Abstract
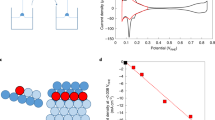
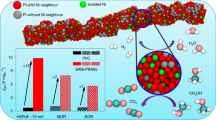
The slow rate of the oxygen reduction reaction in the phosphoric acid fuel cell is the main factor limiting its wide application. Here, we present an approach that can be used for the rational design of cathode catalysts with potential use in phosphoric acid fuel cells, or in any environments containing strongly adsorbing tetrahedral anions. This approach is based on molecular patterning of platinum surfaces with cyanide adsorbates that can efficiently block the sites for adsorption of spectator anions while the oxygen reduction reaction proceeds unhindered. We also demonstrate that, depending on the supporting electrolyte anions and cations, on the same CN-covered Pt(111) surface, the oxygen reduction reaction activities can range from a 25-fold increase to a 50-fold decrease. This behaviour is discussed in the light of the role of covalent and non-covalent interactions in controlling the ensemble of platinum active sites required for high turn over rates of the oxygen reduction reaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
26 August 2010
In the version of this Article originally published, the second author's surname was incorrectly spelt, it should have read María Escudero-Escribano. This has now been corrected on all versions of the Article.
References
Vielstich, W., Lamm, A. & Gasteiger, H. (eds) Handbook of Fuel Cells, Vol. 1, Fundamentals and Survey of Systems (Wiley, 2003).
Schmidt, T. J. & Baurmeister, J. Properties of high-temperature PEFC Celtec®-P 1000 MEAs in start/stop operation mode. J. Power Sources 176, 428–434 (2008).
Seel, D. C., Benicewicz, B. C., Xiao, L. & Schmidt, T. J. in Handbook of Fuel Cells, Fundamentals and Technology, Vol. 5: Advances in Electrocatalysis, Materials and Durability: Part 1 (eds Vielstich, W., Yokokawa, H. & Gasteiger, H. A.) (John Wiley &Sons, 2009).
Markovic, N. M. & Ross, P. N. Surface science studies of model fuel cell electrocatalysts. Surf. Sci. Rep. 45, 117–229 (2002).
Gasteiger, H. A. & Markovic, N. M. Just a dream—or future reality. Science 324, 47–48 (2009).
Stamenkovic, V. R. et al. Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315, 493–497 (2007).
Greeley, J. et al. Alloys of platinum and early transition metals as oxygen reduction electrocatalysts. Nature Chem. 1, 552–556 (2009).
Srivastava, R., Mani, P., Hahn, N. & Strasser, P. Efficient oxygen reduction fuel cell electrocatalysis on voltammetrically dealloyed Pt–Cu–Co nanoparticles. Angew. Chem. Int. Ed. 46, 8988–8991 (2007).
Strasser, P. et al. Lattice-strain control of the activity in dealloyed core–shell fuel cell catalysts. Nature Chem. 2, 454–460 (2010).
Nilekar, A. U. et al. Bimetallic and ternary alloys for improved oxygen reduction catalysis. Top. Catal. 46, 276–284 (2007).
Strmcnik, D. et al. The role of non-covalent interactions in electrocatalytic fuel-cell reactions on platinum. Nature Chem. 1, 466–472 (2009).
Cuesta A. At least three contiguous atoms are necessary for CO formation during methanol electrooxidation on platinum. J. Am. Chem. Soc. 128, 13332–13333 (2006).
Cuesta, A., Escudero, M., Lanova, B. & Baltruschat, H. Cyclic voltammetry, FTIRS and DEMS study of the electrooxidation of carbon monoxide, formic acid and methanol on cyanide-modified Pt(111) electrodes. Langmuir 25, 6500–6507 (2009).
Mostany, J., Herrero, E., Feliu, J. M. & Lipkowski, J. Thermodynamic studies of anion adsorption at stepped platinum (hkl) electrode surfaces in sulfuric acid solutions. J. Phys. Chem. B 106, 12787–12796 (2002).
Mostany, J., Martínez, P., Climent, V., Herrero, E. & Feliu, J. M. Thermodynamic studies of phosphate adsorption on Pt(111) electrode surfaces in perchloric acid solutions. Electrochim. Acta 54, 5836–5843 (2009).
Garcia, N., Climent, V., Orts, J. M., Feliu, J. M. & Aldaz, A. Effect of pH and alkaline metal cations on the voltammetry of Pt(111) single crystal electrodes in sulfuric acid solutions. ChemPhysChem 5, 1221–1227 (2004).
Stickney, J. L., Rosasco, S. D., Salaita, G. N. & Hubbard, A. T. Ordered ionic layers formed on Pt(111) from aqueous solutions. Langmuir 1, 66–71 (1985).
Schardt, B. C. et al. Surface coordination chemistry of well-defined platinum electrodes: surface polyprotic acidity of platinum(111)(2√3×2√3)R30°-hydrogen isocyanide. Inorg. Chem. 24, 1419–1421 (1985).
Frank, D. G. et al. pH and potential dependence of the electrical double layer at well-defined electrode surfaces: Cs+ and Ca2+ ions at Pt(111) (2√3×2√3)R30°–CN, Pt(111) (√13×√13)R14°–CN, and Pt(111) (2×2)–SCN. Langmuir 1, 587–592 (1985).
Hubbard, A. T. Electrochemistry at well-characterized surfaces. Chem. Rev. 88, 633–656 (1988).
Paulissen, V. B. & Korzeniewski, C. Infrared spectroscopy as a probe of the adsorption and electrooxidation of a cyanide monolayer at platinum under aqueous electrochemical conditions. J. Phys. Chem. 96, 4563–4567 (1992).
Kim, C. S. & Korzeniewski, C. Cyanide adsorbed as a monolayer at the low-index surface planes of platinum metal electrodes: an in situ study by infrared spectroscopy. J. Phys. Chem. 97, 9784–9787 (1993).
Stuhlmann, C. Villegas, I. & Weaver, M. J. Scanning tunneling microscopy and infrared spectroscopy as combined in situ probes of electrochemical adlayer structure. Chem. Phys. Lett. 219, 319–324 (1994).
Sawaguchi, T., Yamada, T., Okinaka, Y. & Itaya, K. Electrochemical scanning tunneling microscopy and ultrahigh-vacuum investigation of gold cyanide adlayers on Au(111) formed in aqueous solution. J. Phys. Chem. 99, 14149–14155 (1995).
Stuhlmann, C. Characterization of an electrode adlayer by in situ infrared spectroscopy: cyanide on Pt(111). Surf. Sci. 335, 221–226 (1995).
Friedrich, F. A. et al. In situ spectroscopy of cyanide vibration on Pt(111) and Pt(110) electrode surfaces: potential dependencies and the influence of surface disorder. Surf. Sci. 335, 315–325 (1995).
Kim, Y. G., Yau, S. L. & Itaya, K. Direct observation of complexation of alkali cations on cyanide-modified Pt(111) by scanning tunneling microscopy. J. Am. Chem. Soc. 118, 393–400 (1996).
Inuaki, J., Morioka, Y., Kim, Y. G., Yau, S. L. & Itaya, K. Cation effects on infrared reflection adsorption spectra of cyanide adsorbed on Pt(111) electrode in electrolyte solution. Bull. Chem. Soc. Jpn 70, 1787–1794 (1997).
Huerta, F. J., Morallon, E., Quijada, C., Vazquez, J. L. & Aldaz, A. Spectroelectrochemical study on CN− adsorbed at Pt(111) in sulfuric and perchloric media. Electrochim. Acta 44, 943–948 (1998).
Huerta, F. J., Morallon, E., Vazquez, J. L. & Aldaz, A. Voltammetric and spectrocsopic characterization of cyanide adlayers on Pt(h,k,l) in an acidic medium. Surf. Sci. 396, 400–410 (1998).
Huerta, F. J., Morallon, E. & Vazquez, J. L. Structural effects of adsorbed CN adlayers on the co-adsorption of OH− at the Pt(111) surface in sulfuric acid medium. Surf. Sci. 431, L577–L581 (1999).
Huerta, F., Morallon, E., Quijada, C., Vazquez, J. L. & Berlouis, L. E. A. Potential modulated reflectance spectroscopy of Pt(111) in acidic and alkaline media: cyanide adsorption. J. Electroanal. Chem. 463, 109–115 (1999).
Cuesta, A. & Escudero, M. Electrochemical and FTIRS characterisation of NO adlayers on cyanide-modified Pt(111) electrodes: the mechanism of nitric oxide electroreduction on Pt. Phys. Chem. Chem. Phys. 10, 3628–3634 (2008).
Markovic, N. M., Gasteiger, H. A. & Ross, P. N. Oxygen reduction on platinum low-index single-crystal surfaces in sulfuric acid solution: rotating ring-Pt(hkl) disk studies. J. Phys. Chem. 99, 3411–3415 (1995).
Markovic, N. M., Gasteiger, H. A. & Ross, P. N. Oxygen reduction on platinum low-index single-crystal surfaces in alkaline solution: rotating ring-disk Pt(hkl) studies. J. Phys. Chem. 100, 6715–6721 (1996).
Markovic, N. M., Gasteiger, H. A. & Ross, P. N. Kinetics of oxygen reduction on Pt(hkl) electrodes: implications for the crystallite size effect with supported Pt electrocatalysts. J. Electrochem. Soc. 144, 1591–1597 (1997).
Strmcnik, D. et al. Relationship between the surface coverage of spectator species and the rate of electrocatalytic reactions. J. Phys. Chem. C 111, 18672–18678 (2007).
Schmidt, T. J., Stamenkovic, V. R., Lucas, C., Markovic, N. M. & Ross, P. N. Surface processes and electrocatalysis on the Pt(hkl)/Bi-solution interface. Phys. Chem. Chem. Phys. 3, 3879–3890 (2001).
Clavilier, J., Fernandez-Vega, A., Feliu, J. M. & Aldaz, A. Heterogeneous electrocatalysis on well defined platinum surfaces modified by controlled amounts of irreversibly adsorbed adatoms: Part I. Formic acid oxidation on the Pt (111)-Bi system. J. Electroanal. Chem. 258, 89–100 (1989).
Llorca, M. J., Herrero, J. M., Feliu, J. M. & Aldaz, A. Formic acid oxidation on Pt(111) electrodes modified by irreversibly adsorbed selenium. J. Electroanal. Chem. 373, 217–225 (1994).
Acknowledgements
This work was supported by the Director, Office of Science, Office of Basic Energy Sciences, Division of Materials Sciences, US Department of Energy, under contract no. DE-AC02-06CH11357, and by the DGI (Ministerio de Educación y Ciencia) under project CTQ2006-02109. M.E. acknowledges an FPI fellowship from the DGI and an accommodation grant at the Residencia de Estudiantes from the Madrid City Council.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Strmcnik, D., Escudero-Escribano, M., Kodama, K. et al. Enhanced electrocatalysis of the oxygen reduction reaction based on patterning of platinum surfaces with cyanide. Nature Chem 2, 880–885 (2010). https://doi.org/10.1038/nchem.771
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.771
This article is cited by
-
Parameters Affecting the Fuel Cell Reactions on Platinum Bimetallic Nanostructures
Electrochemical Energy Reviews (2023)
-
Strategies for Sustainable Production of Hydrogen Peroxide via Oxygen Reduction Reaction: From Catalyst Design to Device Setup
Nano-Micro Letters (2023)
-
Sequential reactant water management by complementary multisite catalysts for surpassing platinum hydrogen evolution activity
Nano Research (2023)
-
Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles
Nature Nanotechnology (2021)
-
In situ Raman spectroscopy reveals the structure and dissociation of interfacial water
Nature (2021)