Abstract
Intestinal helminth parasites are potent inducers of T helper type 2 (Th2) response and have a regulatory role, notably on intestinal inflammation. As infection with schistosomes is unlikely to provide a reliable treatment of inflammatory bowel diseases, we have investigated the beneficial effect of a schistosome enzymatic protein, the 28-kDa glutathione S-transferase (P28GST), on the modulation of disease activity and immune responses in experimental colitis. Our results showed that immunization with recombinant P28GST is at least as efficient as established schistosome infection to reduce colitis lesions and expression of pro-inflammatory cytokines. Considering underlying mechanisms, the decrease of inflammatory parameters was associated with the polarization of the immune system toward a Th2 profile, with local and systemic increases of interleukin (IL)-13 and IL-5. Dense eosinophil infiltration was observed in the colons of P28GST-immunized rats and mice. Depletion of eosinophils by treatment with an anti-Siglec-F monoclonal antibody and use of IL-5-deficient mice led to the loss of therapeutic effect, suggesting the crucial role for eosinophils in colitis prevention by P28GST. These findings reveal that immunization with P28GST, a unique recombinant schistosome enzyme, ameliorates intestinal inflammation through eosinophil-dependent modulation of harmful type 1 responses, representing a new immuno-regulatory strategy against inflammatory bowel diseases.
Similar content being viewed by others
INTRODUCTION
Inflammatory bowel diseases (IBD), including Crohn’s disease and ulcerative colitis, are considered as part of immune-mediated inflammatory disorders. The burden of IBD varies in different countries; incidence is currently stable in Northern countries but has increased noticeably in developing countries, suggesting a major role for environmental factors.1 Among other factors, intensive antiparasitic treatment (or deworming) might have disturbed long-term developed regulatory networks, and may be, at least partly, responsible for the emergence of inflammatory disorders.2 The formulation of the IBD hygiene hypothesis is based on epidemiological and experimental evidence and suggests that helminth infections have modulatory effects in several inflammatory disorders, including IBD.2, 3
Although the underlying pathogenesis of IBD is not well understood, it is currently believed that IBD is caused by inappropriate activation of the mucosal immune system, which in turn results in a state of chronic inflammation that is associated with dysregulation of the cytokine network. Crohn’s disease pathology is associated with a T helper type 1 (Th1)/Th17 response, with colitogenic, pro-inflammatory Th1 cells expressing interferon-γ (IFNγ) and tumor necrosis factor (TNF).4 Th17 cells massively infiltrate the inflamed intestine of patients, where they produce interleukin (IL)-17 and other cytokines, triggering and amplifying the inflammatory process.
The role of helminth parasites in controlling wound healing and harmful inflammatory responses has been recently reviewed.5, 6 It is proposed that intestinal helminths induce two separable components of immunity, a type 2 cytokine response and a regulatory response, that synergize to dampen the type 1 pro-inflammatory response. An observed increase of type 2 cytokines, namely IL-5 and IL-13, in the mucosal tissues during helminth infection, is likely caused by the activation of innate lymphoid 2 cells by cytokine alarmins, including IL-25, IL-33, and thymic stromal lymphopoietin.6 Blood and tissue eosinophil increase is a classical feature of helminth infections, and a role for resident innate lymphoid 2 cells in tissue eosinophil accumulation has been emphasized.7
Among other helminths, schistosomes are considered “masters of regulation”.8, 9 During schistosome infections, major Th2 responses follow an early Th1 response and might be associated with a later pathological Th17 response that accompanies fibrosis.6 Earlier studies have indicated a role for schistosome-induced Th2 responses in controlling pathological Th1-type responses.10, 11 Alternatively activated macrophages are involved in the downmodulation of Th1 responses and immunopathology in schistosomiasis.12 The immuno-modulatory properties of schistosomes have been validated in several models of immunopathology. Experimental infections with schistosomes confer protection from immune dysregulatory diseases, such as type 1 diabetes,13 multiple sclerosis,14 and colitis.15 Omega-1, a glycoprotein present in schistosome eggs, has been shown to drive Th2 polarization16; moreover, the effects of excretion–secretion products by the eggs or adult worms of schistosomes in the modulation of experimental colitis have also been described.17 Therefore, it appeared challenging to identify the molecules involved in these regulatory properties and to dissect their mode of action.
One family of potent immunogenic and pharmacologically active enzymatic proteins, the glutathione S-transferases (GSTs), has been identified in schistosomes. Among GSTs, P28GST18 exhibits a unique structural feature19 and has been developed as an antiparasite and antipathology vaccine in several experimental models of schistosomiasis, including in non-human primates.20, 21, 22, 23 The observations that schistosome P28GST induced a strong mucosal immune response, associated with Th2-type and regulatory IL-10 cytokines in experimental animals and in humans,24, 25, 26, 27 prompted us to examine the effect of treatment with recombinant P28GST, on experimentally induced colon inflammation.
We hypothesized that immunization with schistosome-derived P28GST induced an immuno-regulatory response that renders the gut less prone to the development of colitis. Using the model of 2, 4, 6-trinitrobenzene sulfonic acid (TNBS)-induced colitis, which induces a Th1-like inflammation in rats and mice,28 we provide evidence that administration of P28GST in the presence of adjuvant, reduces the severity of intestinal inflammation, and results in modulation of Th responses, associated with regulatory eosinophils.
RESULTS
The unique parasite protein P28GST prevents colitis in rats
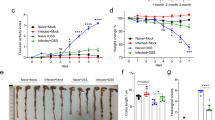
We first used the TNBS acute model of colitis to assess whether P28GST is able to reproduce the modulation of intestinal inflammation observed after infection with the whole parasite. We then compared the effect of subcutaneous immunization of Sprague-Dawley rats with 2 injections of 25 μg kg−1 recombinant P28GST vs. recent (1 week) or established (5 weeks) infection with schistosome larvae, prior to TNBS administration. As expected, the TNBS-positive control group displayed important macroscopic and histological lesions (Figure 1a,b). The established infection led to a significant decrease in clinical and histological scores, a result which was not observed in the recently infected rats (Figure 1a,b). Interestingly, rats that have been immunized by P28GST showed an important decrease in both scores. This was associated with a 50% reduction of the colonic myeloperoxidase (MPO) level in P28GST-immunized rats, similar to the established infection group but in contrast to recently infected rats (Figure 1c). Macroscopic improvements observed in P28GST-immunized rats and in rats with established infection were characterized by the decrease of the colon thickness and the reduction of mucosal ulcerations (number and surface), as compared with the other groups of rats (see Supplementary Figure S1a online). Histological examination of the distal colon of rats administered with TNBS (positive control) or recently infected revealed the complete disorganization of structures (see Supplementary Figure S1b). When rats had been immunized with P28GST or infected for 5 weeks, we observed striking improvement in these histological lesions.
Prevention of 2, 4, 6-trinitrobenzene sulfonic acid (TNBS)-induced colitis in rats by P28GST treatment or established infection with living parasites. Sprague-Dawley rats were immunized with two injections of 25 μg kg−1 P28GST at 1-month interval or infected with living Schistosoma cercariae either 5 weeks (established infection) or 1 week (recent infection) before TNBS injection. TNBS colitis was induced in all the groups, except negative controls, which received ethanol solution. Positive control rats were only injected with TNBS. All rats were killed 4 days after injection of TNBS or ethanol, i.e., 39 days after the first injection or infection. (a) Clinical score of rats with TNBS-induced colitis or negative controls. (b) Histological score of colon tissues assessed blindly by a pathologist to determine the degree of colitis using a score from 0 to 6. (c) Myeloperoxidase (MPO) was evaluated in the colon in the five groups of rats by enzyme-linked immunosorbent assay. Results are expressed as ng per mg of total proteins. (d) Interleukin (IL)-1β, (e) tumor necrosis factor (TNF), and (f) IL-17 mRNA levels analyzed by quantitative PCR were significantly lower in the P28GST-treated rats than in the colitis control group or in the recent infection group. No significant difference was observed between P28GST-treated rats and rats with established infection. Results are expressed as means±s.e.m.; n=20 rats per group. *P<0.05; **P<0.01; NS, not significant.
To further analyze the effect of P28GST immunization on inflammatory responses, the gene expression of inflammatory cytokines was evaluated in colonic tissues. IL-1β, TNF, and IL-17 mRNA levels were markedly lower in P28GST-immunized rats than in TNBS-positive control rats (Figure 1d–f). A similar decrease was observed in rats with established infection but not in recently infected rats. Collectively, we demonstrated that a preventive treatment with recombinant P28GST by subcutaneous route induces a potent intestinal anti-inflammatory effect that reaches the same level of reduction of inflammation markers as an established infection with living schistosome larvae.
Immunization of rats with optimal doses of P28GST is as efficient as a reference treatment
Dose–response experiments with P28GST and comparison with 5-amino-salicylic acid (5-ASA) treatment were then performed. Prior to TNBS injection, rats were immunized with two injections of P28GST at different concentrations, with adjuvant alone or treated with 5-ASA. Animals immunized with 5 or 0.5 μg kg−1 P28GST exhibited significantly decreased clinical and histological scores compared with positive control rats (Figure 2a,b). In contrast, the highest concentrations of P28GST (50 μg kg−1) as well as 150 μg kg−1 (data not shown) had no effect on colitis. In line, high tissue levels of MPO observed in control rats were significantly reduced in P28GST-treated rats only for doses of 5 and 0.5 μg kg−1 (Figure 2c). This dose-dependent decrease of MPO was accompanied with an important drop in the colonic expression of pro-inflammatory cytokines IL-1β, TNF, and IL-17 for the lowest doses of P28GST (Figure 2d–f). The results presented in Figure 2 show that immunization with the optimal doses of P28GST induced the same effects as the reference treatment with 5-ASA.
Dose-dependent immunization of rats with P28GST compared with a reference treatment with 5-amino-salicylic acid (5-ASA). Rats were either immunized with two injections of P28GST at three concentrations (50, 5, or 0.5 μg kg−1) in the presence of aluminum hydroxide as adjuvant, with adjuvant alone before 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) injection, or treated with 5-ASA (30 mM). (a) Clinical score (Wallace’s score) of rats with TNBS-induced colitis. (b) Histological score (Ameho’s score) of colon tissues assessed blindly by a pathologist to determine the degree of colitis using a score from 0 to 8. (c) Myeloperoxidase (MPO) was evaluated in the colon in the six groups of rats by enzyme-linked immunosorbent assay. Results are expressed as ng per mg of total proteins. Levels of (d) interleukin (IL)-1β, (e) tumor necrosis factor (TNF), and (f) IL-17 mRNA in the different groups of rats were assessed by quantitative PCR. Results are expressed as means±s.e.m.; n=10 rats per group. *P<0.05; **P<0.01; ***P<0.001.
P28GST immunization prevents acute experimental colitis in mice
These data were confirmed in BALB/c mice where immunization with lower doses of P28GST also led to stronger improvement of inflammatory lesions and markers (Figure 3). Indeed, BALB/c mice immunized with 25 or 5 μg kg−1 P28GST exhibited significantly decreased weight loss compared with adjuvant-treated mice (Figure 3a), whereas the highest concentration of P28GST (50 μg kg−1) had no effect on colitis. Representative photographs of colon length from the different groups indicated that P28GST treatment at the two optimal doses prevented the colon shortening that is classically observed during colitis (Figure 3b). Parallel decrease of clinical and histological scores was observed for the corresponding concentrations of P28GST (Figure 3c,d). Compared with adjuvant-treated mice, low histological damage was observed in the colons of mice treated with P28GST at 25 and 5 μg kg−1 (Figure 3d). The loss of typical colonic crypt structure in control mice was accompanied by submucosal swelling, microclot formations, and necrosis of the colonic mucosa and that observed in adjuvant-treated mice were significantly reduced by one-half in P28GST-treated mice (Figure 3f).
P28GST immunization protects BALB/c mice from acute 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) colitis. Mice were immunized with three injections of P28GST (50, 25, 5, or 0.5 μg kg−1) in the presence of adjuvant or with adjuvant alone, at 2-week interval. TNBS colitis was induced in all the groups. (a) Percentage of loss of initial weight were expressed as the comparison of body weight of individual mice recorded at day 34 (before diet) and at day 37 (at killing). (b) A representative photograph of colons removed at day 37 (2 days after the induction of TNBS colitis). The colon length is expressed in centimeters (cm). (c) Clinical and (d) histological scores of the colons from mice with acute TNBS colitis. A blinded pathologist assessed different inflammatory characteristics to determine the degree of colitis using macroscopic scoring system described by Wallace and Keenan or histological Ameho’s scoring. (e) Hematoxylin and eosin staining of paraffin-wax-embedded sections of the colon from mice injected with either adjuvant, 5 μg kg−1 P28GST before TNBS injection, or negative controls corresponding to mice without TNBS colitis. (f) Enzyme-linked immunosorbent assay was performed to quantify myeloperoxidase (MPO) in the colon of P28GST- or adjuvant-injected mice. MPO levels expressed as ng per mg of total protein are lower in the P28GST-treated animals. Results are expressed as means±s.e.m.; n=15 mice per group. *P<0.05, **P<0.01; NS, not significant.
Taken together, the data obtained both in rats and mice indicated that the P28GST effect is not species dependent but is optimal for doses ranging from 5 to 25 μg kg−1.
Adjuvant is required for fully efficient anti-inflammatory properties of P28GST
The requirement for adjuvant in the anti-inflammatory effect of P28GST immunization was then evaluated. Prior to TNBS rectal injection, mice were immunized with 25 μg kg−1 P28GST in the presence or absence of aluminum hydroxide as adjuvant. Various parameters of inflammation were compared between immunized mice and control groups of mice that had been injected with TNBS (positive control), adjuvant alone, or treated with 5-ASA as reference treatment (Figure 4). Only treatment with 5-ASA or immunization with P28GST in the presence of adjuvant significantly decreased clinical and histological scores, MPO level, and IL-1β and TNF mRNA levels (Figure 4a–e). Immunization with adjuvant alone did not affect scores, MPO, or IL-1β mRNA levels and actually led to a significant decrease in TNF mRNA. This confirmed that adjuvant is necessary for optimal efficacy of P28GST immunization and that aluminum hydroxide had no pro-inflammatory effect in our experimental immunization conditions.
Mice were immunized either with aluminum hydroxide as adjuvant alone, with P28GST (25 μg kg−1) in the presence, or in the absence of adjuvant (with P28GST re-suspended in physiological serum). 2, 4, 6-Trinitrobenzene sulfonic acid (TNBS) colitis was induced in all the groups, except negative control, which received ethanol solution. Colons were removed after the killing of mice at day 37 and paraffin embedded. Paraffin-wax-embedded sections of the colon were stained with hematoxylin and eosin. (a) Clinical and (b) histological scores of the colons from mice with acute TNBS colitis. (c) Enzyme-linked immunosorbent assay was performed to quantify myeloperoxidase (MPO) in the colon of P28GST-, adjuvant, or 5-amino-salicylic acid (5-ASA)-injected mice. MPO levels expressed as ng per mg of total protein are lower in the P28GST –treated animals. (d) Interleukin (IL)-1β and (e) tumor necrosis factor (TNF) mRNA expression was assessed by quantitative PCR in the colons from the six groups of mice. Results are expressed as means±s.e.m.; n=30 mice per group. *P<0.05; **P<0.01; ***P<0.001.
P28GST treatment induces Th2-type polarization and eosinophil recruitment
To explore underlying mechanisms for P28GST anti-inflammatory role, we investigated tissue immuno-regulatory environment in rats (Figure 5) as well as in mice (Figures 6 and 7).
P28GST-immunization as well as schistosome infection of rats stimulate the production of T helper type 2–type cytokines. Rats were immunized with P28GST (two injections of 25 μg kg−1) or infected with living Schistosoma cercariae either 5 weeks (established infection) or 1 week (recent infection) before 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) injection. TNBS colitis was induced in all groups, except negative control, which received ethanol solution. Animals injected with TNBS were considered as the positive control group. (a) Levels of interferon-γ (IFNγ) mRNA in the different groups of rats were assessed by quantitative PCR (qPCR). The levels of (b) interleukin (IL)-4, (c) IL-13, and (d) IL-10 mRNA assessed by qPCR were increased in P28GST-treated rats by comparison to rats with established infection. (e) Colons were removed after the killing of rats at day 39 and paraffin embedded. Representative images from colon tissues from rats immunized with P28GST or infected with living S. cercariae either 5 weeks (established infection) or 1 week (recent infection) and controls with (positive control) or without (negative control) TNBS. Paraffin-wax-embedded sections of the colon were stained with hematoxylin and eosin. Images are printed at the same final magnification ( × 20). (f) IL-5 mRNA expression was assessed by qPCR in the colons from the five groups of rats. Results are expressed as means±s.e.m.; n=20 rats per group *P<0.05; **P<0.01; NS, not significant.
P28GST immunization of mice alters mRNA colonic levels of pro-inflammatory and T helper type 2–type cytokines. Mice were immunized with P28GST (25 or 5 μg kg−1) or adjuvant and 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) colitis was induced in all the groups. (a) Tumor necrosis factor (TNF) and (b) interleukin (IL)-1β mRNA levels assessed by quantitative PCR are decreased in mice immunized with P28GST (5 μg kg−1) when compared with positive controls. (c) IL-10, (d) IL-5, and (e) eosinophil peroxidase (EPO) mRNA levels increased in P28GST (25 or 5 μg kg−1) immunized mice when compared with adjuvant. n=15 mice per group. (f) Eosinophil quantification in colon sections of negative controls, of mice immunized with adjuvant only, or with P28GST (5 μg kg−1) in the presence of adjuvant. Hematoxylin and eosin–stained colon sections of mice were scored for degree of eosinophil infiltration. Area=90,000 μm2 (area 300 × 300 μm2). n=10 mice per group. Results are expressed as means±s.e.m.; *P<0.05, **P<0.01, ***P<0.001.
P28GST immunization of mice modifies the production by spleen cells of T helper type 2 (Th2)- and Th1-type cytokines. Spleen cells were recovered from the three groups of mice: immunized with P28GST (25 or 5 μg kg−1) or adjuvant. Total spleen cells were stimulated either non-specifically with Concanavalin A (ConA) or specifically with P28GST, and cytokine concentrations were determined in supernatants by Luminex. (a) Interleukin (IL)-4, (b) IL-13, (c) IL-10, (d) IL-5, (e) tumor necrosis factor (TNF), (f) IL-1β, (g) IL-17, and (h) interferon-γ (IFNγ) concentrations were measured. Results are the mean±s.e.m. of a spleen cell pools from 15 mice per group. *P<0.05.
The Th2-polarized immune response is characterized by elevated IL-4, IL-5, and IL-13 and associated with a decrease of Th1-type cytokines, such as IFNγ. To further evaluate the effect of P28GST immunization (25 μg kg−1) on the local immune response profile, quantitative PCR was performed on colonic tissues from the different groups of rats at the time of killing. Reduced IFNγ mRNA levels were observed in P28GST-immunized rats or rats with an established infection (Figure 5a). In contrast, IL-4 and IL-13 mRNA levels were significantly higher in P28GST-immunized rats than in both groups of infected rats (Figure 5b,c). The slight increase in IL-10 mRNA did not reach significance. No difference in transforming growth factor-β mRNA levels was observed between the groups of rats (not shown).
We subjected stained sections from the colons of the different groups of rats to histological examination in order to investigate the cellular mechanisms driven by the Th2-type response (Figure 5e). In TNBS-positive, but not in -negative controls, increased thickness of the colonic wall was observed, with ulceration and infiltration of the lamina propria with polymorphonuclear cells, mainly neutrophils. In contrast, infiltration of the lamina propria with eosinophils was observed in P28GST-immunized rats and in rats with established infection. Levels of colonic IL-5 mRNA support these observations (Figure 5f) Altogether, these findings indicate that immunization of rats with P28GST, a unique recombinant protein from the schistosome helminth, ameliorated inflammation through potent immuno-modulatory effects and changes in cell recruitment.
P28GST treatment alters the cytokine profile in the colons and spleen cells of mice
We next investigated the effect of P28GST immunization on cytokine profiles both in the colons and spleen of mice. We subjected colonic tissues from immunized vs. control BALB/c mice to quantitative PCR 2 days after TNBS administration (Figure 6). When mice immunized with P28GST (25 and 5 μg kg−1) were compared with the adjuvant group, we observed decreased expression of TNF and IL-1β mRNAs (Figure 6a,b). These reductions were associated with marked increases of IL-10 and IL-5 mRNAs (Figure 6c,d); no difference in IFNγ mRNA level was observed (not shown). A significant increase of colonic eosinophil peroxidase mRNA suggested the presence of eosinophils in the colons of P28GST-immunized mice compared with control mice (Figure 6e).
Eosinophil numbers have been determined by microscopy in colon submucosa of mice treated with P28GST (5 μg kg−1) or with adjuvant alone, using a conventional histology staining and scoring as described in the Methods section. Scores for each histological feature examined were significantly higher in P28GST-immunized group as compared with controls (Figure 6f). Extensive infiltration of eosinophils occurs in the mucosal and submucosal layers. Eosinophils were more numerous near lymphoid follicles.
To determine whether this local response was associated with systemic cytokine production, we isolated spleen cells from the different groups of mice. Cytokines produced by spleen cells were measured in supernatants from P28GST-immunized or adjuvant control animals after in vitro activation with either Concanavalin A or P28GST. Concanavalin A stimulation of spleen cells from the different groups of mice induced both Th1 and Th2 cytokine release, confirming the normal T-cell response to a non-specific activator (Figure 7a–h, left panels). Incubation of spleen cells with P28GST led to a significant increased production of IL-4, IL-13, IL-10, and IL-5 by cells from P28GST-immunized mice but not from adjuvant-treated mice (Figure 7a–d). In contrast, TNF, IL-1β, IL-17, and IFNγ levels were low (Figure 7e–h). Collectively, our findings indicate that immunization with the schistosome P28GST favors the production of Th2-type and regulatory cytokines both locally and systemically in the context of acute experimental colitis.
Eosinophil depletion influences the effects of P28GST immunization
The similarities regarding IL-5 mRNA increase and eosinophil infiltration in P28GST-immunized rats and mice, in parallel with reduced inflammation, led us to consider the regulatory role of eosinophils. Treatment of BALB/c mice with anti-Siglec-F monoclonal antibody (mAb)29 induced selective blood eosinophil depletion (see Supplementary Figure S2) and strongly reduced the increases of IL-5 and eosinophil peroxidase mRNAs that had been observed after P28GST immunization (Figure 8a,b). In contrast to isotype control (see Supplementary Figure S3), treatment with anti-Siglec-F mAb partially blunted the preventive effect of P28GST on weight loss, as well as its effect on clinical and histological scores after TNBS injection (Figure 8c–e). Treatment of P28GST-immunized mice with anti-Siglec-F mAb exerted a significant effect on the colonic MPO protein and, to a lesser extent, on TNF mRNA (Figure 8f,g). The highly significant increase of IL-10 mRNA expression in P28GST-immunized mice was no longer observed in eosinophil-depleted mice (Figure 8h). Experiments using IL-5-deficient mice confirmed the results obtained by depletion with anti-Siglec-F mAb, notably demonstrating the lack of decrease of the clinical score and TNF mRNA in P28GST-immunized IL-5 knockout mice vs. their congenic controls (Figure 9a–c). These data indicate that eosinophil depletion, at least partially, blocks the protective effect of P28GST on colitis, which suggests that eosinophils participate in the regulation of P28GST-induced inflammation.
Eosinophils are implied in the protective effect of P28GST. Mice were immunized with P28GST (5 μg kg−1) or adjuvant and were injected intrarectally with 2, 4, 6-trinitrobenzene sulfonic acid (TNBS). To deplete eosinophils, mice received five intraperitoneal injections of anti-Siglec-F mAb (10 μg per mouse) at days 30, 33, 34, 35, and 36. Mice were killed at day 37. (a) Interleukin (IL)-5 and (b) eosinophil peroxidase (EPO) mRNA expression was assessed by quantitative PCR (qPCR). (c) Loss of body weight, (d) macroscopic, and (e) microscopic analysis of the colons were determined. (f) Myeloperoxidase (MPO) concentrations in colon lysates were evaluated by enzyme-linked immunosorbent assay. (g) Tumor necrosis factor (TNF) and (h) IL-10 mRNA expression was assessed by qPCR. Results are expressed as means±s.e.m.; n=10 mice per group. *P<0.05, **P<0.01, ***P<0.001; NS, not significant.
Exacerbation of the 2, 4, 6-trinitrobenzene sulfonic acid (TNBS)-induced colitis despite P28GST immunization in interleukin (IL)-5 knockout (KO) mice. Mice were immunized with P28GST (5 μg kg−1) in the presence of adjuvant or with adjuvant alone. TNBS colitis was induced in all the groups. (a) Clinical score (Wallace’s score) of mice with TNBS-induced colitis. (b) Enzyme-linked immunosorbent assay was performed to quantify myeloperoxidase (MPO) in the colon of P28GST- or adjuvant-injected mice. MPO levels expressed as ng per mg of total protein are lower in the P28GST-treated animals.(c) Tumor necrosis factor (TNF) mRNA expression assessed by quantitative PCR. Results are expressed as means±s.e.m.; n=15 mice per group. *P<0.05, **P<0.01, ***P<0.001; NS, not significant.
DISCUSSION
Epidemiological, experimental, and interventional human studies support the hypothesis that helminth parasitic infections can confer protection against immune dysregulatory diseases.30, 31, 32, 33 Infection with living schistosomes, as well as injection of either schistosome egg antigen or soluble adult worm proteins, ameliorates mouse colitis.17, 6 However, the specific schistosome molecules responsible for these effects remain to be identified. In light of our studies on the immune responses to schistosomes, we have characterized the GST, especially P28GST, as one of the main immunogenic and pharmacologically active enzymatic families.25, 26, 27 In various experimental models of schistosomiasis, including non-human primates, P28GST immunization induces a Th2-type response associated with decreasing immunopathology.20, 21, 22 The safety of a recombinant form of P28GST has been demonstrated in humans24; it therefore represents one of the best characterized helminth-derived molecules, ready for use in humans, owing to its immune-modulating properties.
Consistent with previous reports,17, 6 our data show that preventive treatment of rats by infection with Schistosome cercariae before TNBS injection significantly attenuated macroscopical and histological features of acute colonic inflammation. In contrast with these effects, we observed that recent infection with schistosome larvae, 1 week before TNBS injection, did not induce the same effects. Indeed in this setting, all parameters of colonic inflammation remained unchanged compared with colitis controls. In human schistosomiasis, the immune response during the acute phase is also associated with a Th1 response, with increased production of TNF and IFNγ.32, 34 During the chronic phase, the immune response is of Th2 type, with elevated levels of IL-4 and IL-5 and decreased levels of IFNγ.31, 33, 35, 36 Our data reveal that immunization of rats with P28GST, a recombinant schistosome enzymatic protein, reproduced effects similar to those induced by an established infection with living parasite larvae.
P28GST immunization at the same level as live schistosome infection significantly reduced local expression of pro-inflammatory mediators, including TNF and IL-17, providing a molecular basis for the anti-inflammatory response. Through potent immuno-modulatory effects, P28GST strongly decreases intestinal inflammation in rodent models of IBD.
The cytokine responses that characterize IBD are the key pathophysiological elements that govern the initiation, evolution, and, ultimately, the resolution of inflammation. In Crohn’s disease, gut inflammation is mediated through Th1 and Th17 cells,6, 17 while a strong Th2 immune response is induced during helminth infections.8 Several helminths or helminth extracts, including S. mansoni soluble worm proteins, Ancylostoma ceylanicum adult worm extracts (AcAw), and hookworm excretory/secretory products, attenuate colitis in TNBS or dextran sodium sulfate models, by downregulating Th1 and Th17 cytokines and inducing IL-4-and IL-10 CD4-positive T cells.6, 17 Here we show that immunization of mice with P28GST, a pro-Th2 schistosome enzyme, induced significant changes in cytokine mRNA levels in the colon that were accompanied by a comparable systemic immune response to P28GST with increased levels of IL-4, IL-13, IL-5, and IL-10; in contrast, TNF, IL1β, IL-17, and IFNγ levels remained very low. This finding is in accordance with our previous observations that immunization of humans with P28GST in the presence of aluminum hydroxide induced high levels of serum IL-5, IL-13, and IL-10, after the second administration of P28GST.21, 24 The different dose–effect experiments revealed that immunization by P28GST at 25 or 5 μg kg−1 gives not only significant results in all experiments but also coherent results on reduction of scores and inflammation markers, increase of Th2 cytokines, and eosinophil infiltration. Both in rats and in mice, the highest dose of 50 μg kg−1 appears unable to induce colitis reduction. A possible aggregation, at the highest concentrations, of recombinant P28GST, which is mainly produced under a dimeric form, may prevent an optimal taking up by antigen-presenting cells.
Alhydrogel administration enhances antigen presentation and upregulates the expression of costimulatory molecules on antigen-presenting cells. As recent reports suggest that type II natural killer cells may act as part of the alhydrogel-sensing apparatus and facilitate Th2-driven humoral immunity,37 we have chosen alhydrogel as a pro-Th2 adjuvant. In the present experiments, in both rats and mice immunization with adjuvant alone did not induce any inflammatory reaction under the conditions of immunization. However, alhydrogel appeared essential to induce the immuno-modulatory effect of P28GST, which is in agreement with the helpful adjuvant effects of alum. It is also of note that P28GST immunization of rats as well as of mice induced a reduction of inflammation scores and markers comparable to that observed with 5-ASA used as the reference treatment.
A common feature observed in rats and mice is the increase of IL-13, and IL-5, together with a significant infiltration of the colonic lamina propria by eosinophils, which suggests their participation in the regulatory process. The function of eosinophils in health and disease has been a matter of controversy for a long time, ranging from a protective role in some helminth infections to a cellular target for the treatment of inflammatory disorders.38 This dichotomy between beneficial and non-beneficial eosinophils has recently been reviewed in the context of gastrointestinal disorders,39, 40 and the multifaceted role of gastrointestinal eosinophils appears now more likely.39 Recent studies demonstrating that eosinophils contribute to gut immune homeostasis41 and exert protective effects in acute mouse colitis models42 have opened the discussion about our results, suggesting that eosinophils participate, at least partly, in the attenuation of intestinal inflammation induced by a parasitic protein.
Eosinophils are classically associated with a Th2 profile that includes IL-4, IL-5, IL-10, and IL-13 as growth and stimulation factors, which are notably increased after P28GST immunization. In light of the recent results that emphasize the critical role of eosinophils in the regulation of intestinal inflammation,42 the interesting question is whether or not eosinophils participate in the P28GST-induced attenuation of gut inflammation. To answer this question, we used BALB/c mice, selectively depleted of eosinophils by treatment with anti-Siglec-F mAb as well as IL-5-deficient mice. Eosinophil depletion significantly decreased the effect of P28GST immunization on clinical and histological scores, with a marked effect on IL-10 mRNA level. These results indicate that, at least in this model, eosinophils participate in the immuno-regulatory circuits. One of the known properties of eosinophils is that they release IL-13 and that they are implied in the activation of M2 macrophages.12, 43 Preliminary results in both rats and mice indicate that arginase vs. iNOS mRNA levels are preferentially detected in the colon of P28GST-immunized animals, a result that is in agreement with the decreased inflammation (data not shown).
In agreement with recently published reports, one of the major issue of the findings described in the present study is the consistency of results, pointing to the prominent role of helminths and more interestingly helminth-derived molecules in the regulation of IBD. General interest is increasing in helminth-derived molecules as new promising therapeutics for IBD and autoimmune diseases. Compared with other helminth-derived molecules, P28GST offers several advantages. The first advantage is at the biochemical level: its production at a very high degree of purity in a recombinant form allowed crystallization studies, as well as the precise identification of the active enzymatic domains.19 Second, immunological analysis confirmed the absence of cross-reactivity of these active domains with human GSTs, which is an important parameter to consider in order to avoid any adverse auto-immune response as a result of immunization. Finally, it has to be mentioned that P28GST, which has already been used successfully in phase 1 clinical trials for safety and immunogenicity studies24 (Clinical Trials identifier: NCT 01512277), is presently under investigation for safety in Crohn’s disease patients (Clinical Trials identifier: NCT 02281916).
In conclusion, our data indicate that immunization with the unique parasite enzyme P28GST limits the development of acute colitis by decreasing pro-inflammatory Th1 responses and inducing a Th2 and regulatory response involving eosinophils. These findings suggest that immunotherapy with P28GST represents an additional strategy to improve inflammatory processes in the context of IBD.
METHODS
Animals. Sprague-Dawley rats were purchased from Janvier Europe SAS (Le Genest-Saint-Isle, France) (6–8 weeks of age). BALB/c mice were purchased from Charles River (L’Arbresle, France) (8 weeks of age). Mice heterozygous for the IL5tm1Kopf targeted mutation were purchased from the Jackson Laboratory (Bar Harbor, ME). Homozygous animals were obtained by breeding of heterozygous parents and were identified by PCR analysis.
Animals were maintained in pathogen-free animal-holding facilities. Housing and all of the experimental procedures were approved by the local Animal Care Ethical Committee CEEA (Agreement no. 352012) and were performed in accordance with the French Guide for the Care and Use of Laboratory Animals and the guidelines of the European Union.
P28GST, a recombinant enzyme. Batches of P28GST protein were produced and purified from recombinant Saccharomyces cerevisiae culture under Good Manufacturing Practice conditions by Eurogentec S.A (Seraing, Belgium). P28GST was conserved lyophilized in NH4HCO3 10 mM and 6% lactose. This preparation was re-suspended extemporaneously using aluminum hydroxide 0.2% solution, an adjuvant compatible with animal and human use (Alhydrogel 2% from InvivoGen, Toulouse, France) at the appropriate concentrations.
Adjuvant control is composed of the formulation buffer of recombinant P28GST (NH4HCO3 10 mM and 6% lactose) and was re-suspended extemporaneously using aluminum hydroxide solution at 0.2%.
5-ASA (Sigma-Aldrich, Saint-Quentin Fallavier, France) was used at 30 mM by intrarectal single daily administration 1 week before colitis induction.
Animal models. Six-to-8-week-old male rats and 6-week-old female mice were randomly divided into different groups. Daily body weights were recorded.
Infection protocols. Under anesthesia, rats were infected transcutaneously with 2,000 Schistosome cercariae diluted in 2 ml physiological saline. Two protocols of infections were used. First, an “established infection” model in which rats were infected with S. cercariae 5 weeks before the induction of TNBS colitis. Second, a “recent infection” model in which animals were exposed to schistosome larvae only 1 week before colitis induction.
Immunization protocols. Rats were immunized by two subcutaneous injections of 50, 25, 5, or 0.5 μg kg−1 P28GST with an interval of 4 weeks between the two injections. BALB/c mice were immunized by three subcutaneous injections (50, 25, 5, and 0.5 μg kg−1) of P28GST with an interval of 2 weeks between each injection. In both animal models, the last injection was performed 1 week before TNBS. Control groups were as indicated in the figure legends. IL-5-deficient mice were immunized by three subcutaneous of 5 μg kg−1 P28GST with an interval of 2 weeks.
Colitis induction. All animals were anesthetized before the intrarectal injections. Thirty-five days after the first injection of P28GST with or without adjuvant, and 1 week after the last injection, animals from TNBS groups received an intracolonic injection of TNBS (picrylsulphonic acid solution 1 M in H2O, Sigma-Aldrich) (250 μl, 100 mg kg−1 for rats, or 40 μl, 150 mg kg−1 for mice, diluted in 50% ethanol). TNBS was slowly administered with a catheter inserted 8 cm for rats or 4 cm for mice into the colon through the anus. For the negative control group, animals received an intrarectal injection of ethanol solution. Rats are killed 4 days after TNBS injection, while mice are killed 2 days after TNBS injection. As TNBS is injected 35 days after the first injection of P28GST, the end point is presented at day 39 (for rats) or 37 (for mice).
Treatment with 5-ASA. 5-ASA (30 mM) was used intrarectally by single daily administration 1 week before TNBS injection.
Eosinophil depletion. Before colitis induction, BALB/c mice received five intraperitoneal injections of anti-Siglec-F mAb (clone 238047, R&D Systems, Lille, France) or isotypic control (Monoclonal Rat IgG2A, R&D Systems): 10 μg per mouse at days 30, 33, 34, 35, and 36.
Tissue preparation. Rats were killed at 96 h and mice at 48 h after colitis induction. The colon was removed and opened. After scoring the gross morphological damage, the distal ∼1 cm of the colonic segment was used for histology. The next ∼1 cm segment was used for MPO assay and RNA extraction, and it was immediately frozen and stored at −80 °C.
Spleen was removed and placed on ice, and cell suspension was prepared. Erythrocytes were lysed with Tris-buffered ammonium chloride. Mononuclear cells were washed and re-suspended in RPMI complete medium.
Macroscopic damage score. Macroscopically visible damage of the opened colonic segment was blindly scored on a 0–10 scale using the scoring system for Wallace44 based on inflammation criteria. A score of 0 represents no visible damage whereas overall colitis has a maximal score of 10.
Histology and microscopic damage score. The colonic segment was fixed in 4% formaldehyde and embedded in paraffin. Morphometric analysis was performed on both hematoxylin–eosin-stained 4-μm transverse sections. The extent of colonic inflammatory damage was assessed using the scoring system described by Ameho et al.45 A score of 0 represents no histological damage, whereas extensive colitis with necrosis into the muscularis propria involving 50% of the specimen has a maximal score of 6.
For eosinophil numeration, a conventional histology staining (hematoxylin and eosin) was used. This method can be used for detection of eosinophils in fixed tissue sections.46 Eosinophils had distinctly orange-to-red granular cytoplasmic staining. Five random fields (90,000 μm2) were observed per colon section, in a blinded manner. Eosinophils were counted per viewing field and averaged for each colon. Scoring criteria for eosinophil infiltration were: 1=occasional eosinophils (average: 1–14); 2=mildly increased eosinophils (average: 15–29); 3=moderately increased eosinophils (average: 30–74); and 4=markedly increased eosinophils (average: >75).
Real-time PCR analysis of colon sections. RNA was extracted from colon samples with NucleoSpin RNA II kit (Macherey-Nagel, Hoerdt, France). cDNA was generated from 1 μg total RNA using a High Capacity cDNA Reverse Transcription Kit (Life Technologies, Saint-Aubin, France).
Real-time quantitative PCR was carried out with the Fast Power SYBR Green PCR master mix (Life Technologies) according to the manufacturer’s instructions. The analyses were performed with an ABI StepOnePlus (Applied Biosystems, Life Technologies). Values were normalized to glyceraldehyde-3 phosphate dehydrogenase. Primers are listed in Table 1.
Cytokine and MPO quantification. Spleen cells were suspended in complete medium at 5 × 106 cells ml−1. Cell cultures were stimulated with Concanavalin A (5 μg ml−1) or P28GST (20 μg ml−1). After 72 h at 37 °C, culture supernatants were harvested and assayed for cytokine concentration.
Cytokine levels in the spleen cell supernatants were measured by Luminex assay as recommended by the manufacturer (Milliplex, Millipore, Molsheim, France).
MPO levels were measured in colonic specimens using an Enzyme-Linked Immunosorbent Assay Kit according to the manufacturer’s instructions (Hycult Biotech, Clinisciences, Nanterre, France). Results are expressed as MPO (ng) per protein (mg) ratios.
Statistical analysis. The graphs and statistical analyses were generated using GraphPad Prism 5 (Graph Pad Software, La Jolla, CA) Statistical significance was calculated using unpaired Mann–Whitney U-test or Kruskal–Wallis one-way analysis of variance when more than two groups were compared. All data are expressed as mean±s.e.m. *P<0.05; **P<0.01;***P<0.001.
References
Ng, S.C. et al. Geographical variability and environmental risk factors in inflammatory bowel disease. Gut 62, 630–649 (2013).
Wammes, L., Mpairwe, H., Elliott, A.M. & Yazdanbakhsh, M. Helminth therapy or elimination: epidemiological, immunological, and clinical considerations. Lancet Infect. Dis. 14, 1150–1162 (2014).
Weinstock, J.V. & Elliott, D.E. Helminths and the IBD hygiene hypothesis. Inflamm. Bowel Dis. 15, 128–133 (2009).
Strober, W. & Fuss, I.J. Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology 140, 1756–1767 (2011).
Gause, W.C., Wynn, T.A. & Allen, J.E. Type 2 immunity and wound healing: evolutionary refinement of adaptive immunity by helminths. Nat. Rev. Immunol. 13, 607–614 (2013).
Mishra, P.K., Palma, M., Bleich, D., Loke, P. & Gause, W.C. Systemic impact of intestinal helminth infections. Mucosal Immunol. 7, 753–762 (2014).
Nussbaum, J.C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Maizels, R.M. et al. Helminth parasites—masters of regulation. Immunol. Rev. 201, 89–116 (2004).
Maizels, R.M., McSorley, H.J. & Smyth, D.J. Helminths in the hygiene hypothesis: sooner or later? Clin. Exp. Immunol. 177, 38–46 (2014).
Brunet, L.R., Dunne, D.W. & Pearce, E.J. Cytokine interaction and immune responses during Schistosoma mansoni infection. Parasitol. Today 14, 422–427 (1998).
Brunet, L.R., Finkelman, F.D., Cheever, A.W., Kopf, M.A. & Pearce, E.J. IL-4 protects against TNF-alpha-mediated cachexia and death during acute schistosomiasis. J. Immunol. 159, 777–785 (1997).
Herbert, D.R. et al. Alternative macrophage activation is essential for survival during schistosomiasis and downmodulates T helper 1 responses and immunopathology. Immunity 20, 623–635 (2004).
Cooke, A. et al. Infection with Schistosoma mansoni prevents insulin dependent diabetes mellitus in non-obese diabetic mice. Parasite Immunol. 21, 169–176 (1999).
Kuijk, L.M. et al. Soluble helminth products suppress clinical signs in murine experimental autoimmune encephalomyelitis and differentially modulate human dendritic cell activation. Mol. Immunol. 51, 210–218 (2012).
Moreels, T.G. et al. Concurrent infection with Schistosoma mansoni attenuates inflammation induced changes in colonic morphology, cytokine levels, and smooth muscle contractility of trinitrobenzene sulphonic acid induced colitis in rats. Gut 53, 99–107 (2004).
Everts, B. et al. Schistosome-derived omega-1 drives Th2 polarization by suppressing protein synthesis following internalization by the mannose receptor. J. Exp. Med. 209, 1753–1767 S1751 (2012).
Heylen, M. et al. Of worms, mice and man: an overview of experimental and clinical helminth-based therapy for inflammatory bowel disease. Pharmacol. Ther. 143, 153–167 (2014).
Balloul, J.M. et al. Molecular cloning of a protective antigen of schistosomes. Nature 326, 149–153 (1987).
Johnson, K.A. et al. Crystal structure of the 28 kDa glutathione S-transferase from Schistosoma haematobium. Biochemistry 42, 10084–10094 (2003).
Boulanger, D. et al. Vaccine potential of a recombinant glutathione S-transferase cloned from Schistosoma haematobium in primates experimentally infected with an homologous challenge. Vaccine 17, 319–326 (1999).
Lebens, M. et al. A mucosally administered recombinant fusion protein vaccine against schistosomiasis protecting against immunopathology and infection. Vaccine 21, 514–520 (2003).
Sun, J.B. et al. Intranasal administration of a Schistosoma mansoni glutathione S-transferase-cholera toxoid conjugate vaccine evokes antiparasitic and antipathological immunity in mice. J. Immunol. 163, 1045–1052 (1999).
Capron, A., Riveau, G., Capron, M. & Trottein, F. Schistosomes: the road from host-parasite interactions to vaccines in clinical trials. Trends Parasitol. 21, 143–149 (2005).
Riveau, G. et al. Safety and immunogenicity of rSh28GST antigen in humans: phase 1 randomized clinical study of a vaccine candidate against urinary schistosomiasis. PLoS Negl. Trop. Dis. 6, e1704 (2012).
Khan, C.M. et al. Construction, expression, and immunogenicity of the Schistosoma mansoni P28 glutathione S-transferase as a genetic fusion to tetanus toxin fragment C in a live Aro attenuated vaccine strain of Salmonella. Proc. Natl. Acad. Sci. USA 91, 11261–11265 (1994).
Mielcarek, N. et al. Homologous and heterologous protection after single intranasal administration of live attenuated recombinant Bordetella pertussis. Nat. Biotechnol. 16, 454–457 (1998).
Renauld-Mongenie, G. et al. Induction of mucosal immune responses against a heterologous antigen fused to filamentous hemagglutinin after intranasal immunization with recombinant Bordetella pertussis. Proc. Natl. Acad. Sci. USA 93, 7944–7949 (1996).
Wirtz, S., Neufert, C., Weigmann, B. & Neurath, M.F. Chemically induced mouse models of intestinal inflammation. Nat. Protoc. 2, 541–546 (2007).
Zimmermann, N. et al. Siglec-F antibody administration to mice selectively reduces blood and tissue eosinophils. Allergy 63, 1156–1163 (2008).
Weinstock, J.V. & Elliott, D.E. Translatability of helminth therapy in inflammatory bowel diseases. Int. J. Parasitol. 43, 245–251 (2012).
Weinstock, J.V. & Elliott, D.E. Helminth infections decrease host susceptibility to immune-mediated diseases. J. Immunol. 193, 3239–3247 (2014).
Jouvin, M.H. & Kinet, J.P. Trichuris suis ova: testing a helminth-based therapy as an extension of the hygiene hypothesis. J. Allergy Clin. Immunol. 130, 3–10 (2012).
McSorley, H.J., Hewitson, J.P. & Maizels, R.M. Immunomodulation by helminth parasites: defining mechanisms and mediators. Int. J. Parasitol. 43, 301–310 (2013).
de Jesus, A.R. et al. Clinical and immunologic evaluation of 31 patients with acute schistosomiasis mansoni. J. Infect. Dis. 185, 98–105 (2012).
Joseph, S. et al. Cytokine production in whole blood cultures from a fishing community in an area of high endemicity for Schistosoma mansoni in Uganda: the differential effect of parasite worm and egg antigens. Infect. Immun. 72, 728–734 (2004).
Malaquias, L.C. et al. Cytokine regulation of human immune response to Schistosoma mansoni: analysis of the role of IL-4, IL-5 and IL-10 on peripheral blood mononuclear cell responses. Scand. J. Immunol. 46, 393–398 (1997).
Korsholm, K.S., Petersen, R.V., Agger, E.M. & Andersen, P. T-helper 1 and T-helper 2 adjuvants induce distinct differences in the magnitude, quality and kinetics of the early inflammatory response at the site of injection. Immunology 129, 75–86 (2010).
Rosenberg, H.F., Dyer, K.D. & Foster, P.S. Eosinophils: changing perspectives in health and disease. Nat. Rev. Immunol. 13, 9–22 (2013).
Jung, Y. & Rothenberg, M.E. Roles and regulation of gastrointestinal eosinophils in immunity and disease. J. Immunol. 193, 999–1005 (2014).
Hogan, S.P., Waddell, A. & Fulkerson, P.C. Eosinophils in infection and intestinal immunity. Curr. Opin. Gastroenterol. 29, 7–14 (2013).
Chu, V.T. et al. Eosinophils promote generation and maintenance of immunoglobulin-A-expressing plasma cells and contribute to gut immune homeostasis. Immunity 40, 582–593 (2014).
Masterson, J.C. et al. Eosinophil-mediated signalling attenuates inflammatory responses in experimental colitis. Gut pii: gutjnl-2014-306998 (2014); doi: 10.1136/gutjnl-2014-306998.
Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 332, 243–247 (2011).
Wallace, J.L., MacNaughton, W.K., Morris, G.P. & Beck, P.L. Inhibition of leukotriene synthesis markedly accelerates healing in a rat model of inflammatory bowel disease. Gastroenterology 96, 29–36 (1989).
Ameho, C.K. et al. Prophylactic effect of dietary glutamine supplementation on interleukin 8 and tumour necrosis factor alpha production in trinitrobenzene sulphonic acid induced colitis. Gut 41, 487–493 (1997).
Arantes-Costa, F. et al. Effects of residual oil fly ash (ROFA) in mice with chronic allergic pulmonary inflammation. Toxicol. Pathol. 36, 680–686 (2008).
Acknowledgements
We thank Sylvie Loiseau (Université de Lille, Inserm U995, Lille, France) for performing parasite infection experiments in rats. Furthermore, we thank Anne-Marie Schacht (Lille Pasteur Institute, Inserm U1019, Lille, France) for control procedures of the P28GST batch. This study was supported by ARCIR (Conseil Regional Nord Pas de Calais), ANR (2011 RPIB 021 01), and SATT (project ACROSS).We also acknowledge the support of the DigestScience Foundation.
Author contributions
VD designed, performed, and interpreted most experiments and prepared the manuscript. MEN and MD performed the rat experiments. LD supervised the colitis animal models. CR, CD, AS, SG, and AD participated in rats and mice experiments. GR, JFC, and PD contributed to experimental design. MC initiated and supervised all aspects of this study. All authors participated in revising the manuscript and agreed to the final version.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declared no conflict of interest.
Additional information
SUPPLEMENTARY MATERIAL is linked to the online version of the paper
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Driss, V., El Nady, M., Delbeke, M. et al. The schistosome glutathione S-transferase P28GST, a unique helminth protein, prevents intestinal inflammation in experimental colitis through a Th2-type response with mucosal eosinophils. Mucosal Immunol 9, 322–335 (2016). https://doi.org/10.1038/mi.2015.62
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mi.2015.62
This article is cited by
-
Eosinophils and helminth infection: protective or pathogenic?
Seminars in Immunopathology (2021)
-
Potential application of helminth therapy for resolution of neuroinflammation in neuropsychiatric disorders
Metabolic Brain Disease (2020)
-
Immunity by equilibrium
Nature Reviews Immunology (2016)