Abstract
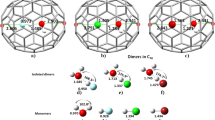
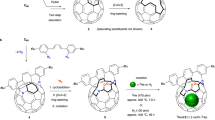
The isolated pentagon rule (IPR) is now widely accepted as a general rule for determining the stability of all-carbon fullerene cages composed of hexagons and pentagons. Fullerenes that violate this rule have been deemed too reactive to be synthesized. The stabilization of non-IPR endohedral fullerenes depends on charge transfer from the encapsulated metal clusters (endoclusters) to fullerene cages, the electronic properties of empty all-carbon cages, the matching size and geometries of fullerene and endocluster, as well as the strong coordination of the metal ions to fused pentagons. The stability of non-IPR exohedral fullerenes can be rationalized primarily by both the 'strain-relief' and 'local-aromaticity' principles. This Review focuses on recent work on stabilization of non-IPR fullerenes, including theoretical and empirical principles, experimental methods, and molecular structures of fused-pentagon fullerenes characterized so far. The special chemical properties of non-IPR fullerenes that distinguish them from IPR-satisfying ones are also emphasized.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kroto, H. W., Heath, J. R., O'Brien, S. C., Curl, R. F. & Smalley, R. E. C60: Buckminsterfullerene. Nature 318, 162–163 (1985).
Jones, D. E. H. Atomization of chemistry. New Scientist 31, 493–496 (1966).
Osawa, E. Superaromaticity. Kagaku (Kyoto, Japan) 25, 854–863 (1970).
Fowler, P. W. & Manolopoulos, D. E. An Atlas of Fullerenes (Oxford Univ. Press, 1995).
Kroto, H. W. The stability of the fullerenes Cn, with n=24, 28, 32, 36, 50, 60 and 70. Nature 329, 529–531 (1986).
Armit, J. W. & Robinson, R. Polynuclear heterocyclic aromatic types. I. Some indenoquinoline derivatives. J. Chem. Soc. Trans. 121, 827–839 (1922).
Bally, T., Chai, S., Neuenschwander, M. & Zhu, Z. Pentalene: formation, electronic, and vibrational structure. J. Am. Chem. Soc. 119, 1869–1875 (1997).
Schmalz, T. G., Seitz, W. A., Klein, D. J. & Hite, G. E. Sixty-carbon-atom carbon cages. Chem. Phys. Lett. 130, 203–207 (1986).
Schmalz, T. G., Seitz, W. A., Klein, D. J. & Hite, G. E. Elemental carbon cages. J. Am. Chem. Soc. 110, 1113–1127 (1988).
Haddon, R. C. Pyramidalization: geometrical interpretation of the π-orbital axis vector in three dimensions. J. Phys. Chem. 91, 3719–3720 (1987).
Haddon, R. C. Chemistry of the fullerenes: the manifestation of strain in a class of continuous aromatic molecules. Science 261, 1545–1550 (1993).
Schein, S. & Friedrich, T. A geometric constraint, the head-to-tail exclusion rule, may be the basis for the isolated-pentagon rule in fullerenes with more than 60 vertices. Proc. Natl Acad. Sci. USA 105, 19142–19147 (2008).
Lu, X. & Chen, Z. F. Curved π-conjugation, aromaticity, and the related chemistry of small fullerenes (< C60) and single-walled carbon nanotubes. Chem. Rev. 105, 3643–3696 (2005).
Heath, J. R. et al. Lanthanum complexes of spheroidal carbon shells. J. Am. Chem. Soc. 107, 7779–7780 (1985).
Shinohara, H. Endohedral metallofullerenes. Rep. Prog. Phys. 63, 843–892 (2000).
Akasaka, T. & Nagase, S. Endofullerenes: A New Family of Carbon Clusters (Kluwer Academic, 2002).
Dunsch, L. & Yang, S. F. Endohedral clusterfullerenes - playing with cluster and cage sizes. Phys. Chem. Chem. Phys. 9, 3067–3081 (2007).
Kadish, K. M. & Ruoff, R. S. Fullerenes: Chemistry, Physics, and Technology (Wiley-Interscience, 2000).
Wang, C. R. et al. Materials sicence: C66 fullerene encaging a scandium dimer. Nature 408, 426–427 (2000).
Stevenson, S. et al. Materials sicence: A stable non-classical metallofullerene family. Nature 408, 427–428 (2000).
Campanera, J. M., Bo, C. & Poblet, J. M. General rule for the stabilization of fullerene cages encapsulating trimetallic nitride templates. Angew. Chem. Int. Ed. 44, 7230–7233 (2005).
Yang, S. F., Kalbac, M., Popov, A. & Dunsch, L. A facile route to the non-IPR fullerene Sc3N@C68: synthesis, spectroscopic characterization, and density functional theory computations (IPR = isolated pentagon rule). Chem. Eur. J. 12, 7856–7863 (2006).
Popov, A. A. & Dunsch, L. Structure, stability, and cluster-cage interactions in nitride clusterfullerenes M3N@C2n (M = Sc, Y; 2n=68–98): a density functional theory study. J. Am. Chem. Soc. 129, 11835–11849 (2007).
Nagase, S., Kobayashi, K. & Akasaka, T. Unconventional cage structures of endohedral metallofullerenes. Theochem-J. Mol. Struct. 461–462, 97–104 (1999).
Shi, Z. Q., Wu, X., Wang, C. R., Lu, X. & Shinohara, H. Isolation and characterization of Sc2C2@C68: a metal-carbide endofullerene with a non-IPR carbon cage. Angew. Chem. Int. Ed. 45, 2107–2111 (2006).
Iiduka, Y. et al. Structural determination of metallofullerene Sc3C82 revisited: a surprising finding. J. Am. Chem. Soc. 127, 12500–12501 (2005).
Stevenson, S. et al. Pyramidalization of Gd3N inside a C80 cage. The synthesis and structure of Gd3N@C80 . Chem. Commun. 2814–2815 (2004).
Olmstead, M. M. et al. Isolation and structural characterization of the endohedral fullerene Sc3N@C78 . Angew. Chem. Int. Ed. 4 0, 1223–1225 (2001).
Popov, A. A., Krause, M., Yang, S., Wong, J. & Dunsch, L. C78 cage isomerism defined by trimetallic nitride cluster size: a computational and vibrational spectroscopic study. J. Phys. Chem. B 111, 3363–3369 (2007).
Akasaka, T. et al. 13C and 139La NMR studies of La2@C80: first evidence for circular motion of metal atoms in endohedral dimetallofullerenes. Angew. Chem. Int. Ed. Engl. 36, 1643–1645 (1997).
Stevenson, S. et al. Small-bandgap endohedral metallofullerenes in high yield and purity. Nature 401, 55–57 (1999).
Yang, S. F., Popov, A. A. & Dunsch, L. Violating the isolated pentagon rule (IPR): the endohedral non-IPR cage of Sc3N@C70 . Angew. Chem. Int. Ed. 46, 1256–1259 (2007).
Yang, S. F., Popov, A. A. & Dunsch, L. The role of an asymmetric nitride cluster on a fullerene cage: The Non-IPR endohedral DySc2N@C76 . J. Phys. Chem. B 111, 13659–13663 (2007).
Summerscales, O. T. & Cloke, F. G. N. The organometallic chemistry of pentalene. Coord. Chem. Rev. 250, 1122–1140 (2006).
Chai, Y. et al. Fullerenes with metals inside. J. Phys. Chem. 95, 7564–7568 (1991).
Kratschmer, W., Lamb, L. D., Fostiropoulos, K. & Huffman, D. R. Solid C60: A new form of carbon. Nature 347, 354–358 (1990).
Yang, S. F. & Dunsch, L. A large family of dysprosium-based trimetallic nitride endohedral fullerenes: Dy3N@C2n (39≤n≤44). J. Phys. Chem. B 109, 12320–12328 (2005).
Bandow, S., Shinohara, H., Saito, Y., Ohkohchi, M. & Ando, Y. High yield synthesis of lanthanofullerenes via lanthanum carbide. J. Phys. Chem. 97, 6101–6103 (1993).
Shinohara, H. et al. Isolation and spectroscopic properties of scandium fullerenes (Sc2@C74, Sc2@C82, and Sc2@C84). J. Phys. Chem. 97, 4259–4261 (1993).
Bolskar, R. D. & Alford, J. M. Chemical oxidation of endohedral metallofullerenes: identification and separation of distinct classes. Chem. Commun. 1292–1293 (2003).
Tsuchiya, T. et al. Reduction of endohedral metallofullerenes: a convenient method for isolation. Chem. Mater. 16, 4343–4346 (2004).
Elliott, B., Yu, L. & Echegoyen, L. A simple isomeric separation of D5h and Ih Sc3N@C80 by selective chemical oxidation. J. Am. Chem. Soc. 127, 10885–10888 (2005).
Ge, Z. X., Duchamp, J. C., Cai, T., Gibson, H. W. & Dorn, H. C. Purification of endohedral trimetallic nitride fullerenes in a single, facile step. J. Am. Chem. Soc. 127, 16292–16298 (2005).
Stevenson, S. et al. Nonchromatographic “stir and filter approach” (SAFA) for isolating Sc3N@C80 metallofullerenes. J. Am. Chem. Soc. 128, 8829–8835 (2006).
Angeli, C. D. et al. Purification of trimetallic nitride templated endohedral metallofullerenes by a chemical reaction of congeners with eutectic 9-methylanthracene. Chem. Mater. 20, 4993–4997 (2008).
Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. App. Cryst. 2, 65–71 (1969).
Collins, D. M. Electron density images from imperfect data by iterative entropy maximization. Nature 298, 49–51 (1982).
Takata, M. et al. Triangle scandium cluster imprisoned in a fullerene cage. Phys. Rev. Lett. 83, 2214–2217 (1999).
Tan, K. & Lu, X. Electronic structure and redox properties of the open-shell metal-carbide Endofullerene Sc3C2@C80: a density functional theory investigation. J. Phys. Chem. A 110, 1171–1176 (2006).
Nishibori, E. et al. High-resolution analysis of (Sc3C2)@C80 metallofullerene by third generation synchrotron radiation X-ray powder diffraction. J. Phys. Chem. B 110, 19215–19219 (2006).
Kobayashi, K. & Nagase, S. A stable unconventional structure of Sc2@C66 found by density functional calculations. Chem. Phys. Lett. 362, 373–379 (2002).
Takata, M., Nishibori, E., Sakata, M., Wang, C. R. & Shinohara, H. Sc2 dimer in IPR-violated C66 fullerene: a covalent bonded metallofullerene. Chem. Phys. Lett. 372, 512–518 (2003).
Olmstead, M. M. et al. Sc3N@C68: folded pentalene coordination in an endohedral fullerene that does not obey the isolated pentagon rule. Angew. Chem. Int. Ed. 42, 900–903 (2003).
Haddon, R. C. π-electrons in three dimensiona. Acc. Chem. Res. 21, 243–249 (1988).
Kobayashi, K., Nagase, S., Yoshida, M. & Sawa, E. Endohedral metallofullerenes. Are the isolated pentagon rule and fullerene structures always satisfied? J. Am. Chem. Soc. 119, 12693–12694 (1997).
Wan, T. S. M. et al. Production, isolation, and electronic properties of missing fullerenes: Ca@C72 and Ca@C74 . J. Am. Chem. Soc. 120, 6806–6807 (1998).
Ichikawa, T. et al. Isolation and characterization of a new isomer of Ca@C72 . Chem. Lett. 33, 1008–1009 (2004).
Wakahara, T. et al. La@C72 having a non-IPR carbon cage. J. Am. Chem. Soc. 128, 14228–14229 (2006).
Kato, H., Taninaka, A., Sugai, T. & Shinohara, H. Structure of a missing-caged metallofullerene: La2@C72 . J. Am. Chem. Soc. 125, 7782–7783 (2003).
Slanina, Z. et al. La2@C72 and Sc2@C72: Computational characterizations. J. Phys. Chem. A 110, 2231–2234 (2006).
Lu, X. et al. Chemical understanding of a non-IPR metallofullerene: stabilization of encaged metals on fused-pentagon bonds in La2@C72 . J. Am. Chem. Soc. 130, 9129–9136 (2008).
Lu, X. et al. Bis-carbene adducts of non-IPR La2@C72: iocalization of high reactivity around fused pentagons and electrochemical properties. Angew. Chem. Int. Ed. 47, 8642–8645 (2008).
Yamada, M. et al. Spectroscopic and theoretical study of endohedral dimetallofullerene having a non-IPR fullerene cage: Ce2@C72 . J. Phys. Chem. A 112, 7627–7631 (2008).
Krause, M., Wong, J. & Dunsch, L. Expanding the world of endohedral fullerenes-the Tm3N@C2n (39≤n≤43) clusterfullerene family. Chem. Eur. J. 11, 706–711 (2005).
Mercado, B. Q. et al. Is the isolated pentagon rule merely a suggestion for endohedral fullerenes? The structure of a second egg-shaped endohedral fullerene-Gd3N@Cs(39663)-C82 . J. Am. Chem. Soc. 130, 7854–7855 (2008).
Beavers, C. M. et al. Tb3N@C84: an improbable, egg-shaped endohedral fullerene that violates the isolated pentagon rule. J. Am. Chem. Soc. 128, 11352–11353 (2006).
Zuo, T. et al. New egg-shaped fullerenes: non-isolated pentagon structures of Tm3N@Cs(51365)C84 and Gd3N@Cs(51365)C84 . Chem. Commun. 1067–1069 (2008).
Hirsch, A. & Brettreich, M. Fullerenes: Chemistry and Reactions (Wiley-VCH, 2005).
Petrie, S. & Bohme, D. K. Enhanced reactivity of fullerene cations containing adjacent pentagons. Nature 365, 426–429 (1993).
Kroto, H. W. & Walton, D. R. M. Stable derivatives of small fullerenes. Chem. Phys. Lett. 214, 353–356 (1993).
Xie, S. Y. et al. Capturing the labile fullerene[50] as C50Cl10 . Science 304, 699–699 (2004).
Han, X. et al. Crystal structures of saturn-like C50Cl10 and pineapple-shaped C64Cl4: geometric implications of double- and triple-pentagon-fused chlorofullerenes. Angew. Chem. Int. Ed. 47, 5340–5343 (2008).
Aihara, J. I. Bond resonance energy and verification of the isolated pentagon rule. J. Am. Chem. Soc. 117, 4130–4136 (1995).
Aihara, J. I., Oe, S., Yoshida, M. & Osawa, E. Further test of the isolated pentagon rule: thermodynamic and kinetic stabilities of C84 fullerene isomers. J. Comput. Chem. 17, 1387–1394 (1996).
Tan, Y. Z. et al. Two Ih-symmetry-breaking C60 isomers stabilized by chlorination. Nature Mater. 7, 790–794 (2008).
Taylor, R. Surprises, serendipity, and symmetry in fullerene chemistry. Synlett 776–793 (2000).
Taylor, R. Why fluorinate fullerenes? J. Fluorine Chem. 125, 359–368 (2004).
Simeonov, K. S., Amsharov, K. Y. & Jansen, M. Connectivity of the chiral D2-symmetric isomer of C76 through a crystal-structure determination of C76Cl18TiCl4 . Angew. Chem. Int. Ed. 46, 8419–8421 (2007).
Simeonov, K. S., Amsharov, K. Y. & Jansen, M. C80Cl12: a chlorine derivative of the chiral D2-C80 isomer-empirical rationale of halogen-atom addition pattern. Chem. Eur. J. 15, 1812–1815 (2009).
Poater, J., Fradera, X., Duran, M. & Sola, M. An insight into the local aromaticities of polycyclic aromatic hydrocarbons and fullerenes. Chem. Eur. J. 9, 1113–1122 (2003).
Hirsch, A., Chen, Z. & Jiao, H. Spherical aromaticity in I h symmetrical fullerenes: the 2(N+1)2 rule. Angew. Chem. Int. Ed. 39, 3915–3917 (2000).
Paquette, L. A. Dodecahedrane-the chemical transliteration of Plato's universe (A Review). Proc. Natl Acad. Sci. USA 79, 4495–4500 (1982).
Troshin, P. A. et al. Isolation of two seven-membered ring C58 fullerene derivatives: C58F17CF3 and C58F18 . Science 309, 278–281 (2005).
Qian, W. Y. et al. C62, a non-classical fullerene incorporating a four-membered ring. J. Am. Chem. Soc. 122, 8333–8334 (2000).
Qian, W. et al. Synthesis of stable derivatives of C62: the first nonclassical fullerene incorporating a four-membered ring. J. Am. Chem. Soc. 125, 2066–2067 (2003).
Prinzbach, H. et al. Gas-phase production and photoelectron spectroscopy of the smallest fullerene, C20 . Nature 407, 60–63 (2000).
Ternansky, R. J., Balogh, D. W. & Paquette, L. A. Dodecahedrane. J. Am. Chem. Soc. 104, 4503–4504 (1982).
Paquette, L. A., Ternansky, R. J. & Balogh, D. W. A strategy for the synthesis of monosubstituted dodecahedrane and the isolation of an isododecahedrane. J. Am. Chem. Soc. 104, 4502–4503 (1982).
Piskoti, C., Yarger, J. & Zettl, A. C36, a new carbon solid. Nature 393, 771–774 (1998).
Koshio, A., Inakuma, M., Sugai, T. & Shinohara, H. A preparative scale synthesis of C36 by high-temperature laser-vaporization: purification and identification of C36H6 and C36H6O. J. Am. Chem. Soc. 122, 398–399 (2000).
Koshio, A., Inakuma, M., Wang, Z. W., Sugai, T. & Shinohara, H. In situ laser-furnace TOF mass spectrometry of C36 and the large-scale production by arc-discharge. J. Phys. Chem. B 104, 7908–7913 (2000).
Loffler, D., Jester, S. S., Weis, P., Bottcher, A. & Kappes, M. M. Cn films (n = 50, 52, 54, 56, and 58) on graphite: cage size dependent electronic properties. J. Chem. Phys. 124, 054705 (2006).
Wahl, F., Woerth, J. & Prinzbach, H. The pagodane route to dodecahedranes: improved access to the C20H20 framework as well as partial and total functionalization. Does C20 fullerene exist? Angew. Chem. Int. Ed. Engl. 32, 1722–1726 (1993).
Prinzbach, H. & Weber, K. From insecticide to Plato's Universe - the pagodane route to dodecahedranes: new routes and new targets. Angew. Chem. Int. Ed. Engl. 33, 2239–2257 (1994).
Wahl, F. et al. Towards perfunctionalized dodecahedranes - en route to C20 fullerene. Chem. Eur. J. 12, 6255–6267 (2006).
Chen, Z. Fullerene derivative: the smaller fullerene C50, isolated as C50Cl10 . Angew. Chem. Int. Ed. 43, 4690–4691 (2004).
Tan, Y. Z. et al. An entrant of smaller fullerene: C56 captured by chlorines and aigned in linear chains. J. Am. Chem. Soc. 130, 15240–15241 (2008).
Chen, D. L., Tian, W. Q., Feng, J. K. & Sun, C. C. Structures and electronic properties of C56Cl8 and C56Cl10 fullerene compounds. ChemPhysChem 8, 2386–2390 (2007).
Stone, A. J. & Wales, D. J. Theoretical studies of icosahedral footballene sixty-carbon-atom molecules and some related species. Chem. Phys. Lett. 128, 501–503 (1986).
Wang, C. R. et al. C64H4: Production, isolation, and structural characterizations of a stable unconventional fulleride. J. Am. Chem. Soc. 128, 6605–6610 (2006).
Yan, Q. B., Zheng, Q. R. & Su, G. Structures, electronic properties, spectroscopies, and hexagonal monolayer phase of a family of unconventional fullerenes C64X4 (X = H, F, Cl, Br). J. Phys. Chem. C 111, 549–554 (2007).
Campanera, J. M., Bo, C. & Poblet, J. M. Exohedral reactivity of trimetallic nitride template (TNT) endohedral metallofullerenes. J. Org. Chem. 71, 46–54 (2006).
Cai, T. et al. Synthesis and characterization of a non-IPR fullerene derivative: Sc3N@C68[C(COOC2H5)(2)]. J. Phys. Chem. C 112, 19203–19208 (2008).
Acknowledgements
The authors thank Michael Blackburn from the University of Sheffield, UK, and Xin Lu from Xiamen University, China, for help in preparing the manuscript. This work was supported by the NNSFC (No. 20525103, 20531050, 20721001) and the 973 Program (No. 2007CB815301).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, YZ., Xie, SY., Huang, RB. et al. The stabilization of fused-pentagon fullerene molecules. Nature Chem 1, 450–460 (2009). https://doi.org/10.1038/nchem.329
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.329
This article is cited by
-
Predicting the new carbon nanocages, fullerynes: a DFT study
Scientific Reports (2021)
-
Radical character of non-IPR isomer 28324 of C80 fullerene
Russian Chemical Bulletin (2021)
-
Diels–Alder cycloaddition of the silicon–silicon bonds at pentagon junctions of Si-doped non-IPR and SW defective fullerenes
Monatshefte für Chemie - Chemical Monthly (2021)
-
Features of molecular structure of small non-IPR fullerenes: the two isomers of C50
Theoretical Chemistry Accounts (2020)
-
Li-Decorated Fullerenes: A DFT Study
Journal of Cluster Science (2019)