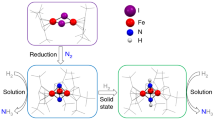
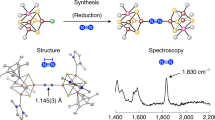
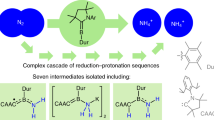
Abstract
The reduction of gaseous nitrogen is a challenge for industrial, biological and synthetic chemists. Major goals include understanding the formation of ammonia for agriculture, and forming N–C and N–Si bonds for the synthesis of fine chemicals. The iron–molybdenum active site of the enzyme nitrogenase has inspired chemists to explore iron and molybdenum complexes in transformations related to N2 reduction. This area of research has gained significant momentum, and the past two years have witnessed a number of significant advances in synthetic Fe–N2 and Mo–N2 chemistry. Furthermore, the identities of all atoms in the iron–molybdenum cofactor of nitrogenase have finally been elucidated, and the discovery of a carbide has generated new questions and targets for coordination chemists. This Perspective summarizes the recent work on iron and molydenum complexes, and highlights the opportunities for continued research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Holland, P. L. in Comprehensive Coordination Chemistry II Vol. 8 (eds McCleverty, J. & Meyer, T. J.) 569–599 (Elsevier, 2004).
MacKay, B. A. & Fryzuk, M. D. Dinitrogen coordination chemistry: on the biomimetic borderlands. Chem. Rev. 104, 385–401 (2004).
Hidai, M. & Mizobe, Y. Recent advances in the chemistry of dinitrogen complexes. Chem. Rev. 95, 1115–1133 (1995).
Hazari, N. Homogeneous iron complexes for the conversion of dinitrogen into ammonia and hydrazine. Chem. Soc. Rev. 39, 4044–4056 (2010).
Crossland, J. L. & Tyler, D. R. Iron-dinitrogen coordination chemistry: dinitrogen activation and reactivity. Coord. Chem. Rev. 254, 1883–1894 (2010).
Chatt, J., Pearman, A. J. & Richards, R. L. Reduction of monocoordinated molecular nitrogen to ammonia in a protic environment. Nature 253, 39–40 (1975).
Chatt, J., Pearman, A. J. & Richards, R. L. Conversion of dinitrogen in its molybdenum and tungsten complexes into ammonia and possible relevance to the nitrogenase reaction. J. Chem. Soc. Dalton Trans. 1852–1860 (1977).
Denisov, N. T., Shuvalov, V. F., Shuvalova, N. I., Shilova, A. K. & Shilov, A. E. Modelling of the biological fixation of nitrogen. Catalytic reduction of nitrogen in protonic media. Dokl. Akad. Nauk SSSR 195, 879–881 (1970).
Yandulov, D. V. & Schrock, R. R. Catalytic reduction of dinitrogen to ammonia at a single molybdenum center. Science 301, 76–78 (2003).
Shiina, K. Reductive silylation of molecular nitrogen via fixation to tris(trialkylsilyl)amine. J. Am. Chem. Soc. 94, 9266–9267 (1972).
Komori, K., Oshita, H., Mizobe, Y. & Hidai, M. Catalytic conversion of molecular nitrogen into silylamines using molybdenum and tungsten dinitrogen complexes. J. Am. Chem. Soc. 111, 1939–1940 (1989).
Tanaka, H. et al. Molybdenum-catalyzed transformation of molecular dinitrogen into silylamine: experimental and DFT study on the remarkable role of ferrocenyldiphosphine ligands. J. Am. Chem. Soc. 133, 3498–3506 (2011).
Arashiba, K. et al. Synthesis and protonation of molybdenum– and tungsten–dinitrogen complexes bearing PNP-type pincer ligands. Organometallics 31, 2035–2041 (2012).
Baumann, J. A. et al. Formation of ammonia and hydrazine from the reactions of acids with bis(dinitrogen) complexes of molybdenum. Inorg. Chem. 24, 3568–3578 (1985).
Arashiba, K., Miyake, Y. & Nishibayashi, Y. A molybdenum complex bearing PNP-type pincer ligands leads to the catalytic reduction of dinitrogen into ammonia. Nature Chem. 3, 120–125 (2011).
Konishita, E. et al. Synthesis and catalytic activity of molybdenum-dinitrogen complexes bearing unsymmetric PNP-type pincer ligands. Organometallics 31, 8437–8443 (2012).
Hebden, T. J., Schrock, R. R., Takase, M. K. & Muller, P. Cleavage of dinitrogen to yield a (t-BuPOCOP)molybdenum nitride. Chem. Commun. 48, 1851–1853 (2012).
Laplaza, C. E. & Cummins, C. C. Dinitrogen cleavage by a three-coordinate molybdenum(III) complex. Science 268, 861–863 (1995).
Curley, J. J., Cozzolino, A. F. & Cummins, C. C. Nitrogen fixation to cyanide at a molybdenum center. Dalton Trans. 40, 2429–2432 (2011).
Peters, J. C. & Mehn, M. P. in Activation of Small Molecules (ed. Tolman, W. B.) 81–119 (VCH, 2006).
Scepaniak, J. J., Young, J. A., Bontchev, R. P. & Smith, J. M. Formation of ammonia from an iron nitrido complex. Angew. Chem. Int. Ed. 48, 3158–3160 (2009).
Scepaniak, J. J., Bontchev, R. P., Johnson, D. L. & Smith, J. M. Snapshots of complete nitrogen atom transfer from an iron(IV) nitrido complex. Angew. Chem. Int. Ed. 50, 6630–6633 (2011).
Scepaniak, J. J. et al. Synthesis, structure, and reactivity of an iron(V) nitride. Science 331, 1049–1052 (2011).
Crossland, J. L., Balesdent, C. G. & Tyler, D. R. Coordination of a complete series of N2 reduction intermediates (N2H2, N2H4, and NH3) to an iron phosphine scaffold. Inorg. Chem. 51, 439–445 (2012).
Field, L. D., Li, H. L. & Magill, A. M. Base-mediated conversion of hydrazine to diazene and dinitrogen at an iron center. Inorg. Chem. 48, 5–7 (2008).
Saouma, C. T., Muller, P. & Peters, J. C. Characterization of structurally unusual diiron NxHy complexes. J. Am. Chem. Soc. 131, 10358–10359 (2009).
Saouma, C. T., Kinney, R. A., Hoffman, B. M. & Peters, J. C. Transformation of an [Fe(η2-N2H3)]+ species to π-delocalized [Fe2(μ-N2H2)]2+/+ complexes. Angew. Chem. Int. Ed. 50, 3446–3449 (2011).
Hoffman, B. M., Dean, D. R. & Seefeldt, L. C. Climbing nitrogenase: toward a mechanism of enzymatic nitrogen fixation. Acc. Chem. Res. 42, 609–619 (2009).
Lee, Y., Mankad, N. P. & Peters, J. C. Triggering N2 uptake via redox-induced expulsion of coordinated NH3 and N2 silylation at trigonal bipyramidal iron. Nature Chem. 2, 558–565 (2010).
Moret, M.-E. & Peters, J. C. Terminal iron dinitrogen and iron imide complexes supported by a tris(phosphino)borane ligand. Angew. Chem. Int. Ed. 50, 2063–2067 (2011).
Moret, M.-E. & Peters, J. C. N2 functionalization at iron metallaboratranes. J. Am. Chem. Soc. 133, 18118–18121 (2011).
Oshita, H., Mizobe, Y. & Hidai, M. Novel disilylation of a dinitrogen ligand. Organometallics 11, 4116–4123 (1992).
Rodriguez, M. M., Bill, E., Brennessel, W. W. & Holland, P. L. N2 Reduction and hydrogenation to ammonia by a molecular iron-potassium complex. Science 334, 780–783 (2011).
Schlögl, R. in Handbook of Heterogeneous Catalysis 2nd Edn, Vol. 5 (eds Ertl, G., Knözinger, H. & Weitkamp, J.) 2501–2575 (Wiley-VCH, 2008).
Mortensen, J. J., Hansen, L. B., Hammer, B. & Norskov, J. K. Nitrogen adsorption and dissociation on Fe(111). J. Catal. 182, 479–488 (1999).
Smith, J. M. et al. Studies of low-coordinate iron dinitrogen complexes. J. Am. Chem. Soc. 128, 756–769 (2006).
Figg, T. M., Holland, P. L. & Cundari, T. R. Cooperativity between low-valent iron and potassium promoters in dinitrogen fixation. Inorg. Chem. 51, 7546–7550 (2012).
Pool, J. A., Lobkovsky, E. & Chirik, P. J. Hydrogenation and cleavage of dinitrogen to ammonia with a zirconium complex. Nature 427, 527–530 (2004).
Askevold, B. et al. Ammonia formation by metal–ligand cooperative hydrogenolysis of a nitrido ligand. Nature Chem. 3, 532–537 (2011).
Yuki, M. et al. Iron-catalysed transformation of molecular dinitrogen into silylamine under ambient conditions. Nature Commun. 3, 1254 (2012).
Einsle, O. et al. Nitrogenase MoFe-protein at 1.16 Å resolution: a central ligand in the FeMo-cofactor. Science 297, 1696–1700 (2002).
Spatzal, T. et al. Evidence for interstitial carbon in nitrogenase FeMo cofactor. Science 334, 940 (2011).
Lancaster, K. M. et al. X-ray emission spectroscopy evidences a central carbon in the nitrogenase iron-molybdenum cofactor. Science 334, 974–977 (2011).
Harris, T. V. & Szilagyi, R. K. Comparative assessment of the composition and charge state of nitrogenase FeMo-cofactor. Inorg. Chem. 50, 4811–4824 (2011).
Wiig, J. A., Lee, C. C., Hu, Y. & Ribbe, M. W. Tracing the interstitial carbide of the nitrogenase cofactor during substrate turnover. J. Am. Chem. Soc. 135, 4982–4983 (2013).
Wiig, J. A., Hu, Y., Lee, C. C. & Ribbe, M. W. Radical SAM-dependent carbon insertion into the nitrogenase M-cluster. Science 337, 1672–1675 (2012).
Anderson, J. S., Moret, M.-E. & Peters, J. C. Conversion of Fe–NH2 to Fe–N2 with release of NH3 . J. Am. Chem. Soc. 135, 534–537 (2013).
Lukoyanov, D. et al. Unification of reaction pathway and kinetic scheme for N2 reduction catalyzed by nitrogenase. Proc. Natl Acad. Sci. USA 109, 5583–5587 (2012).
Hoffman, B. M., Lukoyanov, D., Dean, D. R. & Seefeldt, L. C. Nitrogenase: a draft mechanism. Acc. Chem. Res. 46, 587–595 (2013).
Burgess, B. K. & Lowe, D. J. Mechanism of molybdenum nitrogenase. Chem. Rev. 96, 2983–3011 (1996).
Fryzuk, M. D. Side-on end-on bound dinitrogen: an activated bonding mode that facilitates functionalizing molecular nitrogen. Acc. Chem. Res. 42, 127–133 (2009).
Bernskoetter, W. H., Olmos, A. V., Pool, J. A., Lobkovsky, E. & Chirik, P. J. N-C bond formation promoted by a hafnocene dinitrogen complex: comparison of zirconium and hafnium congeners. J. Am. Chem. Soc. 128, 10696–10697 (2006).
Knobloch, D. J., Toomey, H. E. & Chirik, P. J. Carboxylation of an ansa-zirconocene dinitrogen complex: regiospecific hydrazine synthesis from N2 and CO2 . J. Am. Chem. Soc. 130, 4248–4249 (2008).
Knobloch, D. J. et al. Addition of methyl triflate to a hafnocene dinitrogen complex: stepwise N2 methylation and conversion to a hafnocene hydrazonato compound. J. Am. Chem. Soc. 131, 14903–14912 (2009).
Knobloch, D. J., Lobkovsky, E. & Chirik, P. J. Carbon monoxide-induced dinitrogen cleavage with group 4 metallocenes: reaction scope and coupling to N-H bond formation and CO deoxygenation. J. Am. Chem. Soc. 132, 10553–10564 (2010).
Knobloch, D. J., Lobkovsky, E. & Chirik, P. J. Dinitrogen cleavage and functionalization by carbon monoxide promoted by a hafnium complex. Nature Chem. 2, 30–35 (2010).
Knobloch, D. J., Semproni, S. P., Lobkovsky, E. & Chirik, P. J. Studies into the mechanism of CO-induced N2 cleavage promoted by an ansa-hafnocene complex and C-C bond formation from an observed intermediate. J. Am. Chem. Soc. 134, 3377–3386 (2012).
Semproni, S. P., Milsmann, C. & Chirik, P. J. Structure and reactivity of a hafnocene μ-nitrido prepared from dinitrogen cleavage. Angew. Chem. Int. Ed. 51, 5213–5216 (2012).
Acknowledgements
The authors' research on N2 reduction has been generously supported by the National Institutes of Health (GM065313).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
MacLeod, K., Holland, P. Recent developments in the homogeneous reduction of dinitrogen by molybdenum and iron. Nature Chem 5, 559–565 (2013). https://doi.org/10.1038/nchem.1620
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1620
This article is cited by
-
N2 cleavage by silylene and formation of H2Si(μ-N)2SiH2
Nature Communications (2024)
-
Silicon, Carbon, Germanium, Aluminum Nitride nanocages (Si60, C60, Ge60, Al30N30) as catalysts of N2 reduction to NH3
Silicon (2024)
-
Near ambient N2 fixation on solid electrodes versus enzymes and homogeneous catalysts
Nature Reviews Chemistry (2023)
-
Converting N2 molecules into NH3 with TiO2/Fe3O4 composite covered with a thin water layer under ambient condition
Scientific Reports (2023)
-
High-throughput screening of single-atom catalysts confined in monolayer black phosphorus for efficient nitrogen reduction reaction
Nano Research (2023)