Abstract
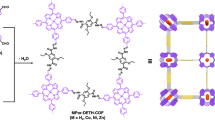
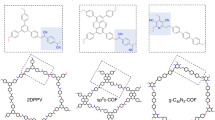
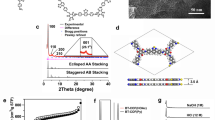
Covalent organic frameworks (COFs) offer a new strategy for assembling organic semiconductors into robust networks with atomic precision and long-range order. General methods for COF synthesis will allow complex building blocks to be incorporated into these emerging materials. Here we report a new Lewis acid-catalysed protocol to form boronate esters directly from protected catechols and arylboronic acids. This transformation also provides crystalline boronate ester-linked COFs from protected polyfunctional catechols and bis(boronic acids). Using this method, we prepared a new COF that features a square lattice composed of phthalocyanine macrocycles joined by phenylene bis(boronic acid) linkers. The phthalocyanines stack in an eclipsed fashion within the COF to form 2.3 nm pores that run parallel to the stacked chromophores. The material's broad absorbance over the solar spectrum, potential for efficient charge transport through the stacked phthalocyanines, good thermal stability and the modular nature of COF synthesis, show strong promise for applications in organic photovoltaic devices.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Anthony, J. E. Functionalized acenes and heteroacenes for organic electronics. Chem. Rev. 106, 5028–5048 (2006).
Park, J.-S. et al. Flexible full color organic light-emitting diode display on polyimide plastic substrate driven by amorphous indium gallium zinc oxide thin-film transistors. Appl. Phys. Lett. 95, 013503 (2009).
Subramanian, V. et al. Progress toward development of all-printed RFID tags: materials, processes, and devices. Proc. IEEE 93, 1330–1338 (2005).
Müllen, K. & Scherf, U. Organic Light Emitting Devices: Synthesis, Properties and Applications (Wiley-VCH, 2006).
Thomas, S. W., Joly, G. D. & Swager, T. M. Chemical sensors based on amplifying fluorescent conjugated polymers. Chem. Rev. 107, 1339–1386 (2007).
Briseno, A. L. et al. Patterning organic single-crystal transistor arrays. Nature 444, 913–917 (2006).
Brabec, C. J. D., Parisi, J. & Sariciftci, N. S. Organic Photovoltaics Concepts and Realization (Springer-Verlag, 2003).
Lloyd, M. T., Anthony, J. E. & Malliaras, G. G. Photovoltaics from soluble small molecules. Mater. Today 10, 34–41 (2007).
Coropceanu, V. et al. Charge transport in organic semiconductors. Chem. Rev. 107, 926–952 (2007).
Anthony, J. E., Brooks, J. S., Eaton, D. L. & Parkin, S. R. Functionalized pentacene: improved electronic properties from control of solid-state order. J. Am. Chem. Soc. 123, 9482–9483 (2001).
Mas-Torrent, M. et al. Correlation between crystal structure and mobility in organic field-effect transistors based on single crystals of tetrathiafulvalene derivatives. J. Am. Chem. Soc. 126, 8546–8553 (2004).
Desiraju, G. R. Cryptic crystallography. Nature Mater. 1, 77–79 (2002).
Côté, A. P. et al. Porous, crystalline, covalent organic frameworks. Science 310, 1166–1170 (2005).
Tilford, R. W., Gemmill, W. R., zur Loye, H. C. & Lavigne, J. J. Facile synthesis of a highly crystalline, covalently linked porous boronate network. Chem. Mater. 18, 5296–5301 (2006).
Kuhn, P., Antonietti, M. & Thomas, A. Porous, covalent triazine-based frameworks prepared by ionothermal synthesis. Angew. Chem. Int. Ed. 47, 3450–3453 (2008).
Furukawa, H. & Yaghi, O. M. Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications. J. Am. Chem. Soc. 131, 8875–8883 (2009).
Wan, S., Guo, J., Kim, J., Ihee, H. & Jiang, D. L. A belt-shaped, blue luminescent, and semiconducting covalent organic framework. Angew. Chem. Int. Ed. 47, 8826–8830 (2008).
Wan, S., Guo, J., Kim, J., Ihee, H. & Jiang, D. L. A photoconductive covalent organic framework: self-condensed arene cubes composed of eclipsed 2D polypyrene sheets for photocurrent generation. Angew. Chem. Int. Ed. 48, 5439–5442 (2009).
Uribe-Romo, F. J. et al. A crystalline imine-linked 3-D porous covalent organic framework. J. Am. Chem. Soc. 131, 4570–4571 (2009).
Hunt, J. R., Doonan, C. J., LeVangie, J. D., Côté, A. P. & Yaghi, O. M. Reticular synthesis of covalent organic borosilicate frameworks. J. Am. Chem. Soc. 130, 11872–11873 (2008).
Tilford, R. W., Mugavero, S. J., Pellechia, P. J. & Lavigne, J. J. Tailoring microporosity in covalent organic frameworks. Adv. Mater. 20, 2741–2748 (2008).
Tong, X., Bailey-Salzman, R. F., Wei, G. & Forrest, S. R. Inverted small molecule organic photovoltaic cells on reflective substrates. Appl. Phys. Lett. 93, 173304 (2008).
Pfuetzner, S., Meiss, J., Petrich, A., Riede, M. & Leo, K. Thick C60:ZnPc bulk heterojunction solar cells with improved performance by film deposition on heated substrates. Appl. Phys. Lett. 94, 253303 (2009).
Reddy, P. Y. et al. Efficient sensitization of nanocrystalline TiO2 films by a near-IR-absorbing unsymmetrical zinc phthalocyanine. Angew. Chem. Int. Ed. 46, 373–376 (2007).
Moreira, L. M. et al. Photodynamic therapy: porphyrins and phthalocyanines as photosensitizers. Aust. J. Chem. 61, 741–754 (2008).
de la Torre, G., Vaquez, P., Agullo-Lopez, F. & Torres, T. Role of structural factors in the nonlinear optical properties of phthalocyanines and related compounds. Chem. Rev. 104, 3723–3750 (2004).
Kadish, K. M., Smith, K. M. & Guilard, R. The Porphyrin Handbook Vol. 15–20 (Academic, 2002).
Seltzman, H. H., Fleming, D. N., Hawkins, G. D. & Carroll, F. I. Facile synthesis and stabilization of 2-arachidonylglycerol via its 1,3-phenylboronate ester. Tetrahedron Lett. 41, 3589–3592 (2000).
Côté, A. P., El-Kaderi, H. M., Furukawa, H., Hunt, J. R. & Yaghi, O. M. Reticular synthesis of microporous and mesoporous 2D covalent organic frameworks. J. Am. Chem. Soc. 129, 12914–12915 (2007).
Baugh, S. D. P., Yang, Z. W., Leung, D. K., Wilson, D. M. & Breslow, R. Cyclodextrin dimers as cleavable carriers of photodynamic sensitizers. J. Am. Chem. Soc. 123, 12488–12494 (2001).
Vanderpol, J. F. et al. A polymer with the mesomorphic order of liquid–crystalline phthalocyanines. Macromolecules 23, 155–162 (1990).
Ruf, M., Lawrence, A. M., Noll, B. C. & Pierpont, C. G. Silicon and zinc coordination to peripheral catechol sites of (2,3,9,10,16,17,23,24-octahydroxyphthalocyaninato)nickel(II). Phthalocyanine coordination chemistry at the edge. Inorg. Chem. 37, 1992–1999 (1998).
Materials Studio Release Notes v.4.4 (Accelrys Software, San Diego, 2008).
Sichel, E. K., Miller, R. E., Abrahams, M. S. & Buiocchi, C. J. Heat capacity and thermal conductivity of hexagonal pyrolytic boron nitride. Phys. Rev. B 13, 4607–4611 (1976).
Liang, J. & Shimizu, G. K. H. Crystalline zinc diphosphonate metal–organic framework with three-dimensional microporosity. Inorg. Chem. Commun. 46, 10449–10451 (2007).
Thallapally, P. K. et al. Carbon dioxide capture in a self-assembled organic nanochannels. Chem. Mater. 19, 3355–3357 (2007).
Mulfort, K. L., Wilson, T. M., Wasielewski, M. R. & Hupp, J. T. Framework reduction and alkali-metal doping of a triply catenating metal–organic framework enhances and then diminishes H2 uptake. Langmuir 25, 503–508 (2009).
Svec, F., Germain, J. & Fréchet, J. M. J. Nanoporous polymers for hydrogen storage. Small 5, 1098–1111 (2009).
Yuan, S. W., Kirklin, S., Dorney, B., Liu, D. J. & Yu, L. P. Nanoporous polymers containing stereocontorted cores for hydrogen storage. Macromolecules 42, 1554–1559 (2009).
Noro, S. et al. Selective gas adsorption in one-dimensional, flexible Cu-II coordination polymers with polar units. Chem. Mater. 21, 3346–3355 (2009).
Barrett, E. P., Joyner, L. G. & Halenda, P. P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 73, 373–380 (2002).
Orilall, M. C. et al. One-pot synthesis of platinum-based nanoparticles incorporated into mesoporous niobium oxide–carbon composites for fuel cell electrodes. J. Am. Chem. Soc. 131, 9389–9395 (2009).
Tant, J. et al. Liquid crystalline metal-free phthalocyanines designed for charge and exciton transport. J. Phys. Chem. B 109, 20315–20323 (2005).
Cornil, J., dos Santos, D. A., Crispin, X., Silbey, R. & Brédas, J. L. Influence of interchain interactions on the absorption and luminescence of conjugated oligomers and polymers: a quantum-chemical characterization. J. Am. Chem. Soc. 120, 1289–1299 (1998).
Huang, X. et al. Self-assembled nanowire networks of aryloxy zinc phthalocyanines based on Zn–O coordination. Langmuir 23, 5167–5172 (2007).
McKeown, N. B., Makhseed, S. & Budd, P. M. Phthalocyanine-based nanoporous network polymers. Chem. Commun. 2780–2781 (2002).
Ivanov, A. V., Svinareva, P. A., Zhukov, I. V. Tomilova, L. G. & Zefirov, N. S. New phthalocyanine complexes based on 4,5-isopropylidenedioxyphthalonitrile. Russ. Chem. Bull. Int. Ed. 52, 1562–1566 (2003).
Acknowledgements
This research was supported by start-up funds provided by Cornell University and the National Science Foundation (NSF)-funded Centers for Chemical Innovation Phase I Center for Molecular Interfacing (CHE-0847926). We also made use of the Cornell Center for Materials Research facilities with support from the NSF Materials Research Science and Engineering Centers program (DMR-0520404). E.L.S. acknowledges the award of the American Competitiveness in Chemistry postdoctoral fellowship from the NSF (CHE-0936988). We thank H. Sai and N. Hoepker for instrument assistance and A. Côté and A. Beeby for discussions.
Author information
Authors and Affiliations
Contributions
Both authors conceived the project, performed and interpreted the results of the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1380 kb)
Rights and permissions
About this article
Cite this article
Spitler, E., Dichtel, W. Lewis acid-catalysed formation of two-dimensional phthalocyanine covalent organic frameworks. Nature Chem 2, 672–677 (2010). https://doi.org/10.1038/nchem.695
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.695
This article is cited by
-
Understanding the electronic pi-system of 2D covalent organic frameworks with Wannier functions
Scientific Reports (2023)
-
Structure determination of a low-crystallinity covalent organic framework by three-dimensional electron diffraction
Communications Chemistry (2023)
-
On-surface synthesis of disilabenzene-bridged covalent organic frameworks
Nature Chemistry (2023)
-
Efficient solution-processed InP quantum-dots light-emitting diodes enabled by suppressing hole injection loss
Nano Research (2023)
-
Two-dimensional covalent organic frameworks for electrocatalysis: Achievements, challenges, and opportunities
Nano Research (2023)