Abstract
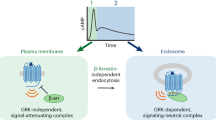
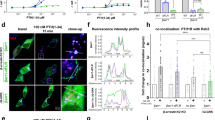
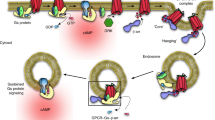
Cells express several G-protein-coupled receptors (GPCRs) at their surfaces, transmitting simultaneous extracellular hormonal and chemical signals into cells. A comprehensive understanding of mechanisms underlying the integrated signaling response induced by distinct GPCRs is thus required. Here we found that the β2-adrenergic receptor, which induces a short cAMP response, prolongs nuclear cAMP and protein kinase A (PKA) activation by promoting endosomal cAMP production in parathyroid hormone (PTH) receptor signaling through the stimulatory action of G protein Gβγ subunits on adenylate cyclase type 2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Feinstein, T.N. et al. Nat. Chem. Biol. 7, 278–284 (2011).
Nagao, M. et al. Proc. Natl. Acad. Sci. USA 108, 17767–17772 (2011).
Wehbi, V.L. et al. Proc. Natl. Acad. Sci. USA 110, 1530–1535 (2013).
Ferrandon, S. et al. Nat. Chem. Biol. 5, 734–742 (2009).
Hanyu, R. et al. Proc. Natl. Acad. Sci. USA 109, 7433–7438 (2012).
Hagiwara, M. et al. Mol. Cell. Biol. 13, 4852–4859 (1993).
Harootunian, A.T. et al. Mol. Biol. Cell 4, 993–1002 (1993).
Isensee, J. et al. J. Cell Sci. 127, 216–229 (2014).
Zhang, P. et al. Science 335, 712–716 (2012).
Manni, S., Mauban, J.H., Ward, C.W. & Bond, M. J. Biol. Chem. 283, 24145–24154 (2008).
Vilardaga, J.P. et al. Nat. Chem. Biol. 4, 126–131 (2008).
Koch, W.J., Hawes, B.E., Inglese, J., Luttrell, L.M. & Lefkowitz, R.J. J. Biol. Chem. 269, 6193–6197 (1994).
Lin, Y. & Smrcka, A.V. Mol. Pharmacol. 80, 551–557 (2011).
Federman, A.D., Conklin, B.R., Schrader, K.A., Reed, R.R. & Bourne, H.R. Nature 356, 159–161 (1992).
Dupré, D.J., Robitaille, M., Rebois, R.V. & Hébert, T.E. Annu. Rev. Pharmacol. Toxicol. 49, 31–56 (2009).
Conley, J.M. et al. J. Pharmacol. Exp. Ther. 347, 276–287 (2013).
Daaka, Y., Luttrell, L.M. & Lefkowitz, R.J. Nature 390, 88–91 (1997).
Baillie, G.S. et al. Proc. Natl. Acad. Sci. USA 100, 940–945 (2003).
Mlakar, V. et al. J. Cell. Mol. Med. 19, 1520–1529 (2015).
Fonseca, T.L. et al. J. Bone Miner. Res. 26, 591–603 (2011).
Cooper, D.M. & Tabbasum, V.G. Biochem. J. 462, 199–213 (2014).
Quitterer, U. & Lohse, M.J. Proc. Natl. Acad. Sci. USA 96, 10626–10631 (1999).
Temkin, P. et al. Nat. Cell Biol. 13, 715–721 (2011).
McGarvey, J.C. et al. J. Biol. Chem. 291, 10986–11002 (2016).
Gidon, A. et al. Nat. Chem. Biol. 10, 707–709 (2014).
Bastepe, M. et al. Mol. Endocrinol. 16, 1912–1919 (2002).
Hynes, T.R. et al. J. Biol. Chem. 279, 30279–30286 (2004).
Zhang, J., Ma, Y., Taylor, S.S. & Tsien, R.Y. Proc. Natl. Acad. Sci. USA 98, 14997–15002 (2001).
Feinstein, T.N. et al. J. Biol. Chem. 288, 27849–27860 (2013).
DiPilato, L.M., Cheng, X. & Zhang, J. Proc. Natl. Acad. Sci. USA 101, 16513–16518 (2004).
Vilardaga, J.P. Curr. Top. Membr. 67, 101–111 (2011).
Gidon, A., Feinstein, T.N., Xiao, K. & Vilardaga, J.P. Methods Cell Biol. 132, 109–126 (2016).
Kahsai, A.W., Rajagopal, S., Sun, J. & Xiao, K. Nat. Protoc. 9, 1301–1319 (2014).
Nobles, K.N. et al. Sci. Signal. 4, ra51 (2011).
Acknowledgements
This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the US National Institutes of Health (NIH) under Award numbers R01 DK087688 and R01 DK102495 (J.-P.V.), and K01AR062598 (J.M.T.), and the Cotswold Foundation Fellowship Award (F.G.J.-A.). The authors thank P. Friedman (University of Pittsburgh) for critical discussion, T. Gardella (Mass general hospital) for providing PTHTMR, and M. Bastepe (Mass general hospital) for providing GαS-knockout MEF cells.
Author information
Authors and Affiliations
Contributions
F.G.J.-A. performed all the experiments with the support of V.L.W. and J.C. K.X., provided expertise and supervised proteomic experiments. M.N. provided expertise with ROS17/2.8 cells. J.M.T. provided expertise with culture of primary osteoblasts, and J.-P.V. was responsible for the overall concept of the study, as well as writing of the manuscript and data analysis with F.G.J.-A.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1 and 2 and Supplementary Figures 1–16. (PDF 863 kb)
Rights and permissions
About this article
Cite this article
Jean-Alphonse, F., Wehbi, V., Chen, J. et al. β2-adrenergic receptor control of endosomal PTH receptor signaling via Gβγ. Nat Chem Biol 13, 259–261 (2017). https://doi.org/10.1038/nchembio.2267
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2267
This article is cited by
-
Precise druggability of the PTH type 1 receptor
Nature Chemical Biology (2022)
-
β-arrestin mediates communication between plasma membrane and intracellular GPCRs to regulate signaling
Communications Biology (2020)
-
Mechanisms of signalling and biased agonism in G protein-coupled receptors
Nature Reviews Molecular Cell Biology (2018)
-
Identification of adenylyl cyclase isoforms mediating parathyroid hormone- and calcitonin-stimulated cyclic AMP accumulation in distal tubule cells
BMC Nephrology (2017)
-
Decoding mixed signals
Nature Chemical Biology (2017)