Abstract
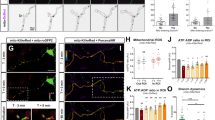
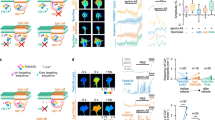
Cyclic AMP (cAMP) and protein kinase A (PKA), classical examples of spatially compartmentalized signaling molecules, are critical axon determinants that regulate neuronal polarity and axon formation, yet little is known about micro-compartmentalization of cAMP and PKA signaling and its role in developing neurons. Here, we revealed that cAMP forms a gradient in developing hippocampal neurons, with higher cAMP levels in more distal regions of the axon compared to other regions of the cell. Interestingly, this cAMP gradient changed according to the developmental stage and depended on proper anchoring of PKA by A-kinase anchoring proteins (AKAPs). Disrupting PKA anchoring to AKAPs increased the cAMP gradient in early-stage neurons and led to enhanced axon elongation. Our results provide new evidence for a local negative-feedback loop, assembled by AKAPs, for the precise control of a growth-stage-dependent cAMP gradient to ensure proper axon growth.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Dotti, C.G., Sullivan, C.A. & Banker, G.A. The establishment of polarity by hippocampal neurons in culture. J. Neurosci. 8, 1454–1468 (1988).
Barnes, A.P. & Polleux, F. Establishment of axon-dendrite polarity in developing neurons. Annu. Rev. Neurosci. 32, 347–381 (2009).
Park, A.J. et al. A presynaptic role for PKA in synaptic tagging and memory. Neurobiol. Learn. Mem. 114, 101–112 (2014).
Pasterkamp, R.J. Getting neural circuits into shape with semaphorins. Nat. Rev. Neurosci. 13, 605–618 (2012).
Cheng, P.L. et al. Self-amplifying autocrine actions of BDNF in axon development. Proc. Natl. Acad. Sci. USA 108, 18430–18435 (2011).
Murray, A.J. & Shewan, D.A. Epac mediates cyclic AMP-dependent axon growth, guidance and regeneration. Mol. Cell. Neurosci. 38, 578–588 (2008).
Muñoz-Llancao, P. et al. Exchange protein directly activated by cAMP (EPAC) regulates neuronal polarization through Rap1B. J. Neurosci. 35, 11315–11329 (2015).
Murray, A.J., Tucker, S.J. & Shewan, D.A. cAMP-dependent axon guidance is distinctly regulated by Epac and protein kinase A. J. Neurosci. 29, 15434–15444 (2009).
Depry, C., Allen, M.D. & Zhang, J. Visualization of PKA activity in plasma membrane microdomains. Mol. Biosyst. 7, 52–58 (2011).
Gervasi, N. et al. Dynamics of protein kinase A signaling at the membrane, in the cytosol, and in the nucleus of neurons in mouse brain slices. J. Neurosci. 27, 2744–2750 (2007).
Shelly, M. et al. Local and long-range reciprocal regulation of cAMP and cGMP in axon/dendrite formation. Science 327, 547–552 (2010).
Nikolaev, V.O. et al. Beta2-adrenergic receptor redistribution in heart failure changes cAMP compartmentation. Science 327, 1653–1657 (2010).
Castro, L.R. et al. Type 4 phosphodiesterase plays different integrating roles in different cellular domains in pyramidal cortical neurons. J. Neurosci. 30, 6143–6151 (2010).
del Puerto, A. et al. Adenylate cyclase 5 coordinates the action of ADP, P2Y1, P2Y13 and ATP-gated P2X7 receptors on axonal elongation. J. Cell Sci. 125, 176–188 (2012).
Murphy, J.G. et al. AKAP-anchored PKA maintains neuronal L-type calcium channel activity and NFAT transcriptional signaling. Cell Rep. 7, 1577–1588 (2014).
Esseltine, J.L. & Scott, J.D. AKAP signaling complexes: pointing towards the next generation of therapeutic targets? Trends Pharmacol. Sci. 34, 648–655 (2013).
Sanderson, J.L. & Dell'Acqua, M.L. AKAP signaling complexes in regulation of excitatory synaptic plasticity. Neuroscientist 17, 321–336 (2011).
Carnegie, G.K. & Scott, J.D. A-kinase anchoring proteins and neuronal signaling mechanisms. Genes Dev. 17, 1557–1568 (2003).
Carr, D.W., Hausken, Z.E., Fraser, I.D., Stofko-Hahn, R.E. & Scott, J.D. Association of the type II cAMP-dependent protein kinase with a human thyroid RII-anchoring protein. Cloning and characterization of the RII-binding domain. J. Biol. Chem. 267, 13376–13382 (1992).
Ventra, C. et al. The differential response of protein kinase A to cyclic AMP in discrete brain areas correlates with the abundance of regulatory subunit II. J. Neurochem. 66, 1752–1761 (1996).
Taylor, S.S., Zhang, P., Steichen, J.M., Keshwani, M.M. & Kornev, A.P. PKA: lessons learned after twenty years. Biochim. Biophys. Acta 1834, 1271–1278 (2013).
Gorshkov, K. & Zhang, J. Visualization of cyclic nucleotide dynamics in neurons. Front. Cell. Neurosci. 8, 395 (2014).
Lohof, A.M., Quillan, M., Dan, Y. & Poo, M.M. Asymmetric modulation of cytosolic cAMP activity induces growth cone turning. J. Neurosci. 12, 1253–1261 (1992).
Shelly, M., Cancedda, L., Heilshorn, S., Sumbre, G. & Poo, M.M. LKB1/STRAD promotes axon initiation during neuronal polarization. Cell 129, 565–577 (2007).
Song, H.J., Ming, G.L. & Poo, M.M. cAMP-induced switching in turning direction of nerve growth cones. Nature 388, 275–279 (1997).
Goslin, K. & Banker, G. Experimental observations on the development of polarity by hippocampal neurons in culture. J. Cell Biol. 108, 1507–1516 (1989).
DiPilato, L.M. & Zhang, J. The role of membrane microdomains in shaping beta2-adrenergic receptor-mediated cAMP dynamics. Mol. Biosyst. 5, 832–837 (2009).
Dessauer, C.W. Adenylyl cyclase-A-kinase anchoring protein complexes: the next dimension in cAMP signaling. Mol. Pharmacol. 76, 935–941 (2009).
Dao, K.K. et al. Epac1 and cAMP-dependent protein kinase holoenzyme have similar cAMP affinity, but their cAMP domains have distinct structural features and cyclic nucleotide recognition. J. Biol. Chem. 281, 21500–21511 (2006).
Vijayaraghavan, S., Goueli, S.A., Davey, M.P. & Carr, D.W. Protein kinase A-anchoring inhibitor peptides arrest mammalian sperm motility. J. Biol. Chem. 272, 4747–4752 (1997).
Bouchard, J.-F. et al. Protein kinase A activation promotes plasma membrane insertion of DCC from an intracellular pool: a novel mechanism regulating commissural axon extension. J. Neurosci. 24, 3040–3050 (2004).
Barnes, A.P. et al. LKB1 and SAD kinases define a pathway required for the polarization of cortical neurons. Cell 129, 549–563 (2007).
Chen, Y. et al. Adenylyl cyclase 6 is selectively regulated by protein kinase A phosphorylation in a region involved in Galphas stimulation. Proc. Natl. Acad. Sci. USA 94, 14100–14104 (1997).
Iwami, G. et al. Regulation of adenylyl cyclase by protein kinase A. J. Biol. Chem. 270, 12481–12484 (1995).
Mika, D. & Conti, M. PDE4D phosphorylation: a coincidence detector integrating multiple signaling pathways. Cell. Signal. 28, 719–724 (2016).
Nicol, X., Hong, K.P. & Spitzer, N.C. Spatial and temporal second messenger codes for growth cone turning. Proc. Natl. Acad. Sci. USA 108, 13776–13781 (2011).
Neves, S.R. et al. Cell shape and negative links in regulatory motifs together control spatial information flow in signaling networks. Cell 133, 666–680 (2008).
Li, L., Gervasi, N. & Girault, J.-A. Dendritic geometry shapes neuronal cAMP signalling to the nucleus. Nat. Commun. 6, 6319 (2015).
Shelly, M. & Poo, M.-M. Role of LKB1-SAD/MARK pathway in neuronal polarization. Dev. Neurobiol. 71, 508–527 (2011).
Ramamurthy, S., Chang, E., Cao, Y., Zhu, J. & Ronnett, G.V. AMPK activation regulates neuronal structure in developing hippocampal neurons. Neuroscience 259, 13–24 (2014).
Zhou, Z. et al. Photoactivated adenylyl cyclase (PAC) reveals novel mechanisms underlying cAMP-dependent axonal morphogenesis. Sci. Rep. 5, 19679 (2016).
Zhong, H. et al. Subcellular dynamics of type II PKA in neurons. Neuron 62, 363–374 (2009).
Diering, G.H., Gustina, A.S. & Huganir, R.L. PKA-GluA1 coupling via AKAP5 controls AMPA receptor phosphorylation and cell-surface targeting during bidirectional homeostatic plasticity. Neuron 84, 790–805 (2014).
Deming, P.B. et al. Anchoring of protein kinase A by ERM (ezrin-radixin-moesin) proteins is required for proper netrin signaling through DCC (deleted in colorectal cancer). J. Biol. Chem. 290, 5783–5796 (2015).
Dodge-Kafka, K.L. et al. The protein kinase A anchoring protein mAKAP coordinates two integrated cAMP effector pathways. Nature 437, 574–578 (2005).
Terrin, A. et al. PKA and PDE4D3 anchoring to AKAP9 provides distinct regulation of cAMP signals at the centrosome. J. Cell Biol. 198, 607–621 (2012).
Gelman, I.H., Tombler, E. & Vargas, J. Jr. A role for SSeCKS, a major protein kinase C substrate with tumour suppressor activity, in cytoskeletal architecture, formation of migratory processes, and cell migration during embryogenesis. Histochem. J. 32, 13–26 (2000).
Piontek, J. & Brandt, R. Differential and regulated binding of cAMP-dependent protein kinase and protein kinase C isoenzymes to gravin in human model neurons: Evidence that gravin provides a dynamic platform for the localization for kinases during neuronal development. J. Biol. Chem. 278, 38970–38979 (2003).
Vardjan, N., Kreft, M. & Zorec, R. Dynamics of β-adrenergic/cAMP signaling and morphological changes in cultured astrocytes. Glia 62, 566–579 (2014).
Soderling, S.H. et al. Loss of WAVE-1 causes sensorimotor retardation and reduced learning and memory in mice. Proc. Natl. Acad. Sci. USA 100, 1723–1728 (2003).
Kawano, Y. et al. CRMP-2 is involved in kinesin-1-dependent transport of the Sra-1/WAVE1 complex and axon formation. Mol. Cell. Biol. 25, 9920–9935 (2005).
Wang, Y. et al. Muscle A-kinase anchoring protein-α is an injury-specific signaling scaffold required for neurotrophic- and cyclic adenosine monophosphate-mediated survival. EBioMedicine 2, 1880–1887 (2015).
Lipscombe, D. et al. Imaging of cytosolic Ca2+ transients arising from Ca2+ stores and Ca2+ channels in sympathetic neurons. Neuron 1, 355–365 (1988).
Willoughby, D., Wachten, S., Masada, N. & Cooper, D.M. Direct demonstration of discrete Ca2+ microdomains associated with different isoforms of adenylyl cyclase. J. Cell Sci. 123, 107–117 (2010).
Yang, J.H., Polanowska-Grabowska, R.K., Smith, J.S., Shields, C.W., IV & Saucerman, J.J. PKA catalytic subunit compartmentation regulates contractile and hypertrophic responses to β-adrenergic signaling. J. Mol. Cell. Cardiol. 66, 83–93 (2014).
Yonaha, M., Chibazakura, T., Kitajima, S. & Yasukochi, Y. Cell cycle-dependent regulation of RNA polymerase II basal transcription activity. Nucleic Acids Res. 23, 4050–4054 (1995).
Kleman, A.M., Yuan, J.Y., Aja, S., Ronnett, G.V. & Landree, L.E. Physiological glucose is critical for optimized neuronal viability and AMPK responsiveness in vitro. J. Neurosci. Methods 167, 292–301 (2008).
Acknowledgements
We thank R. Hand and A. Kolodkin from Johns Hopkins School of Medicine Department of Neuroscience for consultation on various aspects of this manuscript, reagents, and microscopy training. We thank the Malinow lab and I. Hunton at the University California San Diego for use of their lab space and coordinating dissections. We thank Zhang lab members, especially E. Greenwald, for discussions. This work was supported by the UCSD Neuroscience Microscopy Shared Facility Grant P30 NS047101, NSF GRF 1232825 (to K.G.), DGIST Convergence Science Center grant 11-BD-04 (to G.V.R.), R01NS085176 and the Craig H. Neilsen Foundation to (F.-Q.Z.), and NIH R01 DK073368 (to J.Z.).
Author information
Authors and Affiliations
Contributions
K.G. and S.R. performed the experiments; K.G., S.M., and J.Z. designed the experiments and interpreted the data. K.G., S.M., and J.Z. wrote the manuscript; K.G., S.R., S.M., and J.Z. edited the manuscript. Experiments were performed in the laboratories of G.V.R. and J.Z. F.-Q.Z. and J.Z. came up with the initial concept of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results and Supplementary Figures 1–16. (PDF 3055 kb)
Rights and permissions
About this article
Cite this article
Gorshkov, K., Mehta, S., Ramamurthy, S. et al. AKAP-mediated feedback control of cAMP gradients in developing hippocampal neurons. Nat Chem Biol 13, 425–431 (2017). https://doi.org/10.1038/nchembio.2298
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2298
This article is cited by
-
Phosphodiesterase 2A2 regulates mitochondria clearance through Parkin-dependent mitophagy
Communications Biology (2020)
-
AKAP5 complex facilitates purinergic modulation of vascular L-type Ca2+ channel CaV1.2
Nature Communications (2020)
-
Feedback inhibition of cAMP effector signaling by a chaperone-assisted ubiquitin system
Nature Communications (2019)
-
Spatial regulation of axonal cAMP
Nature Chemical Biology (2017)