Abstract
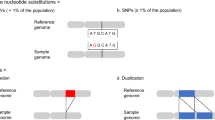
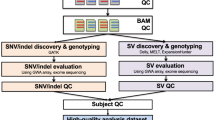
Recent schizophrenia (SCZ) studies have reported an increased burden of de novo copy number variants (CNVs) and identified specific high-risk CNVs, although with variable phenotype expressivity. However, the pathogenesis of SCZ has not been fully elucidated. Using array comparative genomic hybridization, we performed a high-resolution genome-wide CNV analysis on a mainly (92%) Japanese population (1699 SCZ cases and 824 controls) and identified 7066 rare CNVs, 70.0% of which were small (<100 kb). Clinically significant CNVs were significantly more frequent in cases than in controls (odds ratio=3.04, P=9.3 × 10−9, 9.0% of cases). We confirmed a significant association of X-chromosome aneuploidies with SCZ and identified 11 de novo CNVs (e.g., MBD5 deletion) in cases. In patients with clinically significant CNVs, 41.7% had a history of congenital/developmental phenotypes, and the rate of treatment resistance was significantly higher (odds ratio=2.79, P=0.0036). We found more severe clinical manifestations in patients with two clinically significant CNVs. Gene set analysis replicated previous findings (e.g., synapse, calcium signaling) and identified novel biological pathways including oxidative stress response, genomic integrity, kinase and small GTPase signaling. Furthermore, involvement of multiple SCZ candidate genes and biological pathways in the pathogenesis of SCZ was suggested in established SCZ-associated CNV loci. Our study shows the high genetic heterogeneity of SCZ and its clinical features and raises the possibility that genomic instability is involved in its pathogenesis, which may be related to the increased burden of de novo CNVs and variable expressivity of CNVs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Xu B, Roos JL, Levy S, van Rensburg EJ, Gogos JA, Karayiorgou M . Strong association of de novo copy number mutations with sporadic schizophrenia. Nat Genet 2008; 40: 880–885.
Malhotra D, McCarthy S, Michaelson JJ, Vacic V, Burdick KE, Yoon S et al. High frequencies of de novo CNVs in bipolar disorder and schizophrenia. Neuron 2011; 72: 951–963.
Kirov G, Pocklington AJ, Holmans P, Ivanov D, Ikeda M, Ruderfer D et al. De novo CNV analysis implicates specific abnormalities of postsynaptic signalling complexes in the pathogenesis of schizophrenia. Mol Psychiatry 2012; 17: 142–153.
Walsh T, McClellan JM, McCarthy SE, Addington AM, Pierce SB, Cooper GM et al. Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science 2008; 320: 539–543.
Szatkiewicz JP, O'Dushlaine C, Chen G, Chambert K, Moran JL, Neale BM et al. Copy number variation in schizophrenia in Sweden. Mol Psychiatry 2014; 19: 762–773.
International Schizophrenia Consortium. Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 2008; 455: 237–241.
Levinson DF, Duan J, Oh S, Wang K, Sanders AR, Shi J et al. Copy number variants in schizophrenia: confirmation of five previous findings and new evidence for 3q29 microdeletions and VIPR2 duplications. Am J Psychiatry 2011; 168: 302–316.
Rees E, Walters JT, Georgieva L, Isles AR, Chambert KD, Richards AL et al. Analysis of copy number variations at 15 schizophrenia-associated loci. Br J Psychiatry 2014; 204: 108–114.
Sullivan PF, Daly MJ, O'Donovan M . Genetic architectures of psychiatric disorders: the emerging picture and its implications. Nat Rev Genet 2012; 13: 537–551.
Kirov G, Rees E, Walters JT, Escott-Price V, Georgieva L, Richards AL et al. The penetrance of copy number variations for schizophrenia and developmental delay. Biol Psychiatry 2014; 75: 378–385.
Moreno-De-Luca A, Myers SM, Challman TD, Moreno-De-Luca D, Evans DW, Ledbetter DH . Developmental brain dysfunction: revival and expansion of old concepts based on new genetic evidence. Lancet Neurol 2013; 12: 406–414.
Pocklington AJ, Rees E, Walters JT, Han J, Kavanagh DH, Chambert KD et al. Novel findings from CNVs implicate inhibitory and excitatory signaling complexes in schizophrenia. Neuron 2015; 86: 1203–1214.
Purcell SM, Moran JL, Fromer M, Ruderfer D, Solovieff N, Roussos P et al. A polygenic burden of rare disruptive mutations in schizophrenia. Nature 2014; 506: 185–190.
Fromer M, Pocklington AJ, Kavanagh DH, Williams HJ, Dwyer S, Gormley P et al. De novo mutations in schizophrenia implicate synaptic networks. Nature 2014; 506: 179–184.
Ikeda M, Aleksic B, Kirov G, Kinoshita Y, Yamanouchi Y, Kitajima T et al. Copy number variation in schizophrenia in the Japanese population. Biol Psychiatry 2010; 67: 283–286.
Conrad DF, Pinto D, Redon R, Feuk L, Gokcumen O, Zhang YJ et al. Origins and functional impact of copy number variation in the human genome. Nature 2010; 464: 704–712.
DeLisi LE, Friedrich U, Wahlstrom J, Boccio-Smith A, Forsman A, Eklund K et al. Schizophrenia and sex chromosome anomalies. Schizophr Bull 1994; 20: 495–505.
Piton A, Gauthier J, Hamdan FF, Lafreniere RG, Yang Y, Henrion E et al. Systematic resequencing of X-chromosome synaptic genes in autism spectrum disorder and schizophrenia. Mol Psychiatry 2011; 16: 867–880.
Giglia-Mari G, Zotter A, Vermeulen W . DNA damage response. Cold Spring Harb Perspect Biol 2011; 3: a000745.
Catts VS, Catts SV, Jablensky A, Chandler D, Weickert CS, Lavin MF . Evidence of aberrant DNA damage response signalling but normal rates of DNA repair in dividing lymphoblasts from patients with schizophrenia. World J Biol Psychiatry 2012; 13: 114–125.
Stoll G, Pietilainen OP, Linder B, Suvisaari J, Brosi C, Hennah W et al. Deletion of TOP3beta, a component of FMRP-containing mRNPs, contributes to neurodevelopmental disorders. Nat Neurosci 2013; 16: 1228–1237.
Ionita-Laza I, Xu B, Makarov V, Buxbaum JD, Roos JL, Gogos JA et al. Scan statistic-based analysis of exome sequencing data identifies FAN1 at 15q13.3 as a susceptibility gene for schizophrenia and autism. Proc Natl Acad Sci USA 2014; 111: 343–348.
Goto M . Hierarchical deterioration of body systems in Werner's syndrome: implications for normal ageing. Mech Ageing Dev 1997; 98: 239–254.
Lepretre F, Villenet C, Quief S, Nibourel O, Jacquemin C, Troussard X et al. Waved aCGH: to smooth or not to smooth. Nucleic Acids Res 2010; 38: e94.
Vermeesch JR, Brady PD, Sanlaville D, Kok K, Hastings RJ . Genome-wide arrays: quality criteria and platforms to be used in routine diagnostics. Hum Mutat 2012; 33: 906–915.
Johansson MM, Van Geystelen A, Larmuseau MHD, Djurovic S, Andreassen OA, Agartz I et al. Microarray analysis of copy number variants on the human Y chromosome reveals novel and frequent duplications overrepresented in specific haplogroups. PLoS One 2015; 10: e0137223.
Skol AD, Scott LJ, Abecasis GR, Boehnke M . Joint analysis is more efficient than replication-based analysis for two-stage genome-wide association studies. Nat Genet 2006; 38: 209–213.
Kearney HM, Thorland EC, Brown KK, Quintero-Rivera F, South ST . American College of Medical Genetics standards and guidelines for interpretation and reporting of postnatal constitutional copy number variants. Genet Med 2011; 13: 680–685.
Cox DR . Continuity correction. Biometrika 1970; 57: 217–219.
Raychaudhuri S, Korn JM, McCarroll SA, Altshuler D, Sklar P, Purcell S et al. Accurately assessing the risk of schizophrenia conferred by rare copy-number variation affecting genes with brain function. PLoS Genet 2010; 6: e1001097.
Darnell JC, Van Driesche SJ, Zhang C, Hung KY, Mele A, Fraser CE et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 2011; 146: 247–261.
Schizophrenia Psychiatric Genome-Wide Association Study Consortium. Genome-wide association study identifies five new schizophrenia loci. Nat Genet 2011; 43: 969–976.
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM et al. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet 2000; 25: 25–29.
Powell JA . GO2MSIG, an automated GO based multi-species gene set generator for gene set enrichment analysis. BMC Bioinform 2014; 15: 146.
Ramanan VK, Shen L, Moore JH, Saykin AJ . Pathway analysis of genomic data: concepts, methods, and prospects for future development. Trends Genet 2012; 28: 323–332.
Holm S . A simple sequentially rejective multiple test procedure. Scand J Stat 1979; 6: 65–70.
Storey JD, Tibshirani R . Statistical significance for genomewide studies. Proc Natl Acad Sci USA 2003; 100: 9440–9445.
Wang K, Zhang H, Bloss CS, Duvvuri V, Kaye W, Schork NJ et al. A genome-wide association study on common SNPs and rare CNVs in anorexia nervosa. Mol Psychiatry 2011; 16: 949–959.
Pinto D, Darvishi K, Shi X, Rajan D, Rigler D, Fitzgerald T et al. Comprehensive assessment of array-based platforms and calling algorithms for detection of copy number variants. Nat Biotechnol 2011; 29: 512–520.
Newman S, Hermetz KE, Weckselblatt B, Rudd MK . Next-generation sequencing of duplication CNVs reveals that most are tandem and some create fusion genes at breakpoints. Am J Hum Genet 2015; 96: 208–220.
Kunugi H, Lee KB, Nanko S . Cytogenetic findings in 250 schizophrenics: evidence confirming an excess of the X chromosome aneuploidies and pericentric inversion of chromosome 9. Schizophr Res 1999; 40: 43–47.
Williams SR, Mullegama SV, Rosenfeld JA, Dagli AI, Hatchwell E, Allen WP et al. Haploinsufficiency of MBD5 associated with a syndrome involving microcephaly, intellectual disabilities, severe speech impairment, and seizures. Eur J Hum Genet 2010; 18: 436–441.
Hodge JC, Mitchell E, Pillalamarri V, Toler TL, Bartel F, Kearney HM et al. Disruption of MBD5 contributes to a spectrum of psychopathology and neurodevelopmental abnormalities. Mol Psychiatry 2014; 19: 368–379.
Talkowski ME, Mullegama SV, Rosenfeld JA, van Bon WM, Shen YP, Repnikova EA et al. Assessment of 2q23.1 microdeletion syndrome implicates MBD5 as a single causal locus of intellectual disability, epilepsy, and autism spectrum disorder. Am J Hum Genet 2011; 89: 551–563.
Bassett AS, Hodgkinson K, Chow EW, Correia S, Scutt LE, Weksberg R . 22q11 deletion syndrome in adults with schizophrenia. Am J Med Genet 1998; 81: 328–337.
Vu TH, Coccaro EF, Eichler EE, Girirajan S . Genomic architecture of aggression: rare copy number variants in intermittent explosive disorder. Am J Med Genet B 2011; 156B: 808–816.
Schennach R, Riedel M, Musil R, Moller HJ . Treatment response in first-episode schizophrenia. Clin Psychopharmacol Neurosci 2012; 10: 78–87.
Lin AS, Chang SS, Lin SH, Peng YC, Hwu HG, Chen WJ . Minor physical anomalies and craniofacial measures in patients with treatment-resistant schizophrenia. Psychol Med 2015; 45: 1839–1850.
Girirajan S, Rosenfeld JA, Cooper GM, Antonacci F, Siswara P, Itsara A et al. A recurrent 16p12.1 microdeletion supports a two-hit model for severe developmental delay. Nat Genet 2010; 42: 203–209.
Girirajan S, Rosenfeld JA, Coe BP, Parikh S, Friedman N, Goldstein A et al. Phenotypic heterogeneity of genomic disorders and rare copy-number variants. N Engl J Med 2012; 367: 1321–1331.
Fishel ML, Vasko MR, Kelley MR . DNA repair in neurons: so if they don't divide what's to repair? Mutat Res 2007; 614: 24–36.
Flatow J, Buckley P, Miller BJ . Meta-analysis of oxidative stress in schizophrenia. Biol Psychiatry 2013; 74: 400–409.
Brennand K, Savas JN, Kim Y, Tran N, Simone A, Hashimoto-Torii K et al. Phenotypic differences in hiPSC NPCs derived from patients with schizophrenia. Mol Psychiatry 2015; 20: 361–368.
Cabungcal JH, Counotte DS, Lewis EM, Tejeda HA, Piantadosi P, Pollock C et al. Juvenile antioxidant treatment prevents adult deficits in a developmental model of schizophrenia. Neuron 2014; 83: 1073–1084.
Yao JK, Leonard S, Reddy RD . Increased nitric oxide radicals in postmortem brain from patients with schizophrenia. Schizophr Bull 2004; 30: 923–934.
Gawryluk JW, Wang JF, Andreazza AC, Shao L, Young LT . Decreased levels of glutathione, the major brain antioxidant, in post-mortem prefrontal cortex from patients with psychiatric disorders. Int J Neuropsychopharmacol 2011; 14: 123–130.
McKinnon PJ . Maintaining genome stability in the nervous system. Nat Neurosci 2013; 16: 1523–1529.
Hastings PJ, Lupski JR, Rosenberg SM, Ira G . Mechanisms of change in gene copy number. Nat Rev Genet 2009; 10: 551–564.
Arlt MF, Wilson TE, Glover TW . Replication stress and mechanisms of CNV formation. Curr Opin Genet Dev 2012; 22: 204–210.
Weckselblatt B, Rudd MK . Human structural variation: mechanisms of chromosome rearrangements. Trends Genet 2015; 31: 587–599.
Amadio M, Battaini F, Pascale A . The different facets of protein kinases C: old and new players in neuronal signal transduction pathways. Pharmacol Res 2006; 54: 317–325.
Yamauchi T . Neuronal Ca2+/calmodulin-dependent protein kinase II—discovery, progress in a quarter of a century, and perspective: implication for learning and memory. Biol Pharmaceut Bull 2005; 28: 1342–1354.
Lang B, Pu J, Hunter I, Liu M, Martin-Granados C, Reilly TJ et al. Recurrent deletions of ULK4 in schizophrenia: a gene crucial for neuritogenesis and neuronal motility. J Cell Sci 2014; 127: 630–640.
McGuire JL, Hammond JH, Yates SD, Chen D, Haroutunian V, Meador-Woodruff JH et al. Altered serine/threonine kinase activity in schizophrenia. Brain Res 2014; 1568: 42–54.
Costain G, Lionel AC, Merico D, Forsythe P, Russell K, Lowther C et al. Pathogenic rare copy number variants in community-based schizophrenia suggest a potential role for clinical microarrays. Hum Mol Genet 2013; 22: 4485–4501.
Fogel BL, Wexler E, Wahnich A, Friedrich T, Vijayendran C, Gao FY et al. RBFOX1 regulates both splicing and transcriptional networks in human neuronal development. Hum Mol Genet 2012; 21: 4171–4186.
Ye X, Carew TJ . Small G protein signaling in neuronal plasticity and memory formation: the specific role of ras family proteins. Neuron 2010; 68: 340–361.
Harvard C, Strong E, Mercier E, Colnaghi R, Alcantara D, Chow E et al. Understanding the impact of 1q21.1 copy number variant. Orphanet J Rare Dis 2011; 6: 54.
Colnaghi R, Carpenter G, Volker M, O'Driscoll M . The consequences of structural genomic alterations in humans: genomic disorders, genomic instability and cancer. Semin Cell Dev Biol 2011; 22: 875–885.
Bundo M, Toyoshima M, Okada Y, Akamatsu W, Ueda J, Nemoto-Miyauchi T et al. Increased l1 retrotransposition in the neuronal genome in schizophrenia. Neuron 2014; 81: 306–313.
Grice SJ, Liu JL, Webber C . Synergistic interactions between Drosophila orthologues of genes spanned by de novo human CNVs support multiple-hit models of autism. PLoS Genet 2015; 11: e1004998.
Noh HJ, Ponting CP, Boulding HC, Meader S, Betancur C, Buxbaum JD et al. Network topologies and convergent aetiologies arising from deletions and duplications observed in individuals with autism. PLoS Genet 2013; 9: e1003523.
Gottlieb B, Beitel LK, Trifiro MA . Somatic mosaicism and variable expressivity. Trends Genet 2001; 17: 79–82.
Biesecker LG, Spinner NB . A genomic view of mosaicism and human disease. Nat Rev Genet 2013; 14: 307–320.
McConnell MJ, Lindberg MR, Brennand KJ, Piper JC, Voet T, Cowing-Zitron C et al. Mosaic copy number variation in human neurons. Science 2013; 342: 632–637.
Cai X, Evrony GD, Lehmann HS, Elhosary PC, Mehta BK, Poduri A et al. Single-cell, genome-wide sequencing identifies clonal somatic copy-number variation in the human brain. Cell Rep 2014; 8: 1280–1289.
Wei PC, Chang AN, Kao J, Du Z, Meyers RM, Alt FW et al. Long neural genes harbor recurrent DNA break clusters in neural stem/progenitor cells. Cell 2016; 164: 644–655.
Marchini J, Cardon LR, Phillips MS, Donnelly P . The effects of human population structure on large genetic association studies. Nat Genet 2004; 36: 512–517.
Acknowledgements
We thank the patients and their families for participating in this study. This research was supported by research grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Ministry of Health, Labor and Welfare of Japan; 'Integrated research on neuropsychiatric disorders' carried out under the Strategic Research Program for Brain Sciences from the Japan Agency for Medical Research and Development, AMED; the Brain Mapping by Integrated Neurotechnologies for Disease Studies (Brain/MINDS) from AMED; Innovative Areas 'Glial assembly: a new regulatory machinery of brain function and disorders'; Innovative Areas 'Comprehensive Brain Science Network'; Research Group For Schizophrenia; SENSHIN Medical Research Foundation; and The Uehara Memorial Foundation. The ClinVar accession numbers for the clinically significant CNVs identified in patients are SCV000263416–SCV000263544.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Kushima, I., Aleksic, B., Nakatochi, M. et al. High-resolution copy number variation analysis of schizophrenia in Japan. Mol Psychiatry 22, 430–440 (2017). https://doi.org/10.1038/mp.2016.88
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2016.88
This article is cited by
-
Cohen syndrome combined with psychiatric symptoms: a case report
BMC Psychiatry (2024)
-
Pleiotropic contribution of rbfox1 to psychiatric and neurodevelopmental phenotypes in two zebrafish models
Translational Psychiatry (2024)
-
The genetic architecture of schizophrenia: review of large-scale genetic studies
Journal of Human Genetics (2023)
-
Copy Number Variations and Schizophrenia
Molecular Neurobiology (2023)
-
Mice carrying a schizophrenia-associated mutation of the Arhgap10 gene are vulnerable to the effects of methamphetamine treatment on cognitive function: association with morphological abnormalities in striatal neurons
Molecular Brain (2021)