Abstract
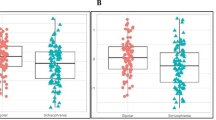
Biomarkers are now used in many areas of medicine but are still lacking for psychiatric conditions such as schizophrenia (SCZ). We have used a multiplex molecular profiling approach to measure serum concentrations of 181 proteins and small molecules in 250 first and recent onset SCZ, 35 major depressive disorder (MDD), 32 euthymic bipolar disorder (BPD), 45 Asperger syndrome and 280 control subjects. Preliminary analysis resulted in identification of a signature comprised of 34 analytes in a cohort of closely matched SCZ (n=71) and control (n=59) subjects. Partial least squares discriminant analysis using this signature gave a separation of 60–75% of SCZ subjects from controls across five independent cohorts. The same analysis also gave a separation of ∼50% of MDD patients and 10–20% of BPD and Asperger syndrome subjects from controls. These results demonstrate for the first time that a biological signature for SCZ can be identified in blood serum. This study lays the groundwork for development of a diagnostic test that can be used as an aid for distinguishing SCZ subjects from healthy controls and from those affected by related psychiatric illnesses with overlapping symptoms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others

References
Saha S, Chant D, Welham J, McGrath J . A systematic review of the prevalence of schizophrenia. PLoS Med 2005; 2: e141.
National Institute of Mental Health. (http://www.nimh.nih.gov/), last updated 15 February 2008.
First MB, Spitzer RL, Gibbon M, Janet BW . Structured Clinical Interview for DSM-IV Axis I Disorders, Clinician Version (SCID-CV). American Psychiatric Press Inc: Washington, DC, 1996.
Csernansky JG, Schuchart EK . Relapse and rehospitalisation rates in patients with schizophrenia: effects of second generation antipsychotics. CNS Drugs 2002; 16: 473–484.
Moller HJ . Course and long-term treatment of schizophrenic psychoses. Pharmacopsychiatry 2004; 37 (Suppl 2): 126–135.
Schwarz E, Izmailov R, Spain M, Barnes A, Mapes JP, Guest PC et al. Validation of a blood-based laboratory test to aid in the confirmation of a diagnosis of schizophrenia. Biomark Insights 2010; 5: 39–47.
Amur S, Frueh FW, Lesko LJ, Huang S-M . Integration and use of biomarkers in drug development, regulation and clinical practice: a US regulatory perspective. Biomakers Med 2008; 2: 305–311.
Eskandari F, Webster JI, Sternberg EM . Neural immune pathways and their connection to inflammatory diseases. Arthritis Res Ther 2003; 5: 251–265.
Harris LW, Wayland M, Lan M, Ryan M, Giger T, Lockstone H et al. The cerebral microvasculature in schizophrenia: a laser capture microdissection study. PLoS One 2008; 3: e3964.
Muller N, Schwarz MJ . Neuroimmune-endocrine crosstalk in schizophrenia and mood disorders. Expert Rev Neurother 2006; 6: 1017–1038.
Bertenshaw GP, Yip P, Seshaiah P, Zhao J, Chen TH, Wiggins WS et al. Multianalyte profiling of serum antigens and autoimmune and infectious disease molecules to identify biomarkers dysregulated in epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev 2008; 17: 2872–2881.
Gurbel PA, Kreutz RP, Bliden KP, DiChiara J, Tantry US . Biomarker analysis by fluorokine multianalyte profiling distinguishes patients requiring intervention from patients with long-term quiescent coronary artery disease: a potential approach to identify atherosclerotic disease progression. Am Heart J 2008; 155: 56–61.
Escobar GP, Lindsey ML . Multi-Analyte Profiling of Post-Myocardial Infarction Plasma Samples. FASEB J 2007; 21 (746.11). [Abstract].
Delaleu N, Immervoll H, Cornelius J, Jonsson R . Biomarker profiles in serum and saliva of experimental Sjogren's syndrome: associations with specific autoimmune manifestations. Arthritis Res Ther 2008; 10: R22.
Kay SR, Fiszbein A, Opler LA . The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 1987; 13: 261–276.
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM et al. Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Standards for Reporting of Diagnostic Accuracy. Clin Chem 2003; 49: 1–6.
Benjamini Y, Hochberg Y . Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Statist Soc Ser B 1995; 57: 289–300.
Seaton BE, Goldstein G, Allen DN . Sources of heterogeneity in schizophrenia: the role of neuropsychological functioning. Neuropsychol Rev 2001; 11: 45–67.
Ferrier IN, Stanton BR, Kelly TP, Scott J . Neuropsychological function in euthymic patients with bipolar disorder. Br J Psychiatry 1999; 175: 246–251.
Raja M, Azzoni A . Asperger's disorder in the emergency psychiatric setting. Gen Hosp Psychiatry 2001; 23: 285–293.
Fleischhacker W . Negative symptoms in patients with schizophrenia with special reference to the primary versus secondary distinction. Encephale 2000; 26 Spec No 1: 12–14.
Domenici E, Wille DR, Tozzi F, Prokopenko I, Miller S, McKeown A et al. Plasma protein biomarkers for depression and schizophrenia by multi analyte profiling of case-control collections. PLoS One 2010; 5: e9166.
Fessel WJ, Solomon GF . Psychosis and systemic lupus erythematosus: a review of the literature and case reports. Calif Med 1960; 92: 266–270.
Goldberg RB . Cytokine and cytokine-like inflammation markers, endothelial dysfunction, and imbalanced coagulation in development of diabetes and its complications. J Clin Endocrinol Metab 2009; 94: 3171–3182.
Volp AC, Alfenas Rde C, Costa NM, Minim VP, Stringueta PC, Bressan J . Inflammation biomarkers capacity in predicting the metabolic syndrome]. Arq Bras Endocrinol Metabol 2008; 52: 537–549.
Muller N, Riedel M, Schwarz MJ, Engel RR . Clinical effects of COX-2 inhibitors on cognition in schizophrenia. Eur Arch Psychiatry Clin Neurosci 2005; 255: 149–151.
Guest P, Wang L, Harris L, Burling K, Levin Y, Ernst A et al. Increased levels of circulating insulin-related peptides in first onset, antipsychotic naïve schizophrenia patients. Mol Psychiatry 2010; 15: 118–119.
Sato T, Hanyu H, Hirao K, Kanetaka H, Sakurai H, Iwamoto T . Efficacy of PPAR-gamma agonist pioglitazone in mild Alzheimer disease. Neurobiol Aging; 16 November 2009 (e-pub ahead of print).
Baptista T, Rangel N, Fernandez V, Carrizo E, El Fakih Y, Uzcategui E et al. Metformin as an adjunctive treatment to control body weight and metabolic dysfunction during olanzapine administration: a multicentric, double-blind, placebo-controlled trial. Schizophr Res 2007; 93: 99–108.
Henderson DC, Fan X, Sharma B, Copeland PM, Borba CP, Boxill R et al. A double-blind, placebo-controlled trial of rosiglitazone for clozapine-induced glucose metabolism impairment in patients with schizophrenia. Acta Psychiatr Scand 2009; 119: 457–465.
Rybakowski JK, Wykretowicz A, Heymann-Szlachcinska A, Wysocki H . Impairment of endothelial function in unipolar and bipolar depression. Biol Psychiatry 2006; 60: 889–891.
Santos LL, Morand EF . Macrophage migration inhibitory factor: a key cytokine in RA, SLE and atherosclerosis. Clin Chim Acta 2009; 399: 1–7.
Burger-Kentischer A, Goebel H, Seiler R, Fraedrich G, Schaefer HE, Dimmeler S et al. Expression of macrophage migration inhibitory factor in different stages of human atherosclerosis. Circulation 2002; 105: 1561–1566.
Bruchfeld A, Carrero JJ, Qureshi AR, Lindholm B, Barany P, Heimburger O et al. Elevated serum macrophage migration inhibitory factor (MIF) concentrations in chronic kidney disease (CKD) are associated with markers of oxidative stress and endothelial activation. Mol Med 2009; 15: 70–75.
Chung CP, Avalos I, Oeser A, Gebretsadik T, Shintani A, Raggi P et al. High prevalence of the metabolic syndrome in patients with systemic lupus erythematosus: association with disease characteristics and cardiovascular risk factors. Ann Rheum Dis 2007; 66: 208–214.
Wajed J, Ahmad Y, Durrington PN, Bruce IN . Prevention of cardiovascular disease in systemic lupus erythematosus--proposed guidelines for risk factor management. Rheumatology (Oxford) 2004; 43: 7–12.
De Hert M, van Winkel R, Van Eyck D, Hanssens L, Wampers M, Scheen A et al. Prevalence of diabetes, metabolic syndrome and metabolic abnormalities in schizophrenia over the course of the illness: a cross-sectional study. Clin Pract Epidemol Ment Health 2006; 2: 14.
Shoelson SE, Lee J, Goldfine AB . Inflammation and insulin resistance. J Clin Invest 2006; 116: 1793–1801.
Steiner J, Walter M, Guest P, Myint AM, Schiltz K, Panteli B et al. Elevated S100B levels in schizophrenia are associated with insulin resistance. Mol Psychiatry 2010; 15: 3–4.
Prabakaran S, Swatton JE, Ryan MM, Huffaker SJ, Huang JT, Griffin JL et al. Mitochondrial dysfunction in schizophrenia: evidence for compromised brain metabolism and oxidative stress. Mol Psychiatry 2004; 9: 684–697, 643.
Uranova NA, Zimina IS, Vikhreva OV, Krukov NO, Rachmanova VI, Orlovskaya DD . Ultrastructural damage of capillaries in the neocortex in schizophrenia. World J Biol Psychiatry 2010; 11: 567–578.
Rybakowski J, Weterle R . Niacin test in schizophrenia and affective illness. Biol Psychiatry 1991; 29: 834–836.
Morrow JD, Awad JA, Oates JA, Roberts II LJ . Identification of skin as a major site of prostaglandin D2 release following oral administration of niacin in humans. J Invest Dermatol 1992; 98: 812–815.
Smesny S, Klemm S, Stockebrand M, Grunwald S, Gerhard UJ, Rosburg T et al. Endophenotype properties of niacin sensitivity as marker of impaired prostaglandin signalling in schizophrenia. Prostaglandins Leukot Essent Fatty Acids 2007; 77: 79–85.
Steiner J, Walter M, Wunderlich MT, Bernstein HG, Panteli B, Brauner M et al. A new pathophysiological aspect of S100B in schizophrenia: potential regulation of S100B by its scavenger soluble RAGE. Biol Psychiatry 2009; 65: 1107–1110.
Riecher-Rossler A, Gschwandtner U, Borgwardt S, Aston J, Pfluger M, Rossler W . Early detection and treatment of schizophrenia: How early? Acta Psychiatr Scand Suppl 2006; 429: 73–80.
Acknowledgements
This study was supported by the Stanley Medical Research Institute (SMRI), Psynova Neurotech and the European Union FP7 SchizDX research programme (grant reference 223427). We want to thank Anke Dudeck, Jeanette Schadow, Dr Wolfgang Jordan, Dr Bernd Hahndorf, Dr Florian Kästner, Dr Anya Pedersen, Dr Ansgar Siegmund, Dr Katja Kölkebeck, Torsten Schoenborn, Dr Christoph W Gerth, Dr Christian Mauss, Dr Brit M Nolden, Dr MA Neatby, Dr Liliana Ruta and Dr Erin Ingudomnukul for their participation in sample characterization and collection. Thanks to all members of the Bahn Laboratory for discussions, help and encouragement. Most of all, thanks to all patients and healthy volunteers for their selfless donation of samples used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Schwarz, E., Guest, P., Rahmoune, H. et al. Identification of a biological signature for schizophrenia in serum. Mol Psychiatry 17, 494–502 (2012). https://doi.org/10.1038/mp.2011.42
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2011.42
Keywords
This article is cited by
-
Consolidation of metabolomic, proteomic, and GWAS data in connective model of schizophrenia
Scientific Reports (2023)
-
Changes in leukocytes and CRP in different stages of major depression
Journal of Neuroinflammation (2022)
-
Dysregulation of complement and coagulation pathways: emerging mechanisms in the development of psychosis
Molecular Psychiatry (2022)
-
Common and disorder-specific upregulation of the inflammatory markers TRAIL and CCL20 in depression and schizophrenia
Scientific Reports (2021)
-
Transcriptome analysis of fibroblasts from schizophrenia patients reveals differential expression of schizophrenia-related genes
Scientific Reports (2020)