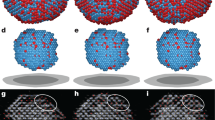
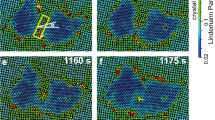
Abstract
How molecules in solution form crystal nuclei, which then grow into large crystals, is a poorly understood phenomenon. The classical mechanism of homogeneous crystal nucleation proceeds via the spontaneous random aggregation of species from liquid or solution. However, a non-classical mechanism suggests the formation of an amorphous dense phase that reorders to form stable crystal nuclei. So far it has remained an experimental challenge to observe the formation of crystal nuclei from five to thirty molecules. Here, using polyoxometallates, we show that the formation of small crystal nuclei is observable by cryogenic transmission electron microscopy. We observe both classical and non-classical nucleation processes, depending on the identity of the cation present. The experiments verify theoretical studies that suggest non-classical nucleation is the lower of the two energy pathways. The arrangement in just a seven-molecule proto-crystal matches the order found by X-ray diffraction of a single bulk crystal, which demonstrates that the same structure was formed in each case.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gibbs, J. W. On the equilibrium of heterogeneous substances (first part). Trans. Connect. Acad. Sci. 3, 108–248 (1876).
Gibbs, J. W. On the equilibrium of heterogeneous substances (concluded). Trans. Connect. Acad. Sci. 16, 343–524 (1878).
Schüth, F., Bussian, P., Ågren, F., Schunk, S. & Lindén, M. Techniques for analyzing the early stages of crystallization reactions. Solid State Sci. 3, 801–808 (2001).
Erdemir, D., Lee, A. Y. & Myerson, A. S. Nucleation of crystals from solution: classical and two-step models. Acc. Chem. Res. 42, 621–629 (2009).
Chayen, N. E., Saridakis, E. & Sear, R. P. Experiment and theory for heterogeneous nucleation of protein crystals in a porous medium. Proc. Natl Acad. Sci. USA 103, 597–601 (2006).
Varanasi, K. K., Hsu, M., Bhate, N., Yang, W. & Deng, T. Spatial control in the heterogeneous nucleation of water. Appl. Phys. Lett. 95, 094101 (2009).
Kashchiev, D. & van Rosmalen, G. M. Review: nucleation in solutions revisited. Cryst. Res. Technol. 38, 555–574 (2003).
Watson, J. N. et al. TPA−silicalite crystallization from homogeneous solution: kinetics and mechanism of nucleation and growth. J. Phys. Chem. B 101, 10094–10104 (1997).
Vekilov, P. G. & Galkin, O. Are nucleation kinetics of protein crystals similar to those of liquid droplets? J. Am. Chem. Soc. 122, 156–163 (2000).
Galkin, O. & Vekilov, P. G. Direct determination of the nucleation rates of protein crystals. J. Phys. Chem. B 103, 10965–10971 (1999).
Banfield, J. F., Welch, S. A., Zhang, H., Ebert, T. T. & Penn, R. L. Aggregation-based crystal growth and microstructure development in natural iron oxyhydroxide biomineralization products. Science 289, 751–754 (2000).
Baumgartner, J. et al. Nucleation and growth of magnetite from solution. Nat. Mater. 12, 310–314 (2013).
Van Driessche, A. E. S. et al. The role and implications of bassanite as a stable precursor phase to gypsum precipitation. Science 336, 69–72 (2012).
Gebauer, D., Volkel, A. & Colfen, H. Stable prenucleation calcium carbonate clusters. Science 322, 1819–1822 (2008).
De Yoreo, J. J. et al. Crystallization by particle attachment in synthetic, biogenic, and geologic environments. Science 349, aaa6760 (2015).
ten Wolde, P. R. & Frenkel, D. Enhancement of protein crystal nucleation by critical density fluctuations. Science 277, 1975–1978 (1997).
Wolf, S. E., Leiterer, J., Kappl, M., Emmerling, F. & Tremel, W. Early homogenous amorphous precursor stages of calcium carbonate and subsequent crystal growth in levitated droplets. J. Am. Chem. Soc. 130, 12342–12347 (2008).
Galkin, O., Chen, K., Nagel, R. L., Hirsch, R. E. & Vekilov, P. G. Liquid–liquid separation in solutions of normal and sickle cell hemoglobin. Proc. Natl Acad. Sci. USA 99, 8479–8483 (2002).
Vekilov, P. G. Nucleation. Cryst. Growth Des. 10, 5007–5019 (2010).
Yuwono, V. M., Burrows, N. D., Soltis, J. A. & Penn, R. L. Oriented aggregation: formation and transformation of mesocrystal intermediates revealed. J. Am. Chem. Soc. 132, 2163–2165 (2010).
Niederberger, M. & Cölfen, H. Oriented attachment and mesocrystals: non-classical crystallization mechanisms based on nanoparticle assembly. Phys. Chem. Chem. Phys. 8, 3271–3287 (2006).
Nielsen, M. H., Aloni, S. & De Yoreo, J. J. In situ TEM imaging of CaCO3 nucleation reveals coexistence of direct and indirect pathways. Science 345, 1158–1162 (2014).
Li, D. et al. Direction-specific interactions control crystal growth by oriented attachment. Science 336, 1014–1018 (2012).
Chen, Q. et al. Interaction potentials of anisotropic nanocrystals from the trajectory sampling of particle motion using in situ liquid phase transmission electron microscopy. ACS Cent. Sci. 1, 33–39 (2015).
Lupulescu, A. I. & Rimer, J. D. In situ imaging of silicalite-1 surface growth reveals the mechanism of crystallization. Science 344, 729–732 (2014).
Yau, S. T. & Vekilov, P. G. Direct observation of nucleus structure and nucleation pathways in apoferritin crystallization. J. Am. Chem. Soc. 123, 1080–1089 (2001).
Pope, M. T. Heteropoly and Isopoly Oxometalates (Springer, 1983).
Qiu, J. & Burns, P. C. Clusters of actinides with oxide, peroxide or hydroxide bridges. Chem. Rev. 113, 1097–1120 (2013).
Liu, T., Diemann, E., Li, H., Dress, A. W. M. & Muller, A. Self-assembly in aqueous solution of wheel-shaped Mo154 oxide clusters into vesicles. Nature 426, 59–62 (2003).
Chauveau, F., Doppelt, P. & Lefebvre, J. Fluorotungstates of the metatungstate family: identification and properties of one compound of the 2–18 series. Inorg. Chem. 19, 2803–2806 (1980).
Sharet, S. et al. Orientations of polyoxometalate anions on gold nanoparticles. Dalton Trans. 41, 9849–9851 (2012).
Soltis, J. A., Wallace, C. M., Penn, R. L. & Burns, P. C. Cation-dependent hierarchical assembly of U60 nanoclusters into macro-ion assemblies imaged via cryogenic transmission electron microscopy. J. Am. Chem. Soc. 138, 191–198 (2016).
Spitsyn, V. I. & Babaev, N. B. Solubility of difficultly soluble alkali metal heteropolyacids. Russ. J. Inorg. Chem. 5, 580–585 (1960).
Cho, H. J. et al. Measurement of ice thickness on vitreous ice embedded cryo-EM grids: investigation of optimizing condition for visualizing macromolecules. J. Anal. Sci. Technol. 4, 7 (2013).
Vasudeva Rao, P. R. & Kolarik, Z. A review of third phase formation in extraction of actinides by neutral organophosphorus extractants. Solvent Extr. Ion Exch. 14, 955–993 (1996).
Schreiber, R. E. et al. Reactivity and O2 formation by Mn(IV)- and Mn(V)-hydroxo species stabilized within a polyfluoroxometalate framework. J. Am. Chem. Soc. 137, 8738–8748 (2015).
Wallace, A. F. et al. Microscopic evidence for liquid–liquid separation in supersaturated CaCO3 solutions. Science 341, 885–889 (2012).
Egerton, R. Physical Principles of Electron Microscopy: An Introduction to TEM, SEM, and AEM (Springer, 2005).
Pronk, S. et al. GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 29, 845–854 (2013).
Wang, J. M., Cieplak, P. & Kollman, P. A. How well does a restrained electrostatic potential (RESP) model perform in calculating conformational energies of organic and biological molecules? J. Comput. Chem. 21, 1049–1074 (2000).
Chaumont, A. & Wipff, G. Ion aggregation in concentrated aqueous and methanol solutions of polyoxometallates Keggin anions: the effect of counterions investigated by molecular dynamics simulations. Phys. Chem. Chem. Phys. 10, 6940–6953 (2008).
López, X., Nieto-Draghi, C., Bo, C., Bonet-Avalos, J. & Poblet, J. M. Polyoxometalates in solution: molecular dynamics simulations on the α-PW12O403− Keggin anion in aqueous media. J. Phys. Chem. A 109, 1216–1222 (2005).
Hay, P. J. & Wadt, W. R. Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 82, 270–283 (1985).
van Lenthe, E., Ehlers, A. & Baerends, E. J. Geometry optimizations in the zero order regular approximation for relativistic effects. J. Chem. Phys. 110, 8943–8953 (1999).
Pye, C. C. & Ziegler, T. An implementation of the conductor-like screening model of solvation within the Amsterdam density functional package. Theor. Chem. Acc. 101, 396–408 (1999).
Dang, L. X. Development of nonadditive intermolecular potentials using molecular-dynamics—solvation of Li+ and F− ions in polarizable water. J. Chem. Phys. 96, 6970–6977 (1992).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926 (1983).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an Nlog(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Bussi, G., Donadio, D. & Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 126, 014101 (2007).
Hockney, R. W., Goel, S. P. & Eastwood, J. W. Quiet high-resolution computer models of a plasma. J. Comp. Phys. 14, 148–158 (1974).
Acknowledgements
The research at the Weizmann Institute of Science was funded by the Israel Science Foundation grant no. 763/14 and electron microscopy experiments were partially funded by Irving and Cherna Moscowitz Center for Nano and Bio-Nano Imaging of the Weizmann Institute of Science. R.N. is the Rebecca and Israel Sieff Professor of Chemistry. Research at Universitat Rovira i Virgili was supported by the Spanish Ministry of Science and Innovation (grant CTQ2014-52774-P) and the Generalitat de Catalunya (2014SGR199 and XRQTC). J.M.P. thanks the ICREA (Catalan Institution for Research and Advanced Studies) Foundation for an ICREA Academia award.
Author information
Authors and Affiliations
Contributions
R.E.S. carried out the majority of the research, R.E.S., L.H. and S.G.W. carried out the electron microscopy measurements and analysed tomography data, G.L. carried out the X-ray diffraction measurements, Z.-l.L., J.J.C. and J.M.P. did the molecular dynamics calculations and R.N. supervised the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2363 kb)
Supplementary information
Crystallographic data for compound Na2K6-MnPFOM. (CIF 735 kb)
Supplementary information
Crystallographic data for compound Cs5.5K2.5-MnPFOM. (CIF 892 kb)
Supplementary movie
Supplementary movie 1 (MP4 4781 kb)
Supplementary movie
Supplementary movie 2 (MP4 868 kb)
Supplementary movie
Supplementary movie 3 (MP4 2823 kb)
Supplementary movie
Supplementary movie 4 (MP4 4518 kb)
Supplementary movie
Supplementary movie 5 (MP4 364 kb)
Supplementary movie
Supplementary movie 6 (MP4 3651 kb)
Supplementary movie
Supplementary movie 7 (MP4 1790 kb)
Supplementary movie
Supplementary movie 8 (MP4 2773 kb)
Supplementary movie
Supplementary movie 9 (MP4 893 kb)
Rights and permissions
About this article
Cite this article
Schreiber, R., Houben, L., Wolf, S. et al. Real-time molecular scale observation of crystal formation. Nature Chem 9, 369–373 (2017). https://doi.org/10.1038/nchem.2675
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2675
This article is cited by
-
Impact of molecular symmetry on crystallization pathways in highly supersaturated KH2PO4 solutions
Nature Communications (2024)
-
Synthesis and characterization of a new copper-based polyoxomolybdate and its catalytic activity for azide-alkyne cycloaddition reaction under UV light irradiation
Scientific Reports (2024)
-
Polyoxometalate Clusters: Inorganic Ligands for Functional Materials
Journal of Cluster Science (2022)
-
Temperature-dependent kinetic pathways of heterogeneous ice nucleation competing between classical and non-classical nucleation
Nature Communications (2021)
-
Crystallisation in a two-dimensional granular system at constant temperature
Scientific Reports (2021)