Abstract
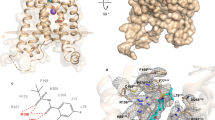
Most G-protein-coupled receptors (GPCRs) are stabilized in common in the inactive state by the formation of the sodium ion–centered water cluster with the conserved Asp2.50 inside the seven-transmembrane domain. We determined the crystal structure of the leukotriene B4 (LTB4) receptor BLT1 bound with BIIL260, a chemical bearing a benzamidine moiety. Surprisingly, the amidine group occupies the sodium ion and water locations, interacts with D662.50, and mimics the entire sodium ion–centered water cluster. Thus, BLT1 is fixed in the inactive state, and the transmembrane helices cannot change their conformations to form the active state. Moreover, the benzamidine molecule alone serves as a negative allosteric modulator for BLT1. As the residues involved in the benzamidine binding are widely conserved among GPCRs, the unprecedented inverse-agonist mechanism by the benzamidine moiety could be adapted to other GPCRs. Consequently, the present structure will enable the rational development of inverse agonists specific for each GPCR.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Canals, M., Sexton, P.M. & Christopoulos, A. Allostery in GPCRs: 'MWC' revisited. Trends Biochem. Sci. 36, 663–672 (2011).
Mahoney, J.P. & Sunahara, R.K. Mechanistic insights into GPCR-G protein interactions. Curr. Opin. Struct. Biol. 41, 247–254 (2016).
Katritch, V. et al. Allosteric sodium in class A GPCR signaling. Trends Biochem. Sci. 39, 233–244 (2014).
Ballesteros, J.A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods in Neurosciences 25, 366–428 (1995).
Gao, Z.G. et al. Identification of essential residues involved in the allosteric modulation of the human A3 adenosine receptor. Mol. Pharmacol. 63, 1021–1031 (2003).
Horstman, D.A. et al. An aspartate conserved among G-protein receptors confers allosteric regulation of α2-adrenergic receptors by sodium. J. Biol. Chem. 265, 21590–21595 (1990).
Liu, W. et al. Structural basis for allosteric regulation of GPCRs by sodium ions. Science 337, 232–236 (2012).
Miller-Gallacher, J.L. et al. The 2.1 Å resolution structure of cyanopindolol-bound β1-adrenoceptor identifies an intramembrane Na+ ion that stabilises the ligand-free receptor. PLoS One 9, e92727 (2014).
Zhang, C. et al. High-resolution crystal structure of human protease-activated receptor 1. Nature 492, 387–392 (2012).
Fenalti, G. et al. Molecular control of δ-opioid receptor signalling. Nature 506, 191–196 (2014).
Rasmussen, S.G. et al. Crystal structure of the β2 adrenergic receptor-Gs protein complex. Nature 477, 549–555 (2011).
Kruse, A.C. et al. Activation and allosteric modulation of a muscarinic acetylcholine receptor. Nature 504, 101–106 (2013).
Huang, W. et al. Structural insights into μ-opioid receptor activation. Nature 524, 315–321 (2015).
Carpenter, B., Nehmé, R., Warne, T., Leslie, A.G. & Tate, C.G. Structure of the adenosine A2A receptor bound to an engineered G protein. Nature 536, 104–107 (2016).
Gutiérrez-de-Terán, H. et al. The role of a sodium ion binding site in the allosteric modulation of the A2A adenosine G protein-coupled receptor. Structure 21, 2175–2185 (2013).
Yokomizo, T., Izumi, T., Chang, K., Takuwa, Y. & Shimizu, T. A G-protein-coupled receptor for leukotriene B4 that mediates chemotaxis. Nature 387, 620–624 (1997).
Birke, F.W., Meade, C.J., Anderskewitz, R., Speck, G.A. & Jennewein, H.M. In vitro and in vivo pharmacological characterization of BIIL 284, a novel and potent leukotriene B4 receptor antagonist. J. Pharmacol. Exp. Ther. 297, 458–466 (2001).
Rosenbaum, D.M. et al. GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function. Science 318, 1266–1273 (2007).
Hori, T. et al. Expression, purification and characterization of leukotriene B4 receptor, BLT1 in Pichia pastoris. Protein Expr. Purif. 72, 66–74 (2010).
Hori, T., Nakamura, M., Yokomizo, T., Shimizu, T. & Miyano, M. The leukotriene B4 receptor BLT1 is stabilized by transmembrane helix capping mutations. Biochem. Biophys. Rep. 4, 243–249 (2015).
Hato, M., Yamashita, J. & Shiono, M. Aqueous phase behavior of lipids with isoprenoid type hydrophobic chains. J. Phys. Chem. B 113, 10196–10209 (2009).
Hato, M., Hosaka, T., Tanabe, H., Kitsunai, T. & Yokoyama, S. A new manual dispensing system for in meso membrane protein crystallization with using a stepping motor-based dispenser. J. Struct. Funct. Genomics 15, 165–171 (2014).
Lehmann, M., Pasamontes, L., Lassen, S.F. & Wyss, M. The consensus concept for thermostability engineering of proteins. Biochim. Biophys. Acta 1543, 408–415 (2000).
Tate, C.G. A crystal clear solution for determining G-protein-coupled receptor structures. Trends Biochem. Sci. 37, 343–352 (2012).
Wu, B. et al. Structures of the CXCR4 chemokine GPCR with small-molecule and cyclic peptide antagonists. Science 330, 1066–1071 (2010).
Tan, Q. et al. Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex. Science 341, 1387–1390 (2013).
Fredriksson, R., Lagerström, M.C., Lundin, L.G. & Schiöth, H.B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharmacol. 63, 1256–1272 (2003).
Zhang, H. et al. Structure of the Angiotensin receptor revealed by serial femtosecond crystallography. Cell 161, 833–844 (2015).
Srivastava, A. et al. High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875. Nature 513, 124–127 (2014).
Hanson, M.A. et al. Crystal structure of a lipid G protein-coupled receptor. Science 335, 851–855 (2012).
Shao, Z. et al. High-resolution crystal structure of the human CB1 cannabinoid receptor. Nature 540, 602–606 (2016).
Palczewski, K. et al. Crystal structure of rhodopsin: A G protein-coupled receptor. Science 289, 739–745 (2000).
Thompson, A.A. et al. Structure of the nociceptin/orphanin FQ receptor in complex with a peptide mimetic. Nature 485, 395–399 (2012).
Thal, D.M. et al. Crystal structures of the M1 and M4 muscarinic acetylcholine receptors. Nature 531, 335–340 (2016).
Kruse, A.C. et al. Structure and dynamics of the M3 muscarinic acetylcholine receptor. Nature 482, 552–556 (2012).
Manglik, A. et al. Structural insights into the dynamic process of β2-adrenergic receptor signaling. Cell 161, 1101–1111 (2015).
Sabirsh, A., Bywater, R.P., Bristulf, J., Owman, C. & Haeggström, J.Z. Residues from transmembrane helices 3 and 5 participate in leukotriene B4 binding to BLT1. Biochemistry 45, 5733–5744 (2006).
Basu, S. et al. Critical role for polar residues in coupling leukotriene B4 binding to signal transduction in BLT1. J. Biol. Chem. 282, 10005–10017 (2007).
Lam, P.Y. et al. Structure-based design of novel guanidine/benzamidine mimics: potent and orally bioavailable factor Xa inhibitors as novel anticoagulants. J. Med. Chem. 46, 4405–4418 (2003).
Rosenbaum, D.M. et al. Structure and function of an irreversible agonist-β2 adrenoceptor complex. Nature 469, 236–240 (2011).
Strange, P.G. Mechanisms of inverse agonism at G-protein-coupled receptors. Trends Pharmacol. Sci. 23, 89–95 (2002).
Khilnani, G. & Khilnani, A.K. Inverse agonism and its therapeutic significance. Indian J. Pharmacol. 43, 492–501 (2011).
Okuno, T. et al. Helix 8 of the leukotriene B4 receptor is required for the conformational change to the low affinity state after G-protein activation. J. Biol. Chem. 278, 41500–41509 (2003).
Kuniyeda, K. et al. Identification of the intracellular region of the leukotriene B4 receptor type 1 that is specifically involved in Gi activation. J. Biol. Chem. 282, 3998–4006 (2007).
Inoue, A. et al. TGFα shedding assay: an accurate and versatile method for detecting GPCR activation. Nat. Methods 9, 1021–1029 (2012).
Warne, T. et al. The structural basis for agonist and partial agonist action on a β1-adrenergic receptor. Nature 469, 241–244 (2011).
Hollenstein, K. et al. Structure of class B GPCR corticotropin-releasing factor receptor 1. Nature 499, 438–443 (2013).
Wu, H. et al. Structure of a class C GPCR metabotropic glutamate receptor 1 bound to an allosteric modulator. Science 344, 58–64 (2014).
Christopher, J.A. et al. Fragment and structure-based drug discovery for a class C GPCR: discovery of the mGlu5 negative allosteric modulator HTL14242 (3-chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J. Med. Chem. 58, 6653–6664 (2015).
Christopoulos, A. & Kenakin, T. G protein-coupled receptor allosterism and complexing. Pharmacol. Rev. 54, 323–374 (2002).
Leach, K., Sexton, P.M. & Christopoulos, A. Allosteric GPCR modulators: taking advantage of permissive receptor pharmacology. Trends Pharmacol. Sci. 28, 382–389 (2007).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Evans, P.R. An introduction to data reduction: space-group determination, scaling and intensity statistics. Acta Crystallogr. D Biol. Crystallogr. 67, 282–292 (2011).
Foadi, J. et al. Clustering procedures for the optimal selection of data sets from multiple crystals in macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 69, 1617–1632 (2013).
Karplus, P.A. & Diederichs, K. Linking crystallographic model and data quality. Science 336, 1030–1033 (2012).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Morris, G.M. et al. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 30, 2785–2791 (2009).
Acknowledgements
We are grateful to Y. Nakamura, H. Tanabe, H. Ago, K. Ida and our colleagues at the RIKEN Center for Life Science Technologies for technical advice and useful discussions on sample preparation, crystallization, data processing, and structural refinement. The synchrotron radiation experiments were performed at BL32XU of SPring-8, with the approval of the Japan Synchrotron Radiation Research Institute (JASRI) (Proposal No. 2016B2726). Some of the vectors for the TGFα-shedding assay were kindly donated by J. Aoki and A. Inoue, at Tohoku University. This work was supported by the Platform Project for Supporting Drug Discovery and Life Science Research funded by Japan Agency for Medical Research and Development (AMED) (S.Y.), by Takeda Science Foundation (T.S.), by MEXT/JSPS KAKENHI Grant Numbers 23770133 (T.H.), 15KK0320, 16K08596 (T.O.), 26460061 (M.N.), 15H05897, 15H05904, and 15H04708 (T.Y.), and by Grants-in-Aid from the Foundation of Strategic Research Projects in Private Universities from MEXT (S1311011, S1411007) (T.Y.) and (2013-2017) (M.M.). M.M. received support for another GPCR project from Takeda Pharmaceutical Co. BIIL260 and BIIL284 were kindly provided by Boehringer Ingelheim Pharma GmbH & Co. KG.
Author information
Authors and Affiliations
Contributions
T.H., T.S., T.Y., M.M., and S.Y. designed the research. T.H. performed all experiments from sample preparation to structure determination and functional analyses, except that T.O. performed the expression of BLT1 mutants using HEK293 cells and the signal assays. K.H., K.Y., Y.K., and M.Y. assisted with the data collection at BL32XU of SPring-8. M.H. constructed the in-house crystallization device and assisted with the crystallization experiment. T.O., M.N., T.S., T.Y., M.M., and S.Y. assisted with the LTB4 binding assay. M.M. supervised the expression, purification, and crystallization in the early stage of the project. S.Y. supervised the purification, crystallization, and structure determination and analysis. T.H. and S.Y. wrote the manuscript, and all of the other authors commented on it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–5, Supplementary Figures 1–14 (PDF 34380 kb)
Rights and permissions
About this article
Cite this article
Hori, T., Okuno, T., Hirata, K. et al. Na+-mimicking ligands stabilize the inactive state of leukotriene B4 receptor BLT1. Nat Chem Biol 14, 262–269 (2018). https://doi.org/10.1038/nchembio.2547
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2547
This article is cited by
-
Bitter taste receptor activation by cholesterol and an intracellular tastant
Nature (2024)
-
G protein-coupled receptors (GPCRs): advances in structures, mechanisms, and drug discovery
Signal Transduction and Targeted Therapy (2024)
-
Evolution and challenges of opioids in pain management: Understanding mechanisms and exploring strategies for safer analgesics
Medicinal Chemistry Research (2024)
-
Structural basis of leukotriene B4 receptor 1 activation
Nature Communications (2022)
-
Structural identification of lysophosphatidylcholines as activating ligands for orphan receptor GPR119
Nature Structural & Molecular Biology (2022)