Abstract
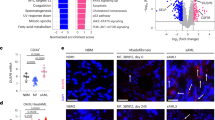
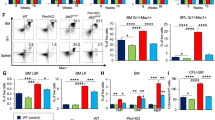
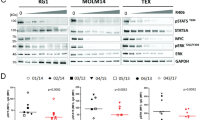
Activating mutations, such as E76K and D61Y, in PTPN11 (SHP2), a protein tyrosine phosphatase implicated in multiple cell signaling processes, are associated with 35% of patients with juvenile myelomonocytic leukemia (JMML), an aggressive childhood myeloproliferative neoplasm (MPN). Here we show that the interaction between leukemia-associated mutant Shp2 and Gab2, a scaffolding protein important for cytokine-induced PI3K/Akt signaling, was enhanced, and that the mTOR pathway was elevated in Ptpn11E76K/+ leukemic cells. Importantly, MPN induced by the Ptpn11E76K/+ mutation was markedly attenuated in Ptpn11E76K/+/Gab2−/− double mutant mice—overproduction of myeloid cells was alleviated, splenomegaly was diminished and myeloid cell infiltration in nonhematopoietic organs was decreased in these double mutants. Excessive myeloid differentiation of stem cells was also normalized by depletion of Gab2. Acute leukemia progression of MPN was reduced in the double mutant mice and, as such, their survival was much prolonged. Furthermore, treatment of Ptpn11E76K/+ mice with Rapamycin, a specific and potent mTOR inhibitor, mitigated MPN phenotypes. Collectively, this study reveals an important role of the Gab2/PI3K/mTOR pathway in mediating the pathogenic signaling of the PTPN11 gain-of-function mutations and a therapeutic potential of Rapamycin for PTPN11 mutation-associated JMML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Birnbaum RA, O'Marcaigh A, Wardak Z, Zhang YY, Dranoff G, Jacks T et al. Nf1 and Gmcsf interact in myeloid leukemogenesis. Mol Cell 2000; 5: 189–195.
Emanuel PD, Bates LJ, Castleberry RP, Gualtieri RJ, Zuckerman KS . Selective hypersensitivity to granulocyte-macrophage colony-stimulating factor by juvenile chronic myeloid leukemia hematopoietic progenitors. Blood 1991; 77: 925–929.
Chang TY, Dvorak CC, Loh ML . Bedside to bench in juvenile myelomonocytic leukemia: insights into leukemogenesis from a rare pediatric leukemia. Blood 2014; 124: 2487–2497.
Liu X, Sabnis H, Bunting KD, Qu CK . Molecular targets for the treatment of juvenile myelomonocytic leukemia. Adv Hematol 2012; 2012: 308252.
Emanuel PD . Juvenile myelomonocytic leukemia and chronic myelomonocytic leukemia. Leukemia 2008; 22: 1335–1342.
Loh ML, Vattikuti S, Schubbert S, Reynolds MG, Carlson E, Lieuw KH et al. Mutations in PTPN11 implicate the SHP-2 phosphatase in leukemogenesis. Blood 2004; 103: 2325–2331.
Tartaglia M, Niemeyer CM, Fragale A, Song X, Buechner J, Jung A et al. Somatic mutations in PTPN11 in juvenile myelomonocytic leukemia, myelodysplastic syndromes and acute myeloid leukemia. Nat Genet 2003; 34: 148–150.
Loh ML, Sakai DS, Flotho C, Kang M, Fliegauf M, Archambeault S et al. Mutations in CBL occur frequently in juvenile myelomonocytic leukemia. Blood 2009; 114: 1859–1863.
Muramatsu H, Makishima H, Jankowska AM, Cazzolli H, O'Keefe C, Yoshida N et al. Mutations of an E3 ubiquitin ligase c-Cbl but not TET2 mutations are pathogenic in juvenile myelomonocytic leukemia. Blood 2010; 115: 1969–1975.
Zhang Y, Taylor BR, Shannon K, Clapp DW . Quantitative effects of Nf1 inactivation on in vivo hematopoiesis. J Clin Invest 2001; 108: 709–715.
Chan IT, Kutok JL, Williams IR, Cohen S, Kelly L, Shigematsu H et al. Conditional expression of oncogenic K-ras from its endogenous promoter induces a myeloproliferative disease. J Clin Invest 2004; 113: 528–538.
Braun BS, Tuveson DA, Kong N, Le DT, Kogan SC, Rozmus J et al. Somatic activation of oncogenic Kras in hematopoietic cells initiates a rapidly fatal myeloproliferative disorder. Proc Natl Acad Sci USA 2004; 101: 597–602.
Araki T, Mohi MG, Ismat FA, Bronson RT, Williams IR, Kutok JL et al. Mouse model of Noonan syndrome reveals cell type- and gene dosage-dependent effects of Ptpn11 mutation. Nat Med 2004; 10: 849–857.
Chan RJ, Leedy MB, Munugalavadla V, Voorhorst CS, Li Y, Yu M et al. Human somatic PTPN11 mutations induce hematopoietic-cell hypersensitivity to granulocyte-macrophage colony-stimulating factor. Blood 2005; 105: 3737–3742.
Chan G, Kalaitzidis D, Usenko T, Kutok JL, Yang W, Mohi MG et al. Leukemogenic Ptpn11 causes fatal myeloproliferative disorder via cell-autonomous effects on multiple stages of hematopoiesis. Blood 2009; 113: 4414–4424.
Xu D, Liu X, Yu WM, Meyerson HJ, Guo C, Gerson SL et al. Non-lineage/stage-restricted effects of a gain-of-function mutation in tyrosine phosphatase Ptpn11 (Shp2) on malignant transformation of hematopoietic cells. J Exp Med 2011; 208: 1977–1988.
Mohi MG, Williams IR, Dearolf CR, Chan G, Kutok JL, Cohen S et al. Prognostic, therapeutic, and mechanistic implications of a mouse model of leukemia evoked by Shp2 (PTPN11) mutations. Cancer Cell 2005; 7: 179–191.
Sanada M, Suzuki T, Shih LY, Otsu M, Kato M, Yamazaki S et al. Gain-of-function of mutated C-CBL tumour suppressor in myeloid neoplasms. Nature 2009; 460: 904–908.
Chan G, Kalaitzidis D, Neel BG . The tyrosine phosphatase Shp2 (PTPN11) in cancer. Cancer Metastasis Rev 2008; 27: 179–192.
Nabinger SC, Chan RJ . Shp2 function in hematopoietic stem cell biology and leukemogenesis. Curr Opin Hematol 2012; 19: 273–279.
Xu D, Qu CK . Protein tyrosine phosphatases in the JAK/STAT pathway. Front Biosci 2008; 13: 4925–4932.
Eck MJ, Pluskey S, Trub T, Harrison SC, Shoelson SE . Spatial constraints on the recognition of phosphoproteins by the tandem SH2 domains of the phosphatase SH-PTP2. Nature 1996; 379: 277–280.
Hof P, Pluskey S, Dhe-Paganon S, Eck MJ, Shoelson SE . Crystal structure of the tyrosine phosphatase SHP-2. Cell 1998; 92: 441–450.
Keilhack H, David FS, McGregor M, Cantley LC, Neel BG . Diverse biochemical properties of Shp2 mutants. Implications for disease phenotypes. J Biol Chem 2005; 280: 30984–30993.
Goodwin CB, Li XJ, Mali RS, Chan G, Kang M, Liu Z et al. PI3K p110delta uniquely promotes gain-of-function Shp2-induced GM-CSF hypersensitivity in a model of JMML. Blood 2014; 123: 2838–2842.
Gritsman K, Yuzugullu H, Von T, Yan H, Clayton L, Fritsch C et al. Hematopoiesis and RAS-driven myeloid leukemia differentially require PI3K isoform p110alpha. J Clin Invest 2014; 124: 1794–1809.
Rodriguez-Viciana P, Warne PH, Dhand R, Vanhaesebroeck B, Gout I, Fry MJ et al. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 1994; 370: 527–532.
Gu H, Neel BG . The 'Gab' in signal transduction. Trends Cell Biol 2003; 13: 122–130.
Nishida K, Wang L, Morii E, Park SJ, Narimatsu M, Itoh S et al. Requirement of Gab2 for mast cell development and KitL/c-Kit signaling. Blood 2002; 99: 1866–1869.
Zhang Y, Diaz-Flores E, Li G, Wang Z, Kang Z, Haviernikova E et al. Abnormal hematopoiesis in Gab2 mutant mice. Blood 2007; 110: 116–124.
Yu WM, Daino H, Chen J, Bunting KD, Qu CK . Effects of a Leukemia-associated Gain-of-Function Mutation of SHP-2 Phosphatase on Interleukin-3 Signaling. J Biol Chem 2006; 281: 5426–5434.
Yu WM, Hawley TS, Hawley RG, Qu CK . Catalytic-dependent and -independent roles of SHP-2 tyrosine Phosphatase in interleukin-3 signaling. Oncogene 2003; 22: 5995–6004.
Liu YL, Castleberry RP, Emanuel PD . PTEN deficiency is a common defect in juvenile myelomonocytic leukemia. Leuk Res 2009; 33: 671–677.
Acknowledgements
We are grateful to Dr Toshio Hirano for Gab2+/− mice. This work was supported by National Institutes of Health grants HL130995 and DK092722, and a Hyundai Hope on Wheels scholar grant (to CKQ).
Author contributions
WL, WMY, and JZ conducted the research and summarized the data. RJC, MLL, ZZ, and KDB provided critical reagents, discussed the work and edited the manuscript. CKQ designed the experiments and provided technical training to the first three authors. WL and CKQ wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, W., Yu, WM., Zhang, J. et al. Inhibition of the Gab2/PI3K/mTOR signaling ameliorates myeloid malignancy caused by Ptpn11 (Shp2) gain-of-function mutations. Leukemia 31, 1415–1422 (2017). https://doi.org/10.1038/leu.2016.326
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.326
This article is cited by
-
Identification of a novel fusion gene, RARA::ANKRD34C, in acute promyelocytic leukemia
Annals of Hematology (2024)
-
Gab2 deficiency prevents Flt3-ITD driven acute myeloid leukemia in vivo
Leukemia (2022)
-
From Stem to Sternum: The Role of Shp2 in the Skeleton
Calcified Tissue International (2022)
-
Pediatric Neoplasms Presenting with Monocytosis
Current Hematologic Malignancy Reports (2021)
-
Molecular insights into phytochemicals exhibiting anti-arthritic activity: systematic review
Inflammation Research (2021)