Abstract
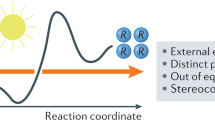
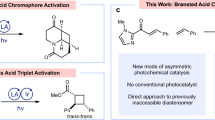
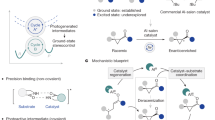
Chiral iminium ions—generated upon condensation of α,β-unsaturated aldehydes and amine catalysts—are used extensively by chemists to make chiral molecules in enantioenriched form. In contrast, their potential to absorb light and promote stereocontrolled photochemical processes remains unexplored. This is despite the fact that visible-light absorption by iminium ions is a naturally occurring event that triggers the mechanism of vision in higher organisms. Herein we demonstrate that the direct excitation of chiral iminium ions can unlock unconventional reaction pathways, enabling enantioselective catalytic photochemical β-alkylations of enals that cannot be realized via thermal activation. The chemistry uses readily available alkyl silanes, which are recalcitrant to classical conjugate additions, and occurs under illumination by visible-light-emitting diodes. Crucial to success was the design of a chiral amine catalyst with well-tailored electronic properties that can generate a photo-active iminium ion while providing the source of stereochemical induction. This strategy is expected to offer new opportunities for reaction design in the field of enantioselective catalytic photochemistry.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dalko, P. I. (ed.) Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications (Wiley-VCH, 2013).
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
Lelais, G. & MacMillan, D. W. C. Modern strategies in organic catalysis: the advent and development of iminium activation. Aldrichim. Acta 39, 79–87 (2006).
Córdova, A. (ed.) Catalytic Asymmetric Conjugate Reactions (Wiley-VCH, 2010).
Wald, G. Molecular basis of visual excitation. Science 162, 230–239 (1968).
Ernst, O. P. et al. Microbial and animal rhodopsins: structures, functions, and molecular mechanisms. Chem. Rev. 114, 126−163 (2014).
Nathans, J., Thomas, D. & Hogness, D. S. Molecular genetics of human color vision: the genes encoding blue, green, and red pigments. Science 232, 193–202 (1986).
Mariano, P. S. The photochemistry of iminium salts and related heteroaromatic systems. Tetrahedron 39, 3845–3879 (1983).
Borg, R. M., Heuckeroth, R. O., Lan, A. J. Y., Quillen, S. L. & Mariano, P. S. Arene-iminium salt electron-transfer photochemistry. Mechanistically interesting photoaddition processes. J. Am. Chem. Soc. 109, 2728–2737 (1987).
Chen, C., Chang, V., Cai, X., Duesler, E. & Mariano, P. S. A general strategy for absolute stereochemical control in enone-olefin [2 + 2] photocycloaddition reactions. J. Am. Chem. Soc. 123, 6433–6434 (2001).
Mariano, P. S. Electron-transfer mechanisms in photochemical transformations of iminium salts. Acc. Chem. Res. 16, 130–144 (1983).
Schultz, D. M. & Yoon, T. P. Solar synthesis: prospects in visible light photocatalysis. Science 343, 1239176 (2014).
Brimioulle, R., Lenhart, D., Maturi, M. M. & Bach, T. Enantioselective catalysis of photochemical reactions. Angew. Chem. Int. Ed. 54, 3872–3890 (2015).
Arceo, E., Jurberg, I. D., Álvarez-Fernández, A. & Melchiorre, P. Photochemical activity of a key donor–acceptor complex can drive stereoselective catalytic α-alkylation of aldehydes. Nat. Chem. 5, 750−756 (2013).
Silvi, M., Arceo, E., Jurberg, I. D., Cassani, C. & Melchiorre, P. Enantioselective organocatalytic alkylation of aldehydes and enals driven by the direct photoexcitation of enamines. J. Am. Chem. Soc. 137, 6120–6123 (2015).
Bahamonde, A. & Melchiorre, P. Mechanism of the stereoselective α-alkylation of aldehydes driven by the photochemical activity of enamines. J. Am. Chem. Soc. 138, 8019−8030 (2016).
Mukherjee, S., Yang, J. W., Hoffmann, S. & List, B. Asymmetric enamine catalysis. Chem. Rev. 107, 5471–5569 (2007).
Balzani, V. Ceroni, P. & Juris, A. in Photochemistry and Photophysics 103–123 (Wiley-VCH, 2014).
Yoshida, J., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Dockery, K. P. et al. Nucleophile-assisted cleavage of benzyltrialkylsilane cation radicals. J. Am. Chem. Soc. 119, 1876–1883 (1997).
Yoon, U. C., Mariano, P. S., Givens, R. S. & Atwater, B. W. in Advances in Electron Transfer Chemistry. Vol. 4, 117–206 (JAI, 1994).
Pirnot, M. T., Rankic, D. A., Martin, D. B. C. & MacMillan, D. W. C. Photoredox activation for the direct β-arylation of ketones and aldehydes. Science 339, 1593–1596 (2013).
Terrett, J. A., Clift, M. D. & MacMillan, D. W. C. Direct β-alkylation of aldehydes via photoredox organocatalysis. J. Am. Chem. Soc. 136, 6858–6861 (2014).
Yoshida, J., Murata, T. & Isoe, S. Electrochemical oxidation of organosilicon compounds I. Oxidative cleavage of carbon-silicon bond in allylsilanes and benzylsilanes. Tetrahedron Lett. 27, 3373–3376 (1986).
Jensen, K. L., Dickmeiss, G., Jiang, H., Albrecht, Ł. & Jørgensen, K. A. The diarylprolinol silyl ether system: a general organocatalyst. Acc. Chem. Res. 45, 248–264 (2012).
Hu, J., Wang, J., Nguyen, T. H. & Zheng, N. The chemistry of amine radical cations produced by visible light photoredox catalysis. Beilstein J. Org. Chem. 9, 1977–2001 (2013).
Müller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Morgenthaler, M. et al. Predicting and tuning physicochemical properties in lead optimization: amine basicities. Chem. Med. Chem. 2, 1100–1115 (2007).
Zimmer, L. E., Sparr, C. & Gilmour, R. Fluorine conformational effects in organocatalysis: an emerging strategy for molecular design. Angew. Chem. Int. Ed. 50, 11860–11871 (2011).
Kim, S.-H. & Rieke, R. D. Benzylic manganese halides, sulfonates, and phosphates: preparation, coupling reactions, and applications in organic synthesis. J. Org. Chem. 65, 2322–2330 (2000).
Van Heerden, P. S., Bezuidenhoudt, B. C. B., Steenkamp, J. A. & Ferreira, D. Conjugate addition of benzyl copper reagents to α,α-enoates and enones. Tetrahedron Lett. 33, 2383–2386 (1992).
Fañanás-Mastral, M. & Feringa, B. L. Copper-catalyzed regio- and enantioselective synthesis of chiral enol acetates and β-substituted aldehydes. J. Am. Chem. Soc. 132, 13152–13153 (2010).
Dell'Amico, L., Companyó, X., Naicker, T., Bräuer, T. M. & Jørgensen, K. A. Asymmetric organocatalytic benzylation of α,β-unsaturated aldehydes with toluenes. Eur. J. Org. Chem. 2013, 5262–5265 (2013).
Li, T. et al. A strategy enabling enantioselective direct conjugate addition of inert aryl methane nucleophiles to enals with a chiral amine catalyst under mild conditions. Chem. Eur. J. 19, 9147–9150 (2013).
Walbiner, M., Wu, J. Q. & Fischer, H. Absolute rate constant for the addition of benzyl and cumyl radicals to alkenes in solution. Helvetica Chim. Acta 78, 910–924 (1995).
Sibi, M. P., Liu, P., Ji, J., Hajra, S. & Chen, J.-x. Free-radical-mediated conjugate additions. enantioselective synthesis of butyrolactone natural products: (–)-enterolactone, (–)-arctigenin, (–)-isoarctigenin, (–)-nephrosteranic acid, and (–)-roccellaric acid. J. Org. Chem. 67, 1738–1745 (2002).
Huo, H., Harms, K. & Meggers, E. Catalytic, enantioselective addition of alkyl radicals to alkenes via visible-light-activated photoredox catalysis with a chiral rhodium complex. J. Am. Chem. Soc. 138, 6936−6939 (2016).
Montanaro, S., Ravelli, D., Merli, D., Fagnoni, M. & Albini, A. Decatungstate as photoredox catalyst: benzylation of electron-poor olefins. Org. Lett. 14, 4218–4221 (2012).
Murphy, J. J., Bastida, D., Paria, S., Fagnoni, M. & Melchiorre, P. Asymmetric catalytic formation of quaternary carbons by iminium ion trapping of radicals. Nature 532, 218−222 (2016).
Acknowledgements
Financial support was provided by the CERCA Programme (Generalitat de Catalunya), MINECO (Severo Ochoa Excellence Accreditation 2014-2018, SEV-2013-0319), and the European Research Council (ERC-2015-CoG 681840 - CATA-LUX). C.V. thanks the Marie Skłodowska-Curie Actions for a postdoctoral fellowship (H2020-MSCA-IF-2014 658980). L.B. thanks MINECO for a predoctoral fellowship (CTQ2013-45938-P). The authors thank M. Moliterno for helpful discussions. This work is dedicated to V. Balzani on the occasion of his 80th birthday.
Author information
Authors and Affiliations
Contributions
M.S. was involved in the discovery and initial development of the light-driven reactions. C.V. and Y.P.R. designed and synthesized the catalysts. C.V., Y.P.R. and L.B. performed the experiments. All of the authors analysed the data and designed the experiments. P.M directed the project and wrote the manuscript with contributions from all of the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 12610 kb)
Supplementary information
Crystallographic data for compound 1c. (CIF 2371 kb)
Supplementary information
Crystallographic data for compound 5a. (CIF 2802 kb)
Supplementary information
Crystallographic data for compound Ic. (CIF 1815 kb)
Rights and permissions
About this article
Cite this article
Silvi, M., Verrier, C., Rey, Y. et al. Visible-light excitation of iminium ions enables the enantioselective catalytic β-alkylation of enals. Nature Chem 9, 868–873 (2017). https://doi.org/10.1038/nchem.2748
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2748
This article is cited by
-
New strategies for asymmetric photocatalysis: asymmetric organocatalytic/photoredox relay catalysis for efficient synthesis of polycyclic compounds containing vicinal amino alcohols
Science China Chemistry (2024)
-
Stereoselective conjugate cyanation of enals by combining photoredox and organocatalysis
Nature Catalysis (2023)
-
Direct electrophilic and radical isoperfluoropropylation with i-C3F7-Iodine(III) reagent (PFPI reagent)
Communications Chemistry (2023)
-
Light-induced phosphine-catalyzed asymmetric functionalization of benzylic C-H bonds
Science China Chemistry (2023)
-
Photocatalytic three-component asymmetric sulfonylation via direct C(sp3)-H functionalization
Nature Communications (2021)