Abstract
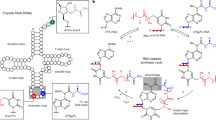
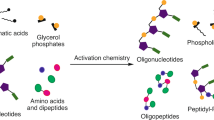
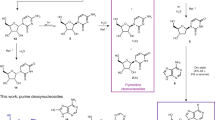
A minimal cell can be thought of as comprising informational, compartment-forming and metabolic subsystems. To imagine the abiotic assembly of such an overall system, however, places great demands on hypothetical prebiotic chemistry. The perceived differences and incompatibilities between these subsystems have led to the widely held assumption that one or other subsystem must have preceded the others. Here we experimentally investigate the validity of this assumption by examining the assembly of various biomolecular building blocks from prebiotically plausible intermediates and one-carbon feedstock molecules. We show that precursors of ribonucleotides, amino acids and lipids can all be derived by the reductive homologation of hydrogen cyanide and some of its derivatives, and thus that all the cellular subsystems could have arisen simultaneously through common chemistry. The key reaction steps are driven by ultraviolet light, use hydrogen sulfide as the reductant and can be accelerated by Cu(I)–Cu(II) photoredox cycling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gánti, T. The Principles of Life (Oxford Univ. Press, 2003).
Dyson, F. Origins of Life 2nd edn (Cambridge Univ. Press, 1999).
Orgel, L. E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 39, 99–123 (2004).
Segré, D., Ben-Eli, D., Deamer, D. W. & Lancet, D. The lipid world. Orig. Life Evol. Biosph. 31, 119–145 (2001).
Wächtershäuser, G. Groundworks for an evolutionary biochemistry: the iron–sulphur world. Prog. Biophys. Molec. Biol. 58, 85–201 (1992).
Powner, M. W., Gerland, B. & Sutherland, J. D. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459, 239–242 (2009).
Mullen, L. B. & Sutherland, J. D. Simultaneous nucleotide activation and synthesis of amino acid amides by a potentially prebiotic multi-component reaction. Angew. Chem. Int. Ed. 46, 8063–8066 (2007).
Ritson, D. & Sutherland, J. D. Prebiotic synthesis of simple sugars by photoredox systems chemistry. Nature Chem. 4, 895–899 (2012).
Ritson, D. J. & Sutherland, J. D. Synthesis of aldehydic ribonucleotide and amino acid precursors by photoredox chemistry. Angew. Chem. Int. Ed. 52, 5845–5847 (2013).
Yaylayan, V. A., Harty-Majors, S. & Ismail, A. A. Investigation of DL-glyceraldehyde-dihydroxyacetone interconversion by FTIR spectroscopy. Carbohydr. Res. 318, 20–25 (1999).
Butzow, J. J. & Eichorn, G. L. Interactions of metal ions with polynucleotides and related compounds. IV. Degradation of polyribonucleotides by zinc and other divalent metal ions. Biopolymers 3, 95–107 (1965).
Lombard, J., López-García, P. & Moreira, D. The early evolution of lipid membranes and the three domains of life. Nature Rev. Microbiol. 10, 507–515 (2012).
Schlesinger, G. & Miller, S. L. Equilibrium and kinetics of glyconitrile formation in aqueous solution. J. Am. Chem. Soc. 95, 3729–3735 (1973).
Kurosawa, K. et al. Hydrogen cyanide production due to mid-size impacts in a redox-neutral N2-rich atmosphere. Orig. Life Evol. Biosph. 43, 221–245 (2013).
Pasek, M. A. & Lauretta, D. S. Aqueous corrosion of phosphide minerals from iron meteorites: a highly reactive source of prebiotic phosphorus on the surface of the early Earth. Astrobiology 5, 515–535 (2005).
Bryant, D. E. & Kee, T. P. Direct evidence for the availability of reactive, water soluble phosphorus on the early Earth. H-Phosphinic acid from the Nantan meteorite. Chem. Commun. 2344–2346 (2006).
Keefe, A. D. & Miller, S. L. Was ferrocyanide a prebiotic reagent? Orig. Life Evol. Biosph. 26, 111–129 (1996).
Gáspár, V. & Beck, M. T. Kinetics of the photoaquation of hexacyanoferrate(II) ion. Polyhedron 2, 387–391 (1983).
Rubin, A. E. Mineralogy of meteorite groups. Meteorit. Planet. Sci. 32, 231–247 (1997).
Pincass, H. Die Bildung von Calciumcyanamid aus Ferrocyancalcium. Chem.-Ztg. 46, 661 (1922).
Seifer, G. B. The thermal decomposition of alkali cyanoferrates(II). Russ. J. Inorg. Chem. 7, 640–643 (1962).
Seifer, G. B. Thermal decomposition of alkaline earth metal and magnesium cyanoferrates(II). Russ. J. Inorg. Chem. 7, 1187–1189 (1962).
Yamanaka, M., Fujita, Y., McLean, A. & Iwase, M. A thermodynamic study of CaCN2 . High Temp. Mater. Processes 19, 275–279 (2000).
Strecker, A. Ueber einen neuen aus Aldehyd-Ammoniak und Blausäure entstehenden Körper. Justus Liebigs Ann. Chem. 91, 349–351 (1854).
Foster, G. W. A. The action of light on potassium ferrocyanide. J. Chem. Soc. 89, 912–920 (1906).
Coderre, F. & Dixon, D. G. Modeling the cyanide heap leaching of cupriferous gold ores. Hydrometallurgy 52, 151–175 (1999).
Hazen, R. M. Paleomineralogy of the Hadean eon: a preliminary species list. Am. J. Sci. 313, 807–843 (2013).
Kurtz, P. Untersuchungen über die Bildung von Nitrilen. Liebigs Ann. Chem. 572, 23–82 (1951).
Buc, S. R., Ford, J. H. & Wise, E. C. An improved synthesis of β-alanine. J. Am. Chem. Soc. 67, 92–94 (1945).
Horváth, O. Photochemistry of copper(I) complexes. Coord. Chem. Rev. 135-136, 303–324 (1994).
Baxendale, J. H. & Westcott, D. T. Kinetics and equilibria in copper(II)–cyanide solutions. J. Chem. Soc. 2347–2351 (1959).
Moureu, C. & Bongrand, J-C. Le cyanoacetylene C3NH. Ann. Chim. (Paris) 14, 47–58 (1920).
Xiang, Y-B., Drenkard, S., Baumann, K., Hickey, D. & Eschenmoser, A. E. Chemie von α-Aminonitrilen. 12. Mitteilung. Sondierungen über thermische Umwandlungen von α-Aminonitrilen. Helv. Chim. Acta 77, 2209–2250 (1994).
Miller, S. L. A production of amino acids under possible primitive Earth conditions. Science 117, 528–529 (1953).
Butlerow, A. Bildung einer zuckerartigen Substanz durch Synthese. Liebigs Ann. Chem. 120, 295–298 (1861).
Oró, J. Synthesis of adenine from ammonium cyanide. Biochem. Biophys. Res. Commun. 2, 407–412 (1960).
Wegner, J., Ceylan, S. & Kirschning, A. Ten key issues in modern flow chemistry. Chem. Commun. 47, 4583–4592 (2011).
de Duve, C. The onset of selection. Nature 433, 581–582 (2005).
Acknowledgements
In memoriam Harry Lonsdale. This work was supported by the Medical Research Council (No. MC_UP_A024_1009), a grant from the Simons Foundation (No. 290362 to J.D.S.) and an award from the Origin of Life Challenge.
Author information
Authors and Affiliations
Contributions
J.D.S. supervised the research and the other authors performed the experiments. All the authors contributed intellectually as the project unfolded. J.D.S. wrote the paper and B.H.P. and C.P. assembled the Supplementary Information, additionally incorporating data from D.J.R. and C.D.D.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 7232 kb)
Rights and permissions
About this article
Cite this article
Patel, B., Percivalle, C., Ritson, D. et al. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nature Chem 7, 301–307 (2015). https://doi.org/10.1038/nchem.2202
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2202
This article is cited by
-
Biogeochemical explanations for the world’s most phosphate-rich lake, an origin-of-life analog
Communications Earth & Environment (2024)
-
To unravel the origin of life, treat findings as pieces of a bigger puzzle
Nature (2024)
-
Thiophosphate photochemistry enables prebiotic access to sugars and terpenoid precursors
Nature Chemistry (2023)
-
From autocatalysis to survival of the fittest in self-reproducing lipid systems
Nature Reviews Chemistry (2023)
-
Detection of HCN and diverse redox chemistry in the plume of Enceladus
Nature Astronomy (2023)