Abstract
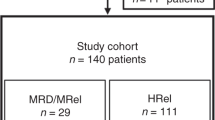
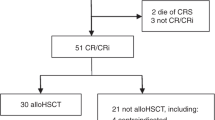
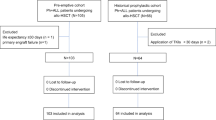
Tyrosine kinase inhibitors (TKIs) directed against the ABL kinase are now used routinely during frontline therapy for Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL) and result in hematologic remission rates exceeding 90%. Minimal residual disease levels are generally lower when TKIs are used in combination with chemotherapy rather than as monotherapy. Although outcome has improved substantially with TKI-based regimens compared with historic controls, allogeneic stem cell transplantation (SCT) in first remission provides the best chance of cure for the majority of patients eligible for SCT. Administration of imatinib after SCT further reduces molecular recurrence and is associated with greatly improved relapse-free and overall survival. The high relapse rate in non-transplanted patients is largely attributable to the emergence of leukemic clones with mutations in the tyrosine kinase domain of BCR-ABL. Ongoing studies with newer TKIs will determine whether these more potent agents are able to sustain remissions without SCT. Assessment of minimal residual disease has become an integral part of the management of Ph+ALL, as it has prognostic importance and is used to guide therapeutic intervention. Novel immunotherapeutic interventions and combinations of TKIs are currently being investigated in clinical trials and may further improve the prognosis of patients with Ph+ALL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ottmann OG, Wassmann B, Pfeifer H, Giagounidis A, Stelljes M, Dührsen U et al. Imatinib compared with chemotherapy as front-line treatment of elderly patients with Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL). Cancer 2007; 109: 2068–2076.
Vignetti M, Fazi P, Cimino G, Martinelli G, Di Raimondo F, Ferrara F et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: results of the Gruppo Italiano Malattie Ematologiche dell’Adulto (GIMEMA) LAL0201-B protocol. Blood 2007; 109: 3676–3678.
Rea D, Legros L, Raffoux E, Thomas X, Turlure P, Maury S et al. High-dose imatinib mesylate combined with vincristine and dexamethasone (DIV regimen) as induction therapy in patients with resistant Philadelphia-positive acute lymphoblastic leukemia and lymphoid blast crisis of chronic myeloid leukemia. Leukemia 2006; 20: 400–403.
Ravandi F, O’Brien S, Thomas DA, Kantarjian HM, Faderl S, Jones D et al. Phase II study of combination of hyperCVAD with dasatinib in frontline therapy of patients with Philadelphia chromosome (Ph) positive acute lymphoblastic leukemia (ALL). Blood 2008; 112: 1005a (abstract 2921).
Rousselot P, Cayuela J-M, Recher C, Leguay T, Salanoubat C, Witz F et al. Dasatinib (Sprycel®) and chemotherapy for first-line treatment in elderly patients with de novo Philadelphia positive ALL: results of the first 22 patients included in the EWALL-Ph-01 trial (on behalf of the European Working Group on Adult ALL (EWALL)). Blood (ASH Annu Meet Abstr) 2008; 112: 2920.
Foà R, Vitale A, Vignetti M, Meloni G, Guarini A, De Propris MS et al. Dasatinib as first-line treatment for adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood 2011; 118: 6521–6528.
Ottmann OG, Pfeifer H . First-line treatment of Philadelphia chromosome-positive acute lymphoblastic leukaemia in adults. Curr Opin Oncol 2009; 21 (Suppl 1): S43–S46.
Wassmann B, Scheuring U, Pfeifer H, Binckebanck A, Käbisch A, Lübbert M et al. Efficacy and safety of imatinib mesylate (Glivec) in combination with interferon-alpha (IFN-alpha) in Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL). Leukemia 2003; 17: 1919–1924.
Rousselot P, Huguet F, Vey N, Bouabdallah K, Delaunay J, Maury S et al. Maintenance therapy by Glivec® and Pegasys® in patients with Philadelphia positive acute lymphocytic leukemia not eligible for hematopoietic stem cell transplantation. Blood (ASH Annu Meet Abstr) 2007; 110: 2812.
Pane F, Cimino G, Izzo B, Camera A, Vitale A, Quintarelli C et al. Significant reduction of the hybrid BCR/ABL transcripts after induction and consolidation therapy is a powerful predictor of treatment response in adult Philadelphia-positive acute lymphoblastic leukemia. Leukemia 2005; 19: 628–635.
Wassmann B, Pfeifer H, Stadler M, Bornhaüser M, Bug G, Scheuring UJ et al. Early molecular response to posttransplantation imatinib determines outcome in MRD+ Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood 2005; 106: 458–463.
Mullighan CG, Goorha S, Radtke I, Miller CB, Coustan-Smith E, Dalton JD et al. Genome-wide analysis of genetic alterations in acute lymphoblastic leukaemia. Nature 2007; 446: 758–764.
Iacobucci I, Ottaviani E, Astolfi A, Lonetti A, Testoni N. High-resolution genomic profiling of Ph-positive acute lymphoblastic leukaemia (ALL) identified recurrent copy number anomalies in genes regulating the cell cycle and the B-cell differentiation. Haematologica 2008; 93: 1084.
Soverini S, Colarossi S, Gnani A, Rosti G, Castagnetti F, Poerio A et al. Contribution of ABL kinase domain mutations to imatinib resistance in different subsets of Philadelphia-positive patients: by the GIMEMA Working Party on Chronic Myeloid Leukemia. Clin Cancer Res 2006; 12: 7374–7379.
Branford S, Rudzki Z, Walsh S, Grigg A, Arthur C, Taylor K et al. High frequency of point mutations clustered within the adenosine triphosphate-binding region of BCR/ABL in patients with chronic myeloid leukemia or Ph-positive acute lymphoblastic leukemia who develop imatinib (STI571) resistance. Blood 2002; 99: 3472–3475.
Pfeifer H, Wassmann B, Pavlova A, Wunderle L, Oldenburg J, Binckebanck A et al. Kinase domain mutations of BCR-ABL frequently precede imatinib-based therapy and give rise to relapse in patients with de novo Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood 2007; 110: 727–734.
Soverini S, Martinelli G, Vitale A, Gnani A, Colarossi S, Paolini S et al. Philadelphia-positive acute lymphoblastic leukemia patients already harbor Bcr-Abl kinase domain mutations at low levels at the time of diagnosis—a report by the GIMEMA ALL Working Party. Blood (ASH Annu Meet Abstr) 2008; 112: 722.
Redaelli S, Piazza R, Rostagno R, Magistroni V, Perini P, Marega M et al. Activity of bosutinib, dasatinib, and nilotinib against 18 imatinib-resistant BCR/ABL mutants. J Clin Oncol 2009; 27: 469–471.
Cortes JE, Kim D-W, Pinilla-Ibarz J, Le Coutre PD, Chuah C, Nicolini FE et al. Ponatinib initial findings from the PACE trial: a pivotal phase 2 study of ponatinib in patients with CML and Ph+ ALL resistant or intolerant to dasatinib or nilotinib, or with the t315i mutation. Blood (ASH Annu Meet Abstr) 2011; 118: 109.
Ottmann O, Dombret H, Martinelli G, Simonsson B, Guilhot F, Larson RA et al. Dasatinib induces rapid hematologic and cytogenetic responses in adult patients with Philadelphia chromosome positive acute lymphoblastic leukemia with resistance or intolerance to imatinib: interim results of a phase 2 study. Blood 2007; 110: 2309–2315.
Topp MS, Kufer P, Gökbuget N, Goebeler M, Klinger M, Neumann S et al. Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J Clin Oncol 2011; 29: 2493–2498.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author received consulting fees for Advisory Board participation and lecture fees from Novartis and Bristol Myers Squibb.
Additional information
This article was published as part of a supplement that was supported by Novartis, MSD Italia, Roche, Celgene, GlaxoSmithKline, Sanofi, Gilead, Adienne, Italfarmaco, Pierre Fabre Pharmaceuticals with an unrestricted educational contribution to AREO—Associazione Ricerche Emato-Oncologiche (Genoa) and AMS—Associazione Malattie del Sangue (Milan) for the purpose of advancing research in acute and chronic leukemia.
Rights and permissions
About this article
Cite this article
Ottmann, O. Management of Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia Suppl 1 (Suppl 2), S7–S9 (2012). https://doi.org/10.1038/leusup.2012.7
Published:
Issue Date:
DOI: https://doi.org/10.1038/leusup.2012.7
Keywords
This article is cited by
-
Leukemia-propagating cells demonstrate distinctive gene expression profiles compared with other cell fractions from patients with de novo Philadelphia chromosome-positive ALL
Annals of Hematology (2018)
-
Phase II study of nilotinib in patients with relapsed or refractory Philadelphia chromosome—positive acute lymphoblastic leukemia
Leukemia (2013)