Abstract
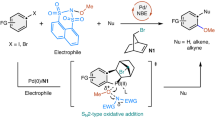
Direct arylation of C–H bonds is in principle a powerful way of preparing value-added molecules that contain carbon–aryl fragments. Unfortunately, currently available synthetic methods are not sufficiently effective to be practical alternatives to conventional cross-coupling reactions. We propose that the main problem lies in the late portion of the catalytic cycle where reductive elimination gives the desired carbon–aryl bond. Accordingly, we have developed a strategy where the Ir(III) centre of the key intermediate is first oxidized to Ir(IV). Density functional theory calculations indicate that the barrier to reductive elimination is reduced by nearly 19 kcal mol–1 for this oxidized complex compared with that of its Ir(III) counterpart. Various experiments confirm this prediction, affording a new methodology capable of directly arylating C–H bonds at room temperature with a broad substrate scope and in good yields. This work highlights how the oxidation states of intermediates can be targeted deliberately to catalyse an otherwise impossible reaction.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
13 February 2018
In the version of this Article originally published, the oxidation states of the iridium centres in figures 2, 4 and 5 were formatted incorrectly. These have been corrected after print.
References
Dyker, G. Handbook of C−H Transformations Applications in Organic Synthesis (Wiley-VCH, 2005).
Ackermann, L., Vicente, R. & Kapdi, A. R. Transition-metal-catalyzed direct arylation of (hetero)arenes by C–H bond cleavage. Angew. Chem. Int. Ed. 48, 9792–9826 (2009).
Liu, C., Zhang, H., Shi, W. & Lei, A. Bond formations between two nucleophiles: transition metal catalyzed oxidative cross-coupling reactions. Chem. Rev. 111, 1780–1824 (2011).
Giri, R., Thapa, S. & Kafle, A. Palladium-catalysed, directed C–H coupling with organometallics. Adv. Synth. Catal. 356, 1395–1411 (2014).
Lapointe, D. & Fagnou, K. Overview of the mechanistic work on the concerted metallation–deprotonation pathway. Chem. Lett. 39, 1118–1126 (2010).
Chu, J.-H., Lin, P.-S. & Wu, M.-J. Palladium(II)-catalyzed ortho arylation of 2-phenoxypyridines with potassium aryltrifluoroborates via C–H functionalization. Organometallics 29, 4058–4065 (2010).
Li, W., Yin, Z., Jiang, X. & Sun, P. Palladium-catalyzed direct ortho C–H arylation of 2-arylpyridine derivatives with aryltrimethoxysilane. J. Org. Chem. 76, 8543–8548 (2011).
Romero-Revilla, J. A., García-Rubia, A., Goméz Arrayás, R., Fernández-Ibáñez, M. Á. & Carretero, J. C. Palladium-catalyzed coupling of arene C–H bonds with methyl- and arylboron reagents assisted by the removable 2-pyridylsulfinyl group. J. Org. Chem. 76, 9525–9530 (2011).
Spangler, J. E., Kobayashi, Y., Verma, P., Wang, D.-H. & Yu, J.-Q. α-Arylation of saturated azacycles and N-methylamines via palladium(II)-catalyzed C(sp3)–H coupling. J. Am. Chem. Soc. 137, 11876–11879 (2015).
Komiya, S., Albright, T. A., Hoffmann, R. & Kochi, J. K. The stability of organogold compounds. Hydrolytic, thermal, and oxidative cleavages of dimethylaurate(I) and tetramethylaurate(III). J. Am. Chem. Soc. 99, 8440–8447 (1977).
Lau, W., Huffman, J. C. & Kochi, J. K. Electrochemical oxidation–reduction of organometallic complexes. Effect of the oxidation state on the pathways for reductive elimination of dialkyliron complexes. Organometallics 1, 155–169 (1982).
Seligson, A. L. & Trogler, W. C. One-electron oxidative cleavage of palladium(II) alkyl and phenoxo bonds. J. Am. Chem. Soc. 114, 7085–7089 (1992).
Pedersen, A. & Tilset, M. Oxidatively induced reductive eliminations. Kinetics and mechanism of the elimination of ethane from the 17-electron cation radical of rhodium complex Cp*Rh(PPh3)(CH3)2 . Organometallics 12, 56–64 (1993).
Koo, K. & Hillhouse, G. L. Carbon–nitrogen bond formation by reductive elimination from nickel(II) amido alkyl complexes. Organometallics 14, 4421–4423 (1995).
Lanci, M. P., Remy, M. S., Kaminsky, W., Mayer, J. M. & Sanford, M. S. Oxidatively induced reductive elimination from (tBu2bpy)Pd(Me)2: palladium(IV) intermediates in a one-electron oxidation reaction. J. Am. Chem. Soc. 131, 15618–15620 (2009).
Bour, J. R. et al. Carbon–carbon bond-forming reductive elimination from isolated nickel(III) complexes. J. Am. Chem. Soc. 138, 16105–16111 (2016).
Watson, M. B., Rath, N. P. & Mirica, L. M. Oxidative C–C bond formation reactivity of organometallic Ni(II), Ni(III), and Ni(IV) complexes. J. Am. Chem. Soc. 139, 35–38 (2017).
Neufeldt, S. R., Seigerman, C. K. & Sanford, M. S. Mild palladium-catalyzed C–H alkylation using potassium alkyltrifluoroborates in combination with MnF3 . Org. Lett. 15, 2302–2305 (2013).
Seigerman, C. K., Micyus, T. M., Neufeldt, S. R. & Sanford, M. S. Palladium-catalyzed C–H arylation using aryltrifluoroborates in conjunction with a MnIII oxidant under mild conditions. Tetrahedron 69, 5580–5587 (2013).
Tasker, S. Z. & Jamison, T. F. Highly regioselective indoline synthesis under nickel/photoredox dual catalysis. J. Am. Chem. Soc. 137, 9531–9534 (2015).
Terrett, J. A., Cuthbertson, J. D., Shurtleff, V. W. & MacMillan, D. W. Switching on elusive organometallic mechanisms with photoredox catalysis. Nature 524, 330–334 (2015).
Corcoran, E. B. et al. Aryl amination using ligand-free Ni(II) salts and photoredox catalysis. Science 353, 279–283 (2016).
Shin, K., Park, S.-W. & Chang, S. Cp*Ir(III)-catalyzed mild and broad C–H arylation of arenes and alkenes with aryldiazonium salts leading to the external oxidant-free approach. J. Am. Chem. Soc. 137, 8584–8592 (2015).
Li, L., Brennessel, W. W. & Jones, W. D. C−H activation of phenyl imines and 2-phenylpyridines with [Cp*MCl2]2 (M = Ir, Rh): regioselectivity, kinetics, and mechanism. Organometallics 28, 3492–3500 (2009).
Yang, S., Li, B., Wan, X. & Shi, Z. Ortho arylation of acetanilides via Pd(II)-catalyzed C–H functionalization. J. Am. Chem. Soc. 129, 6066–6067 (2007).
Zhou, H., Xu, Y.-H., Chung, W.-J. & Loh, T.-P. Palladium-catalyzed direct arylation of cyclic enamides with aryl silanes by sp2 C–H activation. Angew. Chem. Int. Ed. 48, 5355–5357 (2009).
Senthilkumar, N., Parthasarathy, K., Gandeepan, P. & Cheng, C.-H. Synthesis of phenanthridinones from N-methoxybenzamides and aryltriethoxysilanes through Rh(III)-catalyzed C–H and N–H bond activation. Chem. Asian J. 8, 2175–2181 (2013).
Lu, M.-Z., Lu, P., Xu, Y.-H. & Loh, T.-P. Mild Rh(III)-catalyzed direct C–H bond arylation of (hetero)arenes with arylsilanes in aqueous media. Org. Lett. 16, 2614–2617 (2014).
He, J., Takise, R., Fu, H. & Yu, J.-Q. Ligand-enabled cross-coupling of C(sp3)–H bonds with arylsilanes. J. Am. Chem. Soc. 137, 4618–4621 (2015).
Zhao, S., Liu, B., Zhan, B.-B., Zhang, W.-D. & Shi, B.-F. Nickel-catalyzed ortho-arylation of unactivated (hetero)aryl C–H bonds with arylsilanes using a removable auxiliary. Org. Lett. 18, 4586–4589 (2016).
Nareddy, P., Jordan, F. & Szostak, M. Highly chemoselective ruthenium(II)-catalyzed direct arylation of cyclic and N,N-dialkyl benzamides with aryl silanes. Chem. Sci. 8, 3204–3210 (2017).
Shukla, K. H. & DeShong, P. Studies on the mechanism of allylic coupling reactions: a Hammett analysis of the coupling of aryl silicate derivatives. J. Org. Chem. 73, 6283–6291 (2008).
Saijo, H., Ohashi, M. & Ogoshi, S. Fluoroalkylcopper(I) complexes generated by the carbocupration of tetrafluoroethylene: construction of a tetrafluoroethylene-bridging structure. J. Am. Chem. Soc. 136, 15158–15161 (2014).
Nakao, Y., Imanaka, H., Sahoo, A. K., Yada, A. & Hiyama, T. Alkenyl- and aryl[2-(hydroxymethyl)phenyl]dimethylsilanes: an entry to tetraorganosilicon reagents for the silicon-based cross-coupling reaction. J. Am. Chem. Soc. 127, 6952–6953 (2005).
Herron, J. R. & Ball, Z. T. Synthesis and reactivity of functionalized arylcopper compounds by transmetalation of organosilanes. J. Am. Chem. Soc. 130, 16486–16487 (2008).
Oeschger, R. J. & Chen, P. Structure and gas-phase thermochemistry of a Pd/Cu complex: studies on a model for transmetalation transition states. J. Am. Chem. Soc. 139, 1069–1072 (2017).
Nakao, Y. & Hiyama, T. Silicon-based cross-coupling reaction: an environmentally benign version. Chem. Soc. Rev. 40, 4893–4901 (2011).
Amatore, C., Grimaud, L., Le Duc, G. & Jutand, A. Three roles for the fluoride ion in palladium-catalyzed Hiyama reactions: transmetalation of [ArPdFL2] by Ar′Si(OR)3 . Angew. Chem. Int. Ed. 53, 6982–6985 (2014).
Whitaker, D., Burés, J. & Larrosa, I. Ag(I)-catalyzed C–H activation: the role of the Ag(I) salt in Pd/Ag-mediated C–H arylation of electron-deficient arenes. J. Am. Chem. Soc. 138, 8384–8387 (2016).
Lotz, M. D., Camasso, N. M., Canty, A. J. & Sanford, M. S. Role of silver salts in palladium-catalyzed arene and heteroarene C–H functionalization reactions. Organometallics 36, 165–171 (2017).
Shen, Q. & Hartwig, J. F. Lewis acid acceleration of C–N bond-forming reductive elimination from heteroarylpalladium complexes and catalytic amidation of heteroaryl bromides. J. Am. Chem. Soc. 129, 7734–7735 (2007).
Brewster, T. P. et al. An iridium(IV) species, [Cp*Ir(NHC)Cl]+, related to a water-oxidation catalyst. Organometallics 30, 965–973 (2011).
Graeupner, J. et al. Electron-rich CpIr(biphenyl-2,2′-diyl) complexes with π-accepting carbon donor ligands. Organometallics 31, 7158–7164 (2012).
Kang, J. W. & Maitlis, P. M. (Pentamethylcyclopentadienyl)rhodium and -iridium complexes. V. Complexes with oxy-ligands and the exchange of methyl protons by deuterium under basic conditions. J. Organomet. Chem. 30, 127–133 (1971).
Huang, L., Hackenberger, D. & Gooßen, L. J. Iridium-catalyzed ortho-arylation of benzoic acids with arenediazonium salts. Angew. Chem. Int. Ed. 54, 12607–12611 (2015).
Gao, P. et al. Iridium(III)-catalyzed direct arylation of C–H bonds with diaryliodonium salts. J. Am. Chem. Soc. 137, 12231–12240 (2015).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemistry: calling all engineers. Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.201707584 (2017).
Acknowledgements
This research was supported by the Institute for Basic Science (IBS-R010-D1) in Korea.
Author information
Authors and Affiliations
Contributions
K.S., Y.P., M.-H.B. and S.C. conceived and designed the project and wrote the manuscript. K.S. and Y.P. carried out the experiments. Y.P. performed DFT calculations. S.C. organized the research. All authors analysed the data, discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 5361 kb)
Supplementary information
Crystallographic data for compound IIa (CIF 27 kb)
Supplementary information
Structure factors file for compound IIa (FCF 322 kb)
Rights and permissions
About this article
Cite this article
Shin, K., Park, Y., Baik, MH. et al. Iridium-catalysed arylation of C–H bonds enabled by oxidatively induced reductive elimination. Nature Chem 10, 218–224 (2018). https://doi.org/10.1038/nchem.2900
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2900
This article is cited by
-
Unconventional mechanism and selectivity of the Pd-catalyzed C–H bond lactonization in aromatic carboxylic acid
Nature Communications (2022)
-
Transition metal-catalysed directed C–H functionalization with nucleophiles
Nature Synthesis (2022)