Abstract
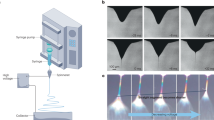
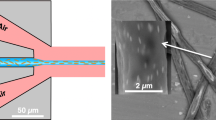
Methods allowing chemical reactions to be carried out on ultra-small scales in a controllable fashion are potentially important for a number of disciplines, including molecular electronics, photonics and molecular biology, and may provide fundamental insight into chemistry in confined spaces. Ultra-small-scale reactions also circumvent potential problems associated with reagent and product toxicity, and reduce energy consumption and waste generation. Here, we report a technique for performing chemical reactions on a zeptomole (10−21 mol) scale. We show that electrospun polymer nanofibres with a diameter of 100–300 nm can be loaded with reactants, and that the junctions formed between crossed nanofibres can function as attolitre-volume reactors. Exposure to heat or solvent vapours fuses the fibres and initiates the reaction. The reaction products can be analysed directly within the nanofibre junctions by fluorescence measurements and mass spectrometry, and solvent extraction of multiple reactors allows product identification by common micromethods such as high-performance liquid chromatography–mass spectrometry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
16 March 2009
In the version of this Article originally published, in paragraph six of the main text, the concentration of reagents in the fibre after evaporation of the solvent was incorrectly given as 0.5 mol l-1, this should have read 0.5 mmol l-1. This has been corrected in the HTML and PDF versions of the Article.
27 March 2009
In the version of this Article originally published, the structure given in Fig. 3a for rhodamine 6G (compound 5) was incorrect. The error has now been corrected in the HTML and PDF versions.
References
Koch, M. V., VandenBussche, K. M. & Chrisman, R. W. (eds) Micro Instrumentation for High Throughput Experimentation and Process Intensification (Wiley-VCH, 2007).
Yoshida, J.-I. Flash Chemistry: Fast Organic Synthesis in Microsystems (Wiley, 2008).
Nie, S., Chiu, D. T. & Zare, R. N. Probing individual molecules with confocal fluorescence microscopy. Science 226, 1018–1021 (1994).
Sauer, M., Hofkens, J. & Enderlein, J. (eds) Handbook of Fluorescence Spectroscopy and Imaging: From Ensemble to Single Molecules (Wiley-VCH, 2009).
Zhang, J., Song, S., Wang, L., Pan, D. & Fan, C. A gold nanoparticle-based chronocoulometric DNA sensor for amplified detection of DNA. Nature Protoc. 2, 2888–2895 (2007).
Yeh, H.-C., Ho, Y.-P., Shih, I.-M. & Wang, T.-H. Homogeneous point mutation detection by quantum dot-mediated two-color fluorescence coincidence analysis. Nucleic Acids Res. 34, e35/1–e35/8 (2006).
Wang, P., Murugaiah, V., Yeung, B., Vouros, P. & Giese, R. W. 2-Phosphoglycolate and glycolate-electrophore detection, including detection of 87 zeptomoles of the latter by gas chromatography–electron-capture mass spectrometry. J. Chromatogr. A 721, 289–296 (1996).
Powell, P. R. & Ewing, A. G. Recent advances in the application of capillary electrophoresis to neuroscience. Anal. Bioanal. Chem. 382, 581–591 (2005).
Lee, J.-Y., Li, H.-W. & Yeung, E. S. Single-molecule spectroscopy for molecular identification in capillary electrophoresis. J. Chromatogr. A 1053, 173–179 (2004).
Page, J. S., Rubakhin, S. S. & Sweedler, J. V. Single-neuron analysis using CE combined with MALDI MS and radionuclide detection. Anal. Chem. 74, 497–503 (2002).
Wirth, T. (ed.) Microreactors in Organic Synthesis and Catalysis (Wiley-VCH, 2008).
Ehrfeld, W., Hessel, V. & Löwe, H. Microreactors: New technology for Modern Chemistry (Wiley-VCH, 2000).
Whitesides, G. M. The origins and the future of microfluidics. Nature 442, 368–373 (2006).
Tabeling, P. Introduction to Microfluidics (Oxford Univ. Press, 2006).
Nguyen, N.-T. & Wereley, S. T. Fundamentals and Applications of Microfluidics 2nd edn (Artech House, 2006).
Velev, O. D., Prevo, B. G. & Bhatt, K. H. On-chip manipulation of free droplets. Nature 426, 515–516 (2003).
Burns, M. A. et al. An integrated nanoliter DNA analysis device. Science 282, 484–487 (1998).
Song, H., Tice, J. D. & Ismagilov, R. F. A microfluidic system for controlling reaction networks in time. Angew. Chem. Int. Ed. 42, 768–772 (2003).
Song, H., Chen, D. L. & Ismagilov, R. F. Reactions in droplets in microfluidic channels. Angew. Chem. Int. Ed. 45, 7336–7356 (2006).
Shestopalov, I., Tice, J. D. & Ismagilov, R. F. Multi-step synthesis of nanoparticles performed on millisecond time scale in a microfluidic droplet-based system. Lab Chip 4, 316–321 (2004).
Zheng, B., Gerdts, C. J. & Ismagilov, R. F. Using nanoliter plugs in microfluidics to facilitate and understand protein crystallization. Curr. Opin. Struct. Biol. 15, 548–555 (2005).
Auroux, P.-A., Koc, Y., deMello, A., Manz, A. & Day, P. J. R. Miniaturised nucleic acid analysis. Lab Chip 4, 534–546 (2004).
Geyer, K., Codee, J. D. C. & Seeberger, P. H. Microreactors as tools for synthetic chemists—the chemists’ round-bottomed flask of the 21st century? Chem. Eur. J. 12, 8434–8442 (2006).
Zheng, B. & Ismagilov, R. F. A microfluidic approach for screening submicroliter volumes against multiple reagents by using preformed arrays of nanoliter plugs in a three-phase liquid/liquid/gas flow. Angew. Chem. Int. Ed. 44, 2520–2523 (2005).
Vriezema, D. M. et al. Self-assembled nanoreactors. Chem. Rev. 105, 1445–1489 (2005).
Tretyakov, Y. D., Lukashin, A. V. & Eliseev, A. A. Synthesis of functional nanocomposites based on solid-phase nanoreactors. Russ. Chem. Rev. 73, 899–921 (2004).
Fan, J. et al. Mesoporous silica nanoreactors for highly efficient proteolysis. Chem. Eur. J. 11, 5391–5396 (2005).
Kim, K. Mechanically interlocked molecules incorporating cucurbituril and their supramolecular assemblies. Chem. Soc. Rev. 31, 96–107 (2002).
Wulff, G. Enzyme-like catalysis by molecularly imprinted polymers. Chem. Rev. 102, 1–28 (2002).
Fiedler, D., Leung, D. H., Bergman, R. G. & Raymond, K. N. Selective molecular recognition, C–H bond activation, and catalysis in nanoscale reaction vessels. Acc. Chem. Res. 38, 351–360 (2005).
Mackay, L. G., Wylie, R. S. & Sanders, J. K. M. Catalytic acyl transfer by a cyclic porphyrin trimer: Efficient turnover without product inhibition. J. Am. Chem. Soc. 116, 3141–3142 (1994).
Breslow, R. & Schmuck, C. Goodness of fit in complexes between substrates and ribonuclease mimics: Effects on binding, catalytic rate constants, and regiochemistry. J. Am. Chem. Soc. 118, 6601–6605 (1996).
Reneker, D. H. & Chun, I. Nanometer diameter fibres of polymer, produced by electrospinning. Nanotechnology 7, 216–223 (1996).
Li, D., Wang, Y. & Xia, Y. Electrospinning nanofibres as uniaxially aligned arrays and layer-by-layer stacked films. Adv. Mater. 16, 361–366 (2004).
Li, D. & Xia, Y. Electrospinning of nanofibres: Reinventing the wheel? Adv. Mater. 16, 1151–1170 (2004).
Jang, S.-Y. et al. Welded electrochromic conductive polymer nanofibres by electrostatic spinning. Adv. Mater. 17, 2177–2180 (2005).
Prodi, L. et al. Dansylated polyamines as fluorescent sensors for metal ions: Photophysical properties and stability of copper(ii) complexes in solution. Helv. Chim. Acta 84, 690–706 (2001).
Lakowicz J. R. Principles of Fluorescence Spectroscopy 3rd edn (Springer, 2006).
Zollinger, H. Diazo Chemistry I: Aromatic and Heteroaromatic Compounds (VCH, 1994).
Szele, I. & Zollinger, H. Azo coupling reactions: Structures and mechanism. Top. Curr. Chem. 112, 1–66 (1983).
Kimball, D. B. & Haley, M. M. Triazenes: A versatile tool in organic synthesis. Angew. Chem. Int. Ed. 41, 3338–3351 (2003).
Markley, J. L. NMR analysis goes nano. Nat. Biotechnol. 25, 750–751 (2007).
Park, H. B. et al. Polymers with cavities tuned for fast selective transport of small molecules and ions. Science 318, 254–258 (2007).
Lomax, G. R. Breathable polyurethane membranes for textile and related industries. J. Mater. Chem. 17, 2775–2784 (2007).
Duda, J. L. & Zielinski, J. M. Free-volume theory. Plastics Eng. 32, 143–171 (1996).
Meares, P. The influence of penetrant concentration on the diffusion and permeation of small molecules in polymers above glass temperature. Eur. Polym. J. 29, 237–243 (1993).
Wustholz, K. L., Sluss, D. R. B., Kahr, B. & Reid, P. J. Applications of single-molecule microscopy to problems in dyed composite materials. Int. Rev. Phys. Chem. 27, 167–200 (2008).
Bellan, L. M., Cross, J. D., Strychalski, E. A., Moran-Mirabal, J. & Craighead, H. G. Individually resolved DNA molecules stretched and embedded in electrospun polymer nanofibers. Nano Lett. 6, 2526–2530 (2006).
Acknowledgements
Financial support from the NSF (CHE No. 0750303, EXP-LA No. 0731153 to P.A.) and Bowling Green State University is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
P.A. and M.A.P. contributed equally to this work.
Corresponding author
Supplementary information
Supplementary information
Supplementary information (PDF 0 kb)
Rights and permissions
About this article
Cite this article
Anzenbacher, P., Palacios, M. Polymer nanofibre junctions of attolitre volume serve as zeptomole-scale chemical reactors. Nature Chem 1, 80–86 (2009). https://doi.org/10.1038/nchem.125
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.125
This article is cited by
-
Microfluidic-spinning construction of black-phosphorus-hybrid microfibres for non-woven fabrics toward a high energy density flexible supercapacitor
Nature Communications (2018)
-
Chinese brushes: From controllable liquid manipulation to template-free printing microlines
Nano Research (2015)
-
Microreactors for peptide synthesis: looking through the eyes of twenty first century !!!
Amino Acids (2014)
-
Nanowire liquid pumps
Nature Nanotechnology (2013)
-
Discreteness-induced concentration inversion in mesoscopic chemical systems
Nature Communications (2012)