Abstract
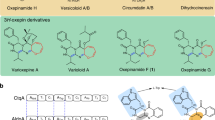
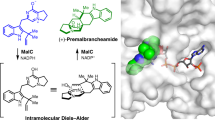
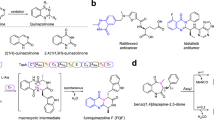
The antitumor fungal metabolite terrequinone A, identified in extracts of Aspergillus sp., is biosynthesized by the five-gene cluster tdiA–tdiE. In this work, we have overproduced all five proteins (TdiA–TdiE) in the bacterial host Escherichia coli, fully reconstituting the biosynthesis of terrequinone A. This pathway involves aminotransferase activity, head-to-tail dimerization and bisprenylation of the scaffold to yield the benzoquinone natural product. We have established that TdiD is a pyridoxal-5′-phosphate–dependent L-tryptophan aminotransferase that generates indolepyruvate for an unusual nonoxidative coupling by the tridomain nonribosomal peptide synthetase TdiA. TdiC, an NADH-dependent quinone reductase, generates the nucleophilic hydroquinone for two distinct rounds of prenylation by the single prenyltransferase TdiB. TdiE is required to shunt the benzoquinone away from an off-pathway monoprenylated species by an as yet unknown mechanism. Overall, we have biochemically characterized the complete biosynthetic pathway to terrequinone A, highlighting the nonoxidative dimerization pathway and the unique asymmetric prenylation involved in its maturation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jerram, W.A. et al. The chemistry of cochliodinol, a metabolite of Chaetomium spp. Can. J. Chem. 53, 727–737 (1975).
Meiler, D. & Taylor, A. The effect of cochliodinol, a metabolite of Chaetomium cochliodes, on the respiration of microsopores of Fusarium oxysporum. Can. J. Microbiol. 17, 83–86 (1971).
Brewer, D., Jerram, W.A. & Taylor, A. The production of cochliodinol and a related metabolite by Chaetomium species. Can. J. Microbiol. 14, 861–866 (1968).
Yamamoto, Y., Kiriyama, S., Shimizu, S. & Koshimura, S. Antitumor activity of asterrequinone, a metabolic product from Aspergillus terreus. Gann 67, 623–624 (1976).
Arai, K. et al. Metabolic products of Aspergillus terreus. IV. Metabolite of the strain IFO 8835. (2). The isolation and structure of indolyl benzoquinone pigments. Chem. Pharm. Bull. (Tokyo) 29, 961–969 (1981).
Mocek, U. et al. Isolation and structure elucidation of five new asterriquinones from Aspergillus, Humicola and Botryotrichum species. J. Antibiot. (Tokyo) 49, 854–859 (1996).
Kaji, A., Saito, R., Nomura, M., Miyamoto, K. & Kiriyama, N. Mechanism of the cytotoxicity of asterriquinone, a metabolite of Aspergillus terreus. Anticancer Res. 17, 3675–3679 (1997).
Shimizu, S., Yamamoto, Y., Inagaki, J. & Koshimura, S. Antitumor effect and structure-activity relationship of asterriquinone analogs. Gann 73, 642–648 (1982).
He, J. et al. Cytotoxic and other metabolites of Aspergillus inhabiting the rhizosphere of Sonoran desert plants. J. Nat. Prod. 67, 1985–1991 (2004).
Bouhired, S., Weber, M., Kempf-Sontag, A., Keller, N.P. & Hoffmeister, D. Accurate prediction of the Aspergillus nidulans terrequinone gene cluster boundaries using the transcriptional regulator LaeA. Fungal Genet. Biol. (2007).
Bok, J.W. et al. Genomic mining for Aspergillus natural products. Chem. Biol. 13, 31–37 (2006).
Marahiel, M.A., Stachelhaus, T. & Mootz, H.D. Modular peptide synthetases involved in nonribosomal peptide synthesis. Chem. Rev. 97, 2651–2674 (1997).
Stein, T. et al. The multiple carrier model of nonribosomal peptide biosynthesis at modular multienzymatic templates. J. Biol. Chem. 271, 15428–15435 (1996).
Belshaw, P.J., Walsh, C.T. & Stachelhaus, T. Aminoacyl-CoAs as probes of condensation domain selectivity in nonribosomal peptide synthesis. Science 284, 486–489 (1999).
Stachelhaus, T., Mootz, H.D., Bergendahl, V. & Marahiel, M.A. Peptide bond formation in nonribosomal peptide biosynthesis. Catalytic role of the condensation domain. J. Biol. Chem. 273, 22773–22781 (1998).
Conti, E., Stachelhaus, T., Marahiel, M.A. & Brick, P. Structural basis for the activation of phenylalanine in the non-ribosomal biosynthesis of gramicidin S. EMBO J. 16, 4174–4183 (1997).
Stachelhaus, T., Huser, A. & Marahiel, M.A. Biochemical characterization of peptidyl carrier protein (PCP), the thiolation domain of multifunctional peptide synthetases. Chem. Biol. 3, 913–921 (1996).
Weber, T., Baumgartner, R., Renner, C., Marahiel, M.A. & Holak, T.A. Solution structure of PCP, a prototype for the peptidyl carrier domains of modular peptide synthetases. Structure 8, 407–418 (2000).
Keating, T.A. & Walsh, C.T. Initiation, elongation, and termination strategies in polyketide and polypeptide antibiotic biosynthesis. Curr. Opin. Chem. Biol. 3, 598–606 (1999).
Williams, R.M., Stocking, E.M. & Sanz-Cervera, J.F. Biosynthesis of prenylated alkaloids derived from tryptophan. Top. Curr. Chem. 209, 97–173 (2000).
Dolence, J.M. & Poulter, C.D. in Comprehensive Natural Product Chemistry (ed. Meth-Cohn, O.) 18473–18500 (Elsevier, Oxford, 1999).
Grundmann, A. & Li, S.M. Overproduction, purification and characterization of FtmPT1, a brevianamide F prenyltransferase from Aspergillus fumigatus. Microbiology 151, 2199–2207 (2005).
Eliot, A.C. & Kirsch, J.F. Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations. Annu. Rev. Biochem. 73, 383–415 (2004).
Magarvey, N.A., Ehling-Schulz, M. & Walsh, C.T. Characterization of the cereulide NRPS alpha-hydroxy acid specifying modules: activation of alpha-keto acids and chiral reduction on the assembly line. J. Am. Chem. Soc. 128, 10698–10699 (2006).
Quadri, L.E. et al. Characterization of Sfp, a Bacillus subtilis phosphopantetheinyl transferase for peptidyl carrier protein domains in peptide synthetases. Biochemistry 37, 1585–1595 (1998).
Nishizawa, T., Aldrich, C.C. & Sherman, D.H. Molecular analysis of the rebeccamycin L-amino acid oxidase from Lechevalieria aerocolonigenes ATCC 39243. J. Bacteriol. 187, 2084–2092 (2005).
Gebler, J.C. & Poulter, C.D. Purification and characterization of dimethylallyl tryptophan synthase from Claviceps purpurea. Arch. Biochem. Biophys. 296, 308–313 (1992).
Lee, S.L., Floss, H.G. & Heinstein, P. Purification and properties of dimethylallylpyrophosphate:tryptopharm dimethylallyl transferase, the first enzyme of ergot alkaloid biosynthesis in Claviceps. sp. SD 58. Arch. Biochem. Biophys. 177, 84–94 (1976).
Unsold, I.A. & Li, S.M. Overproduction, purification and characterization of FgaPT2, a dimethylallyltryptophan synthase from Aspergillus fumigatus. Microbiology 151, 1499–1505 (2005).
Hamahata, A., Takata, Y., Gomi, T. & Fujioka, M. Probing the S-adenosylmethionine-binding site of rat guanidinoacetate methyltransferase. Effect of site-directed mutagenesis of residues that are conserved across mammalian non-nucleic acid methyltransferases. Biochem. J. 317, 141–145 (1996).
de Guzman, F.S. et al. Ochrindoles A-D: new bis-indolyl benzenoids from the sclerotia of Aspergillus ochraceus NRRL 3519. J. Nat. Prod. 57, 634–639 (1994).
Howard-Jones, A.R. & Walsh, C.T. Enzymatic generation of the chromopyrrolic acid scaffold of rebeccamycin by the tandem action of RebO and RebD. Biochemistry 44, 15652–15663 (2005).
Onaka, H., Taniguchi, S., Igarashi, Y. & Furumai, T. Characterization of the biosynthetic gene cluster of rebeccamycin from Lechevalieria aerocolonigenes ATCC 39243. Biosci. Biotechnol. Biochem. 67, 127–138 (2003).
Sanchez, C. et al. The biosynthetic gene cluster for the antitumor rebeccamycin: characterization and generation of indolocarbazole derivatives. Chem. Biol. 9, 519–531 (2002).
Asamizu, S., Kato, Y., Igarashi, Y., Furumai, T. & Onaka, H. Direct formation of chromopyrrolic acid from indole-3-pyruvic acid by StaD, a novel hemoprotein in indolocarbazole biosynthesis. Tetrahedr. Lett. 47, 473–475 (2006).
Howard-Jones, A.R. & Walsh, C.T. Staurosporine and rebeccamycin aglycones are assembled by the oxidative action of StaP, StaC, and RebC on chromopyrrolic acid. J. Am. Chem. Soc. 128, 12289–12298 (2006).
Onaka, H., Taniguchi, S., Igarashi, Y. & Furumai, T. Cloning of the staurosporine biosynthetic gene cluster from Streptomyces sp. TP-A0274 and its heterologous expression in Streptomyces lividans. J. Antibiot. (Tokyo) 55, 1063–1071 (2002).
Balibar, C.J. & Walsh, C.T. In vitro biosynthesis of violacein from L-tryptophan by the enzymes VioA-E from Chromobacterium violaceum. Biochemistry 45, 15444–15457 (2006).
Hibino, S. & Choshi, T. Simple indole alkaloids and those with a nonrearranged monoterpenoid unit. Nat. Prod. Rep. 19, 148–180 (2002).
Sanchez, C., Mendez, C. & Salas, J.A. Indolocarbazole natural products: occurrence, biosynthesis, and biological activity. Nat. Prod. Rep. 23, 1007–1045 (2006).
Bao, B. et al. Bisindole alkaloids of the topsentin and hamacanthin classes from a marine sponge Spongosorites sp. J. Nat. Prod. 70, 2–8 (2007).
Oh, K.B. et al. Bis(indole) alkaloids as sortase A inhibitors from the sponge Spongosorites sp. Bioorg. Med. Chem. Lett. 15, 4927–4931 (2005).
Endo, T., Tsuda, M., Fromont, J. & Kobayashi, J. Hyrtinadine A, a bis-indole alkaloid from a marine sponge. J. Nat. Prod. 70, 423–424 (2007).
Nishizawa, T., Gruschow, S., Jayamaha, D.H., Nishizawa-Harada, C. & Sherman, D.H. Enzymatic assembly of the bis-indole core of rebeccamycin. J. Am. Chem. Soc. 128, 724–725 (2006).
Unsold, I.A. & Li, S.M. Reverse prenyltransferase in the biosynthesis of fumigaclavine C in Aspergillus fumigatus: gene expression, purification, and characterization of fumigaclavine C synthase FGAPT1. ChemBioChem 7, 158–164 (2006).
Stachelhaus, T., Mootz, H.D. & Marahiel, M.A. The specificity-conferring code of adenylation domains in nonribosomal peptide synthetases. Chem. Biol. 6, 493–505 (1999).
Acknowledgements
We gratefully acknowledge the National Institutes of Health grant GM 20011 (to C.T.W.) and a Department of Defense National Defense Science and Engineering Graduate Fellowship (to C.J.B.). We thank P.D. Straight for discussions and for providing a sample of A. nidulans strain A4, and we thank J.A. Read for his critical reading of this manuscript.
Author information
Authors and Affiliations
Contributions
C.J.B. and A.R.H.-J. contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Methods (PDF 221 kb)
Rights and permissions
About this article
Cite this article
Balibar, C., Howard-Jones, A. & Walsh, C. Terrequinone A biosynthesis through L-tryptophan oxidation, dimerization and bisprenylation. Nat Chem Biol 3, 584–592 (2007). https://doi.org/10.1038/nchembio.2007.20
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2007.20
This article is cited by
-
Metabolic engineering for the biosynthesis of bis-indolylquinone terrequinone A in Escherichia coli from L-tryptophan and prenol
Biotechnology for Biofuels and Bioproducts (2023)
-
Fungal quinones: diversity, producers, and applications of quinones from Aspergillus, Penicillium, Talaromyces, Fusarium, and Arthrinium
Applied Microbiology and Biotechnology (2021)
-
Refining and expanding nonribosomal peptide synthetase function and mechanism
Journal of Industrial Microbiology and Biotechnology (2019)
-
Aromatic amino acid aminotransferases in plants
Phytochemistry Reviews (2018)
-
Metabolic engineering of indole pyruvic acid biosynthesis in Escherichia coli with tdiD
Microbial Cell Factories (2017)