Abstract
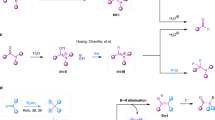
The development of efficient and selective transformations is crucial in synthetic chemistry as it opens new possibilities in the total synthesis of complex molecules. Applying such reactions to the synthesis of ketones is of great importance, as this motif serves as a synthetic handle for the elaboration of numerous organic functionalities. In this context, we report a general and chemoselective method based on an activation/addition sequence on secondary amides allowing the controlled isolation of structurally diverse ketones and ketimines. The generation of a highly electrophilic imidoyl triflate intermediate was found to be pivotal in the observed exceptional functional group tolerance, allowing the facile addition of readily available Grignard and diorganozinc reagents to amides, and avoiding commonly observed over-addition or reduction side reactions. The methodology has been applied to the formal synthesis of analogues of the antineoplastic agent Bexarotene and to the rapid and efficient synthesis of unsymmetrical diketones in a one-pot procedure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Clardy, J. & Walsh, C. Lessons from natural molecules. Nature 432, 829–837 (2004).
Chau, M., Jennewein, S., Walker, K. & Croteau, R. Taxol biosynthesis: molecular cloning and characterization of a cytochrome P450 taxoid 7β-hydroxylase. Chem. Biol. 11, 663–672 (2004).
Hubbard, B. K. & Walsh, C. T. Vancomycin assembly: nature's way. Angew. Chem. Int. Ed. 42, 730–765 (2003).
Schwecke, T. et al. The biosynthetic gene cluster for the polyketide immunosuppressant rapamycin. Proc. Natl Acad. Sci. USA 92, 7839–7843 (1995).
Trost, B. M. Selectivity: a key to synthetic efficiency. Science 219, 245–250 (1983).
Burns, N. Z., Baran, P. S. & Hoffmann, R. W. Redox economy in organic synthesis. Angew. Chem. Int. Ed. 48, 2854–2867 (2009).
Young, I. S. & Baran, P. S. Protecting-group-free synthesis as an opportunity for invention. Nature Chem. 1, 193–205 (2009).
Trost, B. M. & Dong, G. B. Total synthesis of bryostatin 16 using atom-economical and chemoselective approaches. Nature 456, 485–488 (2008).
Afagh, N. A. & Yudin, A. K. Chemoselectivity and the curious reactivity preferences of functional groups. Angew. Chem. Int. Ed. 49, 262–310 (2010).
Huckins, J. R., de Vicente, J. & Rychnovsky, S. D. Synthesis of the C1–C52 fragment of amphidinol 3, featuring a β-alkoxy alkyllithium addition reaction. Org. Lett. 9, 4757–4760 (2007).
Chen, C.-y. et al. Catalytic, enantioselective synthesis of taranabant, a novel, acyclic cannabinoid-1 receptor inverse agonist for the treatment of obesity. Org. Proc. Res. Dev. 11, 616–623 (2007).
Olah, G. A. Friedel–Crafts and Related Reactions Vol. 1, Ch. 11 (Interscience, 1963–1965).
Milstein, D. & Stille, J. K. Mild, selective, general method of ketone synthesis from acyl chlorides and organotin compounds catalyzed by palladium. J. Org. Chem. 44, 1613–1618 (1979).
Brunet, J.-J. & Chauvin, R. Synthesis of diarylketones through carbonylative coupling. Chem. Soc. Rev. 24, 89–95 (1995).
Dieter, R. K. Reaction of acyl chlorides with organometallic reagents: a banquet table of metals for ketone synthesis. Tetrahedron 55, 4177–4236 (1999).
Katritzky, A. R., Le, K. N. B., Khelashvili, L. & Mohapatra, P. P. Alkyl, unsaturated, (hetero)aryl, and N-protected α-amino ketones by acylation of organometallic reagents. J. Org. Chem. 71, 9861–9864 (2006).
Nahm, S. & Weinreb, S. M. N-Methoxy-N-methylamides as effective acylating agents. Tetrahedron Lett. 22, 3815–3818 (1981).
Sibi, M. P. Chemistry of N-methoxy-N-methylamides. Applications in synthesis. A review. Org. Prep. Proced. Int. 25, 15–40 (1993).
Balasubramaniam, S. & Aiden, I. S. The growing synthetic utility of the Weinreb amide. Synthesis, 3707–3738 (2008).
Sengupta, S., Mondal, S. & Das, D. Amino acid derived morpholine amides for nucleophilic α-amino acylation reactions: a new synthetic route to enantiopure α-amino ketones. Tetrahedron Lett. 40, 4107–4110 (1999).
Comins, D. L. & Brown, J. D. Directed lithiation of tertiary β-amino benzamides. J. Org. Chem. 51, 3566–3572 (1986).
Murphy, J. A. et al. Direct conversion of N-methoxy-N-methylamides (Weinreb amides) to ketones via a nonclassical Wittig reaction. Org. Lett. 7, 1427–1429 (2005).
Calosso, M. et al. Enantioselective synthesis of 2,3-disubstituted piperidines. Lett. Org. Chem. 4, 4–6 (2007).
Comins, D. L. The synthetic utility of α-amino alkoxides. Synlett 615–625 (1992).
Wuts, P. G. M. & Greene, T. W. Greene's Protective Groups in Organic Synthesis Ch. 4, Ch. 7 (Wiley, 2007).
Steinig, A. G. & Spero, D. M. Amines via nucleophilic 1,2-addition to ketimines. Construction of nitrogen-substituted quaternary carbon atoms. A review. Org. Prep. Proced. Int. 32, 205–234 (2000).
Reingruber, R. & Bräse, S. 1,2-Addition of trialkylaluminium reagents to N-diphenylphosphinoyl-ketimines in the absence of any additional reagents. Chem. Commun. 105–107 (2008).
Chen, Q., Ilies, L. & Nakamura, E. Cobalt-catalyzed ortho-alkylation of secondary benzamide with alkyl chloride through directed C–H bond activation. J. Am. Chem. Soc. 133, 428–429 (2011).
Yoo, E. J., Ma, S., Mei, T.-S., Chan, K. S. L. & Yu, J. Q. Pd-catalyzed intermolecular C–H amination with alkylamines. J. Am. Chem. Soc. 133, 7652–7655 (2011).
Marcoux, D. & Charette, A. B. Trans-directing ability of amide groups in cyclopropanation: application to the asymmetric cyclopropanation of alkenes with diazo reagents bearing two carboxy groups. Angew. Chem. Int. Ed. 47, 10155–10158 (2008).
Barbe, G. & Charette, A. B. Highly chemoselective metal-free reduction of tertiary amides. J. Am. Chem. Soc. 130, 18–19 (2008).
Pelletier, G., Bechara, W. S. & Charette, A. B. Controlled and chemoselective reduction of secondary amides. J. Am. Chem. Soc. 132, 12817–12819 (2010).
Medley, J. W. & Movassaghi, M. Direct dehydrative N-pyridinylation of amides. J. Org. Chem. 74, 1341–1344 (2009).
Myers, A. G., Tom, N. J., Fraley, M. E., Cohen, S. B. & Madar, D. J. A convergent synthetic route to (+)-dynemicin A and analogs of wide structural variability. J. Am. Chem. Soc. 119, 6072–6094 (1997).
Movassaghi, M. & Hill, M. D. Single-step synthesis of pyrimidine derivatives. J. Am. Chem. Soc. 128, 14254–14255 (2006).
Charette, A. B. & Grenon, M. Spectroscopic studies of the electrophilic activation of amides with triflic anhydride and pyridine. Can. J. Chem. 79, 1694–1703 (2001).
Bartoli, J. et al. New azole antifungals. 2. Synthesis and antifungal activity of heterocyclecarboxamide derivatives of 3-amino-2-aryl-1-azolyl-2-butanol. J. Med. Chem. 41, 1855–1868 (1998).
Jamieson, C., Miller, D. D., Rami, H. K. & Thompson, M. Preparation of piperidinecarboxamide and cyclohexanecarboxamide derivatives as vanilloid receptor modulators. International patent WO2005016915 (24 February 2005).
Krasovskiy, A. & Knochel, P. A LiCl-mediated Br/Mg exchange reaction for the preparation of functionalized aryl- and heteroarylmagnesium compounds from organic bromides. Angew. Chem. Int. Ed. 43, 3333–3336 (2004).
Small, B. L., Brookhart, M. & Bennett, A. M. A. Highly active iron and cobalt catalysts for the polymerization of ethylene. J. Am. Chem. Soc. 120, 4049–4050 (1998).
Charette, A. B. Chiral Amine Synthesis: Methods, Developments and Applications Ch. 1 (Wiley, 2010).
Faul, M. M., Ratz, A. M., Sullivan, K. A., Trankle, W. G. & Winneroski, L. L. Synthesis of novel retinoid X receptor-selective retinoids. J. Org. Chem. 66, 5772–5782 (2001).
Canan Koch, S. S. et al. Synthesis of retinoid X recetor-specific ligands that are potent inducers of adipogenesis in 3T4-LI cells. J. Med. Chem. 42, 742–750 (1999).
Hudlicky, T. & Price, J. D. Anionic approaches to the construction of cyclopentanoids. Chem. Rev. 89, 1467–1486 (1989).
Özkay, Y., Işikdağ, I., İncesu, Z. & Akalin, G. Synthesis of 2-substituted-N-[4-(1-methyl-4,5-diphenyl-1H-imidazole-2-yl) phenyl]acetamide derivatives and evaluation of their anticancer activity. Eur. J. Med. Chem. 45, 3320–3328 (2010).
Prasad, K. R. & Penchalaiah, K. Total synthesis of (–)-anamarine. J. Org. Chem. 76, 6889–6893 (2011).
Sibi, M. P., Sharma, R. & Paulson, K. L. N,N′-Dimethoxy-N,N′-dimethylethanediamide: a useful α-oxo-N-methoxy-N-methylamide and 1,2-diketone synthon. Tetrahedron Lett. 33, 1941–1944 (1992).
Acknowledgements
This work was supported by the Natural Science and Engineering Research Council of Canada (NSERC), the Canada Research Chair Program, the Canada Foundation for Innovation, the FQRNT Centre in Green Chemistry and Catalysis (CGCC) and the Université de Montréal. W.S.B. and G.P. are grateful to NSERC, FQRNT and the Université de Montréal for postgraduate scholarships. The authors would like to thank A. Lemire and P. Lavallée (Université de Montréal), P. Lapointe (IRIC) and M. Grenon (Lundbeck) for supplying starting materials and reagents.
Author information
Authors and Affiliations
Contributions
W.S.B. and G.P. carried out the experimental work, organized the research, analysed data and wrote the manuscript. All the authors contributed to the design of the experiments and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 14101 kb)
Rights and permissions
About this article
Cite this article
Bechara, W., Pelletier, G. & Charette, A. Chemoselective synthesis of ketones and ketimines by addition of organometallic reagents to secondary amides. Nature Chem 4, 228–234 (2012). https://doi.org/10.1038/nchem.1268
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1268
This article is cited by
-
Ketones from aldehydes via alkyl C(sp3)−H functionalization under photoredox cooperative NHC/palladium catalysis
Nature Communications (2023)
-
Direct alkylation of N,N-dialkyl benzamides with methyl sulfides under transition metal-free conditions
Communications Chemistry (2021)
-
Benzylic aroylation of toluenes with unactivated tertiary benzamides promoted by directed ortho-lithiation
Science China Chemistry (2021)
-
Upgrading ketone synthesis direct from carboxylic acids and organohalides
Nature Communications (2020)
-
Chemodivergent transformations of amides using gem-diborylalkanes as pro-nucleophiles
Nature Communications (2020)