Abstract
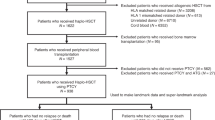
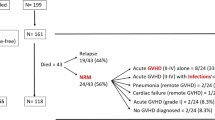
Chronic graft-versus-host disease (GVHD) remains a serious complication after allogeneic hematopoietic stem cell transplantation (HCT). In 2005 the National Institutes of Health (NIH) established new criteria for chronic GVHD based on retrospective data and expert recommendations. We prospectively evaluated the incidence of NIH-defined chronic GVHD and its prognostic impact in 178 consecutive patients. The cumulative incidence of chronic GVHD at 3 years was 64, 48 and 16% for chronic classic GVHD and overlap syndrome. Prior acute GVHD and myeloablative conditioning were significantly associated with increased risk of chronic GVHD. Three-year survival (overall survival (OS)) for late-acute GVHD, chronic classic and overlap chronic GVHD when assigned on day 100 were 69, 83 and 73%. OS was significantly worse for patients with platelet counts below 100 g/l at onset of chronic GVHD (35% versus 86%, P<0.0001) and progressive as compared with de novo and quiescent onset of chronic GVHD (54.5% versus 89.5% versus 84%, P=0.022 and 0.001). Peak severity of chronic GVHD had no impact on non-relapse mortality (NRM) and OS. Recurrent acute GVHD, platelet counts below 100 g/l at diagnosis of chronic GVHD, progressive onset of chronic GVHD and advanced disease stage prior to HCT were significantly associated with increased NRM. This prospective analysis provides for the first-time data on the incidence rates of NIH-defined chronic GVHD categories and identified risk factors for the occurrence of chronic GVHD. A prognostic value of thrombocytopenia and progressive onset type of chronic GVHD for survival after HCT was observed in NIH-defined chronic GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lee SJ, Vogelsang G, Flowers MED . Chronic graft-versus-host disease. Biol Blood Marrow Transplant 2003; 9: 215–233.
Vogelsang GB, Higman MA . Chronic graft versus host disease. Br J Haematol 2004; 125: 435–454.
Socie G, Stone JV, Wingard JR, Weisdorf D, Henslee-Downey PJ, Bredeson C et al. Long-term survival and late deaths after allogeneic bone marrow transplantation. Late Effects Working Committee of the International Bone Marrow Transplant Registry. N Engl J Med 1999; 341: 14–21.
Syrjala KL, Chapko MK, Vitaliano PP, Cummings C, Sullivan KM . Recovery after allogeneic marrow transplantation: prospective study of predictors of long-term physical and psychosocial functioning. Bone Marrow Transplant 1993; 11: 319–327.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease. I. Diagnosis and staging working group report. Biol Blood Marrow Transplant 2005; 11: 945–956.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE et al. Chronic graft-versus-host syndrome in man: a long-term clinicopathologic study of 20 Seattle patients. Am J Med 1980; 69: 204–217.
Sullivan KM . Graft-versus-host disease. In: Thomas ED, Blume KG, Forman SJ (eds). Hematopoietic Cell Transplantation. Blackwell Sciences: Malden, MA, 1999, pp 515–536.
Jagasia M, Giglia J, Chinratanalab W, Dixon S, Chen H, Frangoul H et al. Incidence and outcome of chronic graft-versus-host disease using National Institutes of Health Consensus criteria. Biol Blood Marrow Transplant 2007; 13: 1207–1215.
Vigorito AC, Campregher PV, Storer BE, Carpenter PA, Moravec CK, Kiem HP et al. Evaluation of NIH consensus criteria for classification of late acute and chronic GVHD. Blood 2009; 114: 702–708.
Arora M, Nagaraj S, Witte J, DeFor TE, MacMillan M, Burns LJ et al. New classification of chronic GVHD: added clarity from the consensus diagnoses. Bone Marrow Transplant 2009; 43: 149–153.
Cho BS, Min CK, Eom KS, Kim YJ, Kim HJ, Lee S et al. Feasibility of NIH consensus criteria for chronic graft-versus-host disease. Leukemia 2009; 23: 78–84.
Kim DY, Lee JH, Lee JH, Kim SH, Lim SN, Kim SD et al. Reevaluation of the National Institutes of Health criteria for classification and scoring of chronic GVHD. Bone Marrow Transplant 2009; 45: 1174–1180.
Couriel DR, Saliba R, Escalon MP, Hsu Y, Ghosh S, Ippoliti C et al. Sirolimus in combination with tacrolimus and corticosteroids for the treatment of resistant chronic graft-versus-host disease. Br J Haematol 2005; 130: 409–417.
Schmid C, Schleuning M, Schwerdtfeger R, Hertenstein B, Mischak-Weissinger E, Bunjes D et al. Long-term survival in refractory acute myeloid leukemia after sequential treatment with chemotherapy and reduced-intensity conditioning for allogeneic stem cell transplantation. Blood 2006; 108: 1092–1099.
Tomblyn M, Chiller T, Einsele H, Gress R, Sepkowitz K, Storek J . Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant 2009; 15: 1143–1238.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. Consensus conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Fine JP, Gray RJ . A proportional hazards model for the subdistribution of competing risk. J Am Stat Assoc 1999; 94: 496–509.
Perez-Simon JA, Encinas C, Silva F, Arcos MJ, Diez-Campelo M, Sanchez-Guijo FM et al. Prognostic factors of chronic graft-versus-host disease following allogeneic peripheral blood stem cell transplantation: the National Institutes of Health scale plus the type of onset can predict survival rates and duration of immunosuppressive therapy. Biol Blood Marrow Transplant 2008; 14: 1163–1171.
Thepot S, Zhou J, Perrot A, Robin M, Xhaard A, de Latour RP et al. The graft-versus-leukemia effect is mainly restricted to NIH-defined chronic graft-versus-host disease after reduced intensity conditioning before allogeneic stem cell transplantation. Leukemia 2010; 24: 1852–1858.
Arai S, Jagasia M, Storer B, Chai X, Pidala J, Cutlrt C et al. Global and organ-specific chronic graft-versus-host disease severity according to the 2005 NIH consensus criteria. Blood 2011; e-pub ahead of print 26 July 2011.
Storb R, Prentice RL, Sullivan KM, Shulman HM, Deeg HJ, Doney KC et al. Predictive factors in chronic graft-versus-host disease in patients with aplastic anemia treated by marrow transplantation from HLA-identical siblings. Ann Intern Med 1983; 98: 461–466.
Carlens S, Ringden O, Remberger M, Lonnqvist B, Hagglund H, Klaesson S et al. Risk factors for chronic graft-versus-host disease after bone marrow transplantation: a retrospective single centre analysis. Bone Marrow Transplant 1998; 22: 755–761.
Flowers MED, Inamoto Y, Carpenter PA, Lee SJ, Kiem HP, Petersdorf EW et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to National Institutes of Health consensus criteria. Blood 2011; 117: 3214–3219.
Lee SJ, Klein JP, Barrett AJ, Ringden O, Antin JH, Cahn JY et al. Severity of chronic graft-versus-host disease: association with treatment-related mortality and relapse. Blood 2002; 100: 406–414.
Greinix HT, Fae I, Schneider B, Rosenmayr A, Mitterschiffthaler A, Pelzmann B et al. Impact of HLA class I high-resolution mismatches on chronic graft-versus-host disease and survival of patients given hematopoietic stem cell grafts from unrelated donors. Bone Marrow Transplant 2005; 35: 57–62.
Finke J, Bethge WA, Schmoor C, Ottinger HD, Stelljes M, Zander AR et al. Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: a randomised, open-label, multicentre phase 3 trial. Lancet Oncol 2009; 10: 855–864.
Soiffer RJ, LeRademacher J, Ho V, Kan F, Artz A, Champlin RE et al. Impact of immune modulation with anti-T-cell antibodies on the outcome of reduced-intensity allogeneic hematopoietic stem cell transplantation for hematologic malignancies. Blood 2011; 117: 6963–6970.
Cutler C, Giri S, Jeyapalan S, Paniagua D, Viswanathan A, Antin JH . Acute and chronic graft-versus-host disease after allogeneic peripheral blood stem-cell and bone marrow transplantation: a meta-analysis. J Clin Oncol 2001; 19: 3685–3691.
Zaucha JM, Gooley T, Bensinger WI, Heimfeld S, Chauncey TR, Zaucha R et al. CD34 dose in granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell grafts affects engraftment kinetics and development of extensive chronic graft-versus-host disease after human leukocyte antigen-identical sibling transplantation. Blood 2001; 98: 3221–3227.
Wolff A, Gerbitz A, Ayuk F, Kiani A, Hildebrandt GC, Vogelsang GB et al. Consensus conference on clinical practice in chronic graft-versus-host disease (GVHD): first-line and topical treatment of chronic GVHD. Biol Blood Marrow Transplant 2010; 16: 1611–1628.
Wolff D, Schleuning M, von Harsdorf S, Bacher U, Gerbitz A, Stadler M et al. Consensus conference on clinical practice in chronic GVHD: second-line treatment of chronic graft-versus-host disease. Biol Blood Marrow Transplant 2011; 17: 1–17.
Sullivan KM, Witherspoon RP, Storb R, Weiden P, Flournoy N, Dahlberg S et al. Prednisone and azathioprine compared with prednisone and placebo for treatment of chronic graft-versus-host disease: prognostic influence of prolonged thrombocytopenia after allogeneic marrow transplantation. Blood 1988; 72: 546–554.
Akpek G, Lee SJ, Flowers ME, Pavletic SZ, Arora M, Lee S et al. Performance of a new clinical grading system for chronic graft-versus-host disease: a multi-center study. Blood 2003; 102: 802–809.
Arora M, Burns LJ, Davies SM, MacMillan ML, Defor TE, Miller WJ et al. Chronic graft-versus-host disease: a prospective cohort study. Biol Blood Marrow Transplant 2003; 9: 38–45.
Stewart BL, Storer B, Storek J, Deeg HJ, Storb R, Hansen JA et al. Duration of immunosuppressive treatment for chronic graft-versus-host disease. Blood 2004; 104: 3501–3506.
Pavletic SZ, Smith LM, Bishop MR, Lynch JC, Tarantolo SR, Vose JM et al. Prognostic factors of chronic graft-versus-host disease after allogeneic blood stem-cell transplantation. Am J Hematol 2005; 78: 265–274.
Przepiorka D, Anderlini P, Saliba R, Cleary K, Mehra R, Khouri I et al. Chronic graft-versus-host disease after allogeneic blood stem cell transplantation. Blood 2001; 98: 1695–1700.
Pulanic D, Lozier JN, Pavletic SZ . Thrombocytopenia and hemostatic disorders in chronic graft versus host disease. Bone Marrow Transplant 2009; 44: 393–403.
Arora M, Klein JP, Weisdorf DJ, Hassebroek A, Flowers MED, Cutler CS et al. Chronic GVHD risk score: a Center for International Blood and Marrow Transplant Research analysis. Blood 2011; 117: 6714–6720.
Wingard JR, Piantadosi S, Vogelsang GB, Farmer ER, Jabs DA, Levin S et al. Predictors of death from chronic graft-versus-host disease after bone marrow transplantation. Blood 1989; 74: 1428–1435.
Jagasia MH, Savani BN, Stricklin G, Engelhardt B, Kassim A, Dixon S et al. Classic and overlap chronic graft-versus-host disease (cGVHD) is associated with superior outcome after extracorporeal photopheresis (ECP). Biol Blood Marrow Transplant 2009; 15: 1288–1295.
Inamoto Y, Flowers MED, Lee SJ, Carpenter PA, Warren EH, Deeg HJ et al. Influence of immunosuppressive treatment on risk of recurrent malignancy after allogeneic hematopoietic cell transplantation. Blood 2011; 118: 456–463.
Ishiyama K, Takami A, Shiobara S, Koizumi S, Nakao S . Graft-versus-leukemia effect of allogeneic stem cell transplantation: a Japanese single center study. Haematologica 2004; 89: 887–889.
Baron F, Maris MB, Sandmaier BM, Storer BE, Sorror M, Diaconescu R et al. Graft-versus-tumor effects after allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. J Clin Oncol 2005; 23: 1993–2003.
Pavletic SZ, Martin P, Lee SJ, Mitchell S, Jacobsohn D, Cowen EW et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006; 12: 252–266.
Herzberg PY, Heussner P, Mumm F, Horak M, Hilgendorf I, von Harsdorf S et al. Validation of the Human Activity Profile Questionnaire in patients after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2010; 16: 1707–1717.
The Chronic GVHD Consortium. Rationale and design of the chronic GVHD cohort study: improving outcomes assessment in chronic GVHD. Biol Blood Marrow Transplant 2011; 17: 1114–1120.
Greinix HT, Loddenkemper C, Pavletic SZ, Holler E, Socie G, Lawitschka A et al. Diagnosis and staging of chronic graft-versus-host disease in the clinical practice. Biol Blood Marrow Transplant 2011; 17: 167–175.
Acknowledgements
We thank Dr Peter Bauer from the Institute of Medical Statistics of the Medical University of Vienna for his assistance in statistical analyses. The study was supported by European Commission Grant 037703 STEMDIAGNOSTICS and a grant by Austrotransplant.
Author contributions
HTG designed the research study, analyzed and interpreted the data, and co-authored the manuscript; ZK performed the clinical research, collected and analyzed data; AB, SE, LV and PK performed clinical assessments of chronic GVHD according to the NIH criteria; VP, KK, GS, UJ and JN performed assessments of defined organ manifestations of chronic GVHD according to the NIH consensus criteria; EP performed the statistical analyses; RK and NW were involved in clinical assessments and therapy of patients.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Preliminary data of this manuscript were presented at the Annual Meeting of the European Group for Blood and Marrow Transplantation in March 2010 in Vienna, Austria.
Rights and permissions
About this article
Cite this article
Kuzmina, Z., Eder, S., Böhm, A. et al. Significantly worse survival of patients with NIH-defined chronic graft-versus-host disease and thrombocytopenia or progressive onset type: results of a prospective study. Leukemia 26, 746–756 (2012). https://doi.org/10.1038/leu.2011.257
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.257
Keywords
This article is cited by
-
Ikaros expression is associated with an increased risk of chronic graft-versus-host disease
Scientific Reports (2023)
-
Applying Rituximab During the Conditioning Regimen Prevents Epstein–Barr Virus Infection Following Allogeneic Hematopoietic Stem Cell Transplant in a Children’s Cohort: A Retrospective Case–Control Study
Infectious Diseases and Therapy (2023)
-
Post-transplant multimorbidity index and quality of life in patients with chronic graft-versus-host disease—results from a joint evaluation of a prospective German multicenter validation trial and a cohort from the National Institutes of Health
Bone Marrow Transplantation (2021)
-
Steroid-refractory chronic graft-versus-host disease: treatment options and patient management
Bone Marrow Transplantation (2021)
-
Allogene Blutstammzelltransplantation: Etabliertes und Neues
InFo Hämatologie + Onkologie (2021)