islet amyloid polypeptide toxicity is membrane disruption by intermediate-sized toxic amyloid particles. Diabetes 48, 491–498 (1999).
Sousa, M.M. & Saraiva, M.J. Neurodegeneration in familial amyloid polyneuropathy: from pathology to molecular signaling. Prog. Neurobiol. 71, 385–400 (2003).
Merlini, G. & Bellotti, V. Molecular mechanisms of amyloidosis. N. Engl. J. Med. 349, 583–596 (2003).
Silveira, J.R. et al. The most infectious prion protein particles. Nature 437, 257–261 (2005).
Rahimi, F., Shanmugam, A. & Bitan, G. Structure-function relationships of pre-fibrillar protein assemblies in Alzheimer's disease and related disorders. Curr. Alzheimer Res. 5, 319–341 (2008).
Cookson, M.R. & van der Brug, M. Cell systems and the toxic mechanism(s) of alpha-synuclein. Exp. Neurol. 209, 5–11 (2008).
Kayed, R. et al. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300, 486–489 (2003).
Rosano, C. et al. Crystal structure and anion binding in the prokaryotic hydrogenase maturation factor HypF acylphosphatase-like domain. J. Mol. Biol. 321, 785–796 (2002).
Chiti, F. et al. Solution conditions can promote formation of either amyloid protofilaments or mature fibrils from the HypF N-terminal domain. Protein Sci. 10, 2541–2547 (2001).
Marcon, G. et al. Amyloid formation from HypF-N under conditions in which the protein is initially in its native state. J. Mol. Biol. 347, 323–335 (2005).
Campioni, S. et al. Conformational properties of the aggregation precursor state of HypF-N. J. Mol. Biol. 379, 554–567 (2008).
Bucciantini, M. et al. Inherent toxicity of aggregates implies a common mechanism for protein misfolding diseases. Nature 416, 507–511 (2002).
Bucciantini, M. et al. Prefibrillar amyloid protein aggregates share common features of cytotoxicity. J. Biol. Chem. 279, 31374–31382 (2004).
Baglioni, S. et al. Prefibrillar amyloid aggregates could be generic toxins in higher organisms. J. Neurosci. 26, 8160–8167 (2006).
Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 65, 55–63 (1983).
Cecchi, C. et al. Seladin-1/DHCR24 protects neuroblastoma cells against Abeta toxicity by increasing membrane cholesterol content. J. Cell. Mol. Med. 12, 1990–2002 (2008).
Cecchi, C. et al. Replicating neuroblastoma cells in different cell cycle phases display different vulnerability to amyloid toxicity. J. Mol. Med. 86, 197–209 (2008).
Downs, T.R. & Wilfinger, W.W. Fluorometric quantification of DNA in cells and tissue. Anal. Biochem. 131, 538–547 (1983).
Birks, J.B. Excimers and exciplexes. Nature 214, 1187–1190 (1967).
Hammarström, P. et al. Structural mapping of an aggregation nucleation site in a molten globule intermediate. J. Biol. Chem. 274, 32897–32903 (1999).
Krishnan, R. & Lindquist, S.L. Structural insights into a yeast prion illuminate nucleation and strain diversity. Nature 435, 765–772 (2005).
Dong, D.C. & Winnik, M.A. The Py scale of solvent polarities. Can. J. Chem. 62, 2560–2565 (1984).
Christensen, P.A., Pedersen, J.S., Christiansen, G. & Otzen, D.E. Spectroscopic evidence for the existence of an obligate pre-fibrillar oligomer during glucagon fibrillation. FEBS Lett. 582, 1341–1345 (2008).
Hammarström, P. et al. Structural mapping of an aggregation nucleation site in a molten globule intermediate. J. Biol. Chem. 274, 32897–32903 (1999).
Thirunavukkuarasu, S., Jares-Erijman, E.A. & Jovin, T.M. Multiparametric fluorescence detection of early stages in the amyloid protein aggregation of pyrene-labeled alpha-synuclein. J. Mol. Biol. 378, 1064–1073 (2008).
Semisotnov, G.V. et al. Study of the “molten globule” intermediate state in protein folding by a hydrophobic fluorescent probe. Biopolymers 31, 119–128 (1991).
Cardamone, M. & Puri, N.K. Spectrofluorimetric assessment of the surface hydrophobicity of proteins. Biochem. J. 282, 589–593 (1992).
Kayed, R. et al. Permeabilization of lipid bilayers is a common conformation-dependent activity of soluble amyloid oligomers in protein misfolding diseases. J. Biol. Chem. 279, 46363–46366 (2004).
Demuro, A. et al. Calcium dysregulation and membrane disruption as a ubiquitous neurotoxic mechanism of soluble amyloid oligomers. J. Biol. Chem. 280, 17294–17300 (2005).
Petkova, A.T. et al. Self-propagating, molecular-level polymorphism in Alzheimer's beta-amyloid fibrils. Science 307, 262–265 (2005).
Inaba, S., Okada, T., Konakahara, T. & Kodaka, M. Specific binding of amyloid-beta-protein to IMR-32 neuroblastoma cell membrane. J. Pept. Res. 65, 485–490 (2005).
Lee, S., Fernandez, E.J. & Good, T.A. Role of aggregation conditions in structure, stability, and toxicity of intermediates in the Abeta fibril formation pathway. Protein Sci. 16, 723–732 (2007).
Kayed, R. et al. Annular protofibrils are a structurally and functionally distinct type of amyloid oligomer. J. Biol. Chem. 284, 4230–4237 (2009).
Nekooki-Machida, Y. et al. Distinct conformations of in vitro and in vivo amyloids of huntingtin-exon1 show different cytotoxicity. Proc. Natl. Acad. Sci. USA 106, 9679–9684 (2009).
Yoshiike, Y., Akagi, T. & Takashima, A. Surface structure of amyloid-beta fibrils contributes to cytotoxicity. Biochemistry 46, 9805–9812 (2007).
Hung, L.W. et al. Amyloid-beta peptide (Abeta) neurotoxicity is modulated by the rate of peptide aggregation: Abeta dimers and trimers correlate with neurotoxicity. J. Neurosci. 28, 11950–11958 (2008).
Kheterpal, I. & Wetzel, R. Hydrogen/deuterium exchange mass spectrometry—a window into amyloid structure. Acc. Chem. Res. 39, 584–593 (2006).
Kremer, J.J., Pallitto, M.M., Sklansky, D.J. & Murphy, R.M. Correlation of β-amyloid aggregate size and hydrophobicity with decreased bilayer fluidity of model membranes. Biochemistry 39, 10309–10318 (2000).
Oma, Y., Kino, Y., Sasagawa, N. & Ishiura, S. Intracellular localization of homopolymeric amino acid-containing proteinexpressed in mammalian cells. J. Biol. Chem. 279, 21217–21222 (2004).
Oma, Y., Kino, Y., Sasagawa, N. & Ishiura, S. Comparative analysis of the cytotoxicity of homopolymeric amino acids. Biochim. Biophys. Acta 1748, 174–179 (2005).
Cheon, M. et al. Structural reorganisation and potential toxicity of oligomeric species formed during the assembly of amyloid fibrils. PLoS Comput. Biol. 3, 1727–1738 (2007).
Haugland, R.P. Thiol-reactive probes excited with ultraviolet light. in Handbook of Fluorescent Probes and Research Products 9th edn. (ed. Gregory, J.) 95 (Molecular Probes, Eugene, Oregon, USA, 2002).
Roseman, M.A. Hydrophilicity of polar amino acid side-chains is markedly reduced by flanking peptide bonds. J. Mol. Biol. 200, 513–522 (1988).
Koradi, R., Billeter, M. & Wüthrich, K. MOLMOL: a program for display and analysis of macromolecular structures. J. Mol. Graph. 14, 51–55 (1996).
Acknowledgements
This work was supported by the European Union (Project EURAMY), by the Italian Ministero dell'Istruzione, Università e Ricerca (FIRB RBNE03PX83, PRIN 2006058958 and PRIN 2007XY59Z), Fondazione Cariplo, Ente Cassa di Risparmio di Firenze (project “Lipid rafts” 2008) and the European Molecular Biology Organization Young Investigator Programme. We are grateful to S. Torrassa and D. Nichino for assistance with AFM measurements and to I. Shalova for assistance in the purification and labeling of some of the variants used in this work.
Author information
Authors and Affiliations
Contributions
S.C. and F.C. designed the variants and the experiments to investigate the structure of HypF-N oligomers. S.C. (in part also B.M.) produced, purified and labeled all the variants and performed all the fluorescence experiments (PM, ThT and ANS). A.P. and E.E. performed the MTT assays. M.Z. performed the confocal microscopy experiments. C.P. and A.R. designed, performed and analyzed the AFM data. M.S. and C.C. supervised the experiments on cell cultures. F.C. supervised all the experiments. S.C., C.M.D. and F.C. wrote the manuscript with contributions from A.R., C.C. and M.S.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Results (PDF 3700 kb)
Rights and permissions
About this article
Cite this article
Campioni, S., Mannini, B., Zampagni, M. et al. A causative link between the structure of aberrant protein oligomers and their toxicity. Nat Chem Biol 6, 140–147 (2010). https://doi.org/10.1038/nchembio.283
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.283
This article is cited by
-
Intermolecular interactions underlie protein/peptide phase separation irrespective of sequence and structure at crowded milieu
Nature Communications (2023)
-
Effects of oligomer toxicity, fibril toxicity and fibril spreading in synucleinopathies
Cellular and Molecular Life Sciences (2022)
-
Misfolded protein oligomers induce an increase of intracellular Ca2+ causing an escalation of reactive oxidative species
Cellular and Molecular Life Sciences (2022)
-
Divergent CPEB prion-like domains reveal different assembly mechanisms for a generic amyloid-like fold
BMC Biology (2021)
-
Probing ion channel functional architecture and domain recombination compatibility by massively parallel domain insertion profiling
Nature Communications (2021)