Abstract
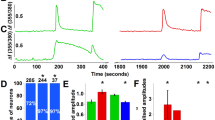
Zinc is an essential biological trace element. It is required for the structure or function of over 300 proteins, and it is increasingly recognized for its role in cell signaling. However, high concentrations of zinc have cytotoxic effects, and overexposure to zinc can cause pain and inflammation through unknown mechanisms. Here we show that zinc excites nociceptive somatosensory neurons and causes nociception in mice through TRPA1, a cation channel previously shown to mediate the pungency of wasabi and cinnamon through cysteine modification. Zinc activates TRPA1 through a unique mechanism that requires zinc influx through TRPA1 channels and subsequent activation via specific intracellular cysteine and histidine residues. TRPA1 is highly sensitive to intracellular zinc, as low nanomolar concentrations activate TRPA1 and modulate its sensitivity. These findings identify TRPA1 as an important target for the sensory effects of zinc and support an emerging role for zinc as a signaling molecule that can modulate sensory transmission.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Vallee, B.L. & Falchuk, K.H. The biochemical basis of zinc physiology. Physiol. Rev. 73, 79–118 (1993).
Frederickson, C.J., Koh, J.Y. & Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 6, 449–462 (2005).
Barceloux, D.G. Zinc. J. Toxicol. Clin. Toxicol. 37, 279–292 (1999).
Blanc, P.D., Boushey, H.A., Wong, H., Wintermeyer, S.F. & Bernstein, M.S. Cytokines in metal fume fever. Am. Rev. Respir. Dis. 147, 134–138 (1993).
Adamson, I.Y., Prieditis, H., Hedgecock, C. & Vincent, R. Zinc is the toxic factor in the lung response to an atmospheric particulate sample. Toxicol. Appl. Pharmacol. 166, 111–119 (2000).
Kuschner, W.G., D'Alessandro, A., Wong, H. & Blanc, P.D. Early pulmonary cytokine responses to zinc oxide fume inhalation. Environ. Res. 75, 7–11 (1997).
Ozaktay, A.C. et al. Effects of interleukin-1 beta, interleukin-6, and tumor necrosis factor on sensitivity of dorsal root ganglion and peripheral receptive fields in rats. Eur. Spine J. 15, 1529–1537 (2006).
Patapoutian, A., Tate, S. & Woolf, C.J. Transient receptor potential channels: targeting pain at the source. Nat. Rev. Drug Discov. 8, 55–68 (2009).
Caterina, M.J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 (1997).
Story, G.M. et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112, 819–829 (2003).
Bandell, M. et al. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 41, 849–857 (2004).
Jordt, S.E. et al. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427, 260–265 (2004).
Macpherson, L.J. et al. The pungency of garlic: activation of TRPA1 and TRPV1 in response to allicin. Curr. Biol. 15, 929–934 (2005).
Obata, K. et al. TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury. J. Clin. Invest. 115, 2393–2401 (2005).
Bautista, D.M. et al. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 124, 1269–1282 (2006).
Kwan, K.Y. et al. TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron 50, 277–289 (2006).
Dhaka, A., Viswanath, V. & Patapoutian, A. TRP ion channels and temperature sensation. Annu. Rev. Neurosci. 29, 135–161 (2006).
Riera, C.E., Vogel, H., Simon, S.A. & le Coutre, J. Artificial sweeteners and salts producing a metallic taste sensation activate TRPV1 receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 293, R626–R634 (2007).
Macpherson, L.J. et al. An ion channel essential for sensing chemical damage. J. Neurosci. 27, 11412–11415 (2007).
McNamara, C.R. et al. TRPA1 mediates formalin-induced pain. Proc. Natl. Acad. Sci. USA 104, 13525–13530 (2007).
Macpherson, L.J. et al. More than cool: promiscuous relationships of menthol and other sensory compounds. Mol. Cell. Neurosci. 32, 335–343 (2006).
Gee, K.R., Zhou, Z.L., Ton-That, D., Sensi, S.L. & Weiss, J.H. Measuring zinc in living cells. A new generation of sensitive and selective fluorescent probes. Cell Calcium 31, 245–251 (2002).
Xu, H., Delling, M., Jun, J.C. & Clapham, D.E. Oregano, thyme and clove-derived flavors and skin sensitizers activate specific TRP channels. Nat. Neurosci. 9, 628–635 (2006).
Karashima, Y. et al. Bimodal action of menthol on the transient receptor potential channel TRPA1. J. Neurosci. 27, 9874–9884 (2007).
Wang, Y.Y., Chang, R.B., Waters, H.N., McKemy, D.D. & Liman, E.R. The nociceptor ion channel TRPA1 is potentiated and inactivated by permeating calcium ions. J. Biol. Chem. 283, 32691–32703 (2008).
Patel, K., Kumar, A. & Durani, S. Analysis of the structural consensus of the zinc coordination centers of metalloprotein structures. Biochim. Biophys. Acta 1774, 1247–1253 (2007).
Macpherson, L.J. et al. Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature 445, 541–545 (2007).
Hinman, A., Chuang, H.H., Bautista, D.M. & Julius, D. TRP channel activation by reversible covalent modification. Proc. Natl. Acad. Sci. USA 103, 19564–19568 (2006).
Barnhart, S. & Rosenstock, L. Cadmium chemical pneumonitis. Chest 86, 789–791 (1984).
Nordberg, G. Excursions of intake above ADI: case study on cadmium. Regul. Toxicol. Pharmacol. 30, S57–S62 (1999).
Yamasaki, S. et al. Zinc is a novel intracellular second messenger. J. Cell Biol. 177, 637–645 (2007).
Mathie, A., Sutton, G.L., Clarke, C.E. & Veale, E.L. Zinc and copper: pharmacological probes and endogenous modulators of neuronal excitability. Pharmacol. Ther. 111, 567–583 (2006).
Hershfinkel, M., Moran, A., Grossman, N. & Sekler, I. A zinc-sensing receptor triggers the release of intracellular Ca2+ and regulates ion transport. Proc. Natl. Acad. Sci. USA 98, 11749–11754 (2001).
Krezel, A., Hao, Q. & Maret, W. The zinc/thiolate redox biochemistry of metallothionein and the control of zinc ion fluctuations in cell signaling. Arch. Biochem. Biophys. 463, 188–200 (2007).
Jo, S.M., Danscher, G., Schroder, H.D. & Suh, S.W. Depletion of vesicular zinc in dorsal horn of spinal cord causes increased neuropathic pain in mice. Biometals 21, 151–158 (2008).
Velazquez, R.A., Cai, Y., Shi, Q. & Larson, A.A. The distribution of zinc selenite and expression of metallothionein-III mRNA in the spinal cord and dorsal root ganglia of the rat suggest a role for zinc in sensory transmission. J. Neurosci. 19, 2288–2300 (1999).
Danscher, G. et al. Inhibitory zinc-enriched terminals in mouse spinal cord. Neuroscience 105, 941–947 (2001).
Kosugi, M., Nakatsuka, T., Fujita, T., Kuroda, Y. & Kumamoto, E. Activation of TRPA1 channel facilitates excitatory synaptic transmission in substantia gelatinosa neurons of the adult rat spinal cord. J. Neurosci. 27, 4443–4451 (2007).
Larson, A.A. & Kitto, K.F. Manipulations of zinc in the spinal cord, by intrathecal injection of zinc chloride, disodium-calcium-EDTA, or dipicolinic acid, alter nociceptive activity in mice. J. Pharmacol. Exp. Ther. 282, 1319–1325 (1997).
Safieh-Garabedian, B. et al. Zinc reduces the hyperalgesia and upregulation of NGF and IL-1 beta produced by peripheral inflammation in the rat. Neuropharmacology 35, 599–603 (1996).
Nelson, M.T. et al. Reducing agents sensitize C-type nociceptors by relieving high-affinity zinc inhibition of T-type calcium channels. J. Neurosci. 27, 8250–8260 (2007).
King, J.C., Shames, D.M. & Woodhouse, L.R. Zinc homeostasis in humans. J. Nutr. 130, 1360S–1366S (2000).
Gee, K.R., Zhou, Z.L., Qian, W.J. & Kennedy, R. Detection and imaging of zinc secretion from pancreatic beta-cells using a new fluorescent zinc indicator. J. Am. Chem. Soc. 124, 776–778 (2002).
Qian, W.J., Aspinwall, C.A., Battiste, M.A. & Kennedy, R.T. Detection of secretion from single pancreatic beta-cells using extracellular fluorogenic reactions and confocal fluorescence microscopy. Anal. Chem. 72, 711–717 (2000).
Razavi, R. et al. TRPV1+ sensory neurons control beta cell stress and islet inflammation in autoimmune diabetes. Cell 127, 1123–1135 (2006).
Bandell, M., Macpherson, L.J. & Patapoutian, A. From chills to chilis: mechanisms for thermosensation and chemesthesis via thermoTRPs. Curr. Opin. Neurobiol. 17, 490–497 (2007).
Wang, G., Strang, C., Pfaffinger, P.J. & Covarrubias, M. Zn2+-dependent redox switch in the intracellular T1–T1 interface of a Kv channel. J. Biol. Chem. 282, 13637–13647 (2007).
Dhaka, A. et al. TRPM8 is required for cold sensation in mice. Neuron 54, 371–378 (2007).
Bandell, M. et al. High-throughput random mutagenesis screen reveals TRPM8 residues specifically required for activation by menthol. Nat. Neurosci. 9, 493–500 (2006).
Petrus, M. et al. A role of TRPA1 in mechanical hyperalgesia is revealed by pharmacological inhibition. Mol. Pain 3, 40 (2007).
Acknowledgements
We thank J. Mathur, T. Earley, J. Watson and M. Garrett for excellent technical support, T. Miyamoto (The Scripps Research Institute) and B. Xiao (The Scripps Research Institute) for supplying mutant TRPA1 constructs, and T. Jegla and members of the Patapoutian lab for helpful discussions. We thank D. Corey (Harvard Medical School) for the Trpa1−/− mice. We also thank the following individuals for generously sharing reagents: M. Caterina (Johns Hopkins University) for rat TRPV1 and N. Prevarskaya (Universite des Sciences et Technologies de Lille) for human TRPM8. This research was supported by the US National Institutes of Health and the Novartis Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1 and 2 (PDF 955 kb)
Rights and permissions
About this article
Cite this article
Hu, H., Bandell, M., Petrus, M. et al. Zinc activates damage-sensing TRPA1 ion channels. Nat Chem Biol 5, 183–190 (2009). https://doi.org/10.1038/nchembio.146
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.146
This article is cited by
-
Zinc is an important inter-kingdom signal between the host and microbe
Veterinary Research (2021)
-
Zinc drives vasorelaxation by acting in sensory nerves, endothelium and smooth muscle
Nature Communications (2021)
-
TRPA1 gene variants hurting our feelings
Pflügers Archiv - European Journal of Physiology (2020)
-
FK506 (tacrolimus) causes pain sensation through the activation of transient receptor potential ankyrin 1 (TRPA1) channels
The Journal of Physiological Sciences (2019)
-
Contribution of Zinc-Dependent Delayed Calcium Influx via TRPC5 in Oxidative Neuronal Death and its Prevention by Novel TRPC Antagonist
Molecular Neurobiology (2019)