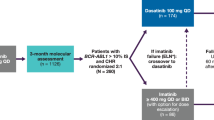
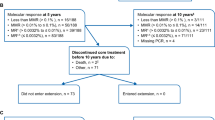
Abstract
In patients with chronic myeloid leukemia (CML), disease in the initial chronic phase (CP) and subsequent progression are driven by the oncogenic activity of the BCR–ABL fusion kinase. Imatinib, a tyrosine kinase inhibitor of BCR–ABL, has been the mainstay of first-line therapy for CML for 10 years. Although patients with CML–CP respond well to imatinib, those who have delayed reductions in leukemic burden during imatinib therapy, such as not achieving a complete cytogenetic response (CCyR) by 12 months, have an increased risk of disease progression. It has been recognized, with 8 years of observation, that patients who achieve an early major molecular response (MMR) on imatinib have a very low probability of disease progression. Recent randomized phase 3 trials have shown that first-line treatment with dasatinib or nilotinib—more potent BCR–ABL inhibitors—results in significantly higher rates and more rapid achievement of CCyR and MMR in comparison with standard-dose imatinib. These trials suggest that CML treatment can be improved with more potent BCR–ABL inhibition during initial therapy, but further follow-up is needed to confirm that the improved response rates with dasatinib and nilotinib are maintained long term.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med 2006; 355: 2408–2417.
Hochhaus A, O’Brien SG, Guilhot F, Druker BJ, Branford S, Foroni L et al. Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia 2009; 23: 1054–1061.
Hughes TP, Hochhaus A, Branford S, Muller MC, Kaeda JS, Foroni L et al. Long-term prognostic significance of early molecular response to imatinib in newly diagnosed chronic myeloid leukemia: an analysis from the international randomized study of interferon versus STI571 (IRIS). Blood 2010; 116: 3758–3765.
Deininger M, O’Brien SG, Guilhot F, Goldman JM, Hochhaus A, Hughes TP et al. International randomized study of interferon vs STI571 (IRIS) 8-year follow up: sustained survival and low risk for progression or events in patients with newly diagnosed chronic myeloid leukemia in chronic phase treated with imatinib. Blood 2009; 114 (Suppl): 462 (abstract 1126).
de Lavallade H, Apperley JF, Khorashad JS, Milojkovic D, Reid AG, Bua M et al. Imatinib for newly diagnosed patients with chronic myeloid leukemia: incidence of sustained responses in an intention-to-treat analysis. J Clin Oncol 2008; 26: 3358–3363.
Lucas CM, Wang L, Austin GM, Knight K, Watmough SJ, Shwe KH et al. A population study of imatinib in chronic myeloid leukaemia demonstrates lower efficacy than in clinical trials. Leukemia 2008; 22: 1963–1966.
Michallet M, Tulliez M, Corm S, Gardembas M, Huguet F, Oukessou A et al. Management of chronic myeloid leukaemia in clinical practice in France: results of the French subset of patients from the UNIC study. Curr Med Res Opin 2010; 26: 307–317.
O’Hare T, Walters DK, Stoffregen EP, Jia T, Manley PW, Mestan J et al. In vitro activity of Bcr-Abl inhibitors AMN107 and BMS-354825 against clinically relevant imatinib-resistant Abl kinase domain mutants. Cancer Res 2005; 65: 4500–4505.
Puttini M, Coluccia AM, Boschelli F, Cleris L, Marchesi E, Donella-Deana A et al. In vitro and in vivo activity of SKI-606, a novel Src-Abl inhibitor, against imatinib-resistant Bcr-Abl+ neoplastic cells. Cancer Res 2006; 66: 11314–11322.
Redaelli S, Piazza R, Rostagno R, Magistroni V, Perini P, Marega M et al. Activity of bosutinib, dasatinib, and nilotinib against 18 imatinib-resistant BCR/ABL mutants. J Clin Oncol 2009; 27: 469–471.
Weisberg E, Manley PW, Breitenstein W, Bruggen J, Cowan-Jacob SW, Ray A et al. Characterization of AMN107, a selective inhibitor of native and mutant Bcr-Abl. Cancer Cell 2005; 7: 129–141.
Boschelli DH, Ye F, Wang YD, Dutia M, Johnson SL, Wu B et al. Optimization of 4-phenylamino-3-quinolinecarbonitriles as potent inhibitors of Src kinase activity. J Med Chem 2001; 44: 3965–3977.
Lombardo LJ, Lee FY, Chen P, Norris D, Barrish JC, Behnia K et al. Discovery of N-(2-chloro-6-methyl-phenyl)-2-(6-(4-(2-hydroxyethyl)-piperazin-1-yl)-2-methylpyrimidin-4-ylamino)thiazole-5-carboxamide (BMS-354825), a dual Src/Abl kinase inhibitor with potent antitumor activity in preclinical assays. J Med Chem 2004; 47: 6658–6661.
Schittenhelm MM, Shiraga S, Schroeder A, Corbin AS, Griffith D, Lee FY et al. Dasatinib (BMS-354825), a dual SRC/ABL kinase inhibitor, inhibits the kinase activity of wild-type, juxtamembrane, and activation loop mutant KIT isoforms associated with human malignancies. Cancer Res 2006; 66: 473–481.
Tokarski JS, Newitt JA, Chang CY, Cheng JD, Wittekind M, Kiefer SE et al. The structure of dasatinib (BMS-354825) bound to activated ABL kinase domain elucidates its inhibitory activity against imatinib-resistant ABL mutants. Cancer Res 2006; 66: 5790–5797.
Laneuville P, DiLea C, Yin OQ, Woodman RC, Mestan J, Manley PW . Comparative in vitro cellular data alone are insufficient to predict clinical responses and guide the choice of BCR-ABL inhibitor for treating imatinib-resistant chronic myeloid leukemia. J Clin Oncol 2010; 28: e169–e171.
Apperley JF . Part I: mechanisms of resistance to imatinib in chronic myeloid leukaemia. Lancet Oncol 2007; 8: 1018–1029.
Cortes J, Jabbour E, Kantarjian H, Yin CC, Shan J, O’Brien S et al. Dynamics of BCR-ABL kinase domain mutations in chronic myeloid leukemia after sequential treatment with multiple tyrosine kinase inhibitors. Blood 2007; 110: 4005–4011.
Hughes T, Saglio G, Branford S, Soverini S, Kim DW, Muller MC et al. Impact of baseline BCR-ABL mutations on response to nilotinib in patients with chronic myeloid leukemia in chronic phase. J Clin Oncol 2009; 27: 4204–4210.
Khorashad JS, Milojkovic D, Mehta P, Anand M, Ghorashian S, Reid AG et al. In vivo kinetics of kinase domain mutations in CML patients treated with dasatinib after failing imatinib. Blood 2008; 111: 2378–2381.
Müller MC, Cortes JE, Kim DW, Druker BJ, Erben P, Pasquini R et al. Dasatinib treatment of chronic-phase chronic myeloid leukemia: analysis of responses according to preexisting BCR-ABL mutations. Blood 2009; 114: 4944–4953.
Shah NP, Skaggs BJ, Branford S, Hughes TP, Nicoll JM, Paquette RL et al. Sequential ABL kinase inhibitor therapy selects for compound drug-resistant BCR-ABL mutations with altered oncogenic potency. J Clin Invest 2007; 117: 2562–2569.
Soverini S, Gnani A, Colarossi S, Castagnetti F, Abruzzese E, Paolini S et al. Philadelphia-positive patients who already harbor imatinib-resistant Bcr-Abl kinase domain mutations have a higher likelihood of developing additional mutations associated with resistance to second- or third-line tyrosine kinase inhibitors. Blood 2009; 114: 2168–2171.
Gambacorti-Passerini C, Khoury HJ, Pinczowski H, Masszi T, Kim DW, Zanichelli M et al. Clinical activity of bosutinib by mutational status in patients with previously treated Philadelphia chromosome-positive leukemias. Blood 2010; 116 (Suppl) abstract 3434.
Bradeen HA, Eide CA, O’Hare T, Johnson KJ, Willis SG, Lee FY et al. Comparison of imatinib mesylate, dasatinib (BMS-354825), and nilotinib (AMN107) in an N-ethyl-N-nitrosourea (ENU)-based mutagenesis screen: high efficacy of drug combinations. Blood 2006; 108: 2332–2338.
von Bubnoff N, Manley PW, Mestan J, Sanger J, Peschel C, Duyster J . Bcr-Abl resistance screening predicts a limited spectrum of point mutations to be associated with clinical resistance to the Abl kinase inhibitor nilotinib (AMN107). Blood 2006; 108: 1328–1333.
Brave M, Goodman V, Kaminskas E, Farrell A, Timmer W, Pope S et al. Sprycel for chronic myeloid leukemia and Philadelphia chromosome-positive acute lymphoblastic leukemia resistant to or intolerant of imatinib mesylate. Clin Cancer Res 2008; 14: 352–359.
Hazarika M, Jiang X, Liu Q, Lee SL, Ramchandani R, Garnett C et al. Tasigna for chronic and accelerated phase Philadelphia chromosome—positive chronic myelogenous leukemia resistant to or intolerant of imatinib. Clin Cancer Res 2008; 14: 5325–5331.
Kantarjian H, Sawyers C, Hochhaus A, Guilhot F, Schiffer C, Gambacorti-Passerini C et al. Hematologic and cytogenetic responses to imatinib mesylate in chronic myelogenous leukemia. N Engl J Med 2002; 346: 645–652.
O’Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 2003; 348: 994–1004.
Kantarjian HM, Cortes JE, O’Brien S, Giles F, Garcia-Manero G, Faderl S et al. Imatinib mesylated therapy in newly diagnosed patients with Philadelphia chromosome-positive chronic myelogenous leukemia: high incidence of early complete and major cytogenetic responses. Blood 2003; 101: 97–100.
Palandri F, Iacobucci I, Quarantelli F, Castagnetti F, Cilloni D, Baccarani M . Long-term molecular responses to imatinib in patients with chronic myeloid leukemia: comparison between complete cytogenetic responders treated in early and in late chronic phase. Haematologica 2007; 92: 1579–1580.
Calabretta B, Perrotti D . The biology of CML blast crisis. Blood 2004; 103: 4010–4022.
Hehlmann R, Saussele S . Treatment of chronic myeloid leukemia in blast crisis. Haematologica 2008; 93: 1765–1769.
Cortes J, O’Dwyer ME . Clonal evolution in chronic myelogenous leukemia. Hematol Oncol Clin North Am 2004; 18: 671–684.
Goldman JM, Melo JV . Targeting the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001; 344: 1084–1086.
Soverini S, Martinelli G, Rosti G, Bassi S, Amabile M, Poerio A et al. ABL mutations in late chronic phase chronic myeloid leukemia patients with up-front cytogenetic resistance to imatinib are associated with a greater likelihood of progression to blast crisis and shorter survival: a study by the GIMEMA Working Party on Chronic Myeloid Leukemia. J Clin Oncol 2005; 23: 4100–4109.
White D, Saunders V, Grigg A, Arthur C, Filshie R, Leahy MF et al. Measurement of in vivo BCR-ABL kinase inhibition to monitor imatinib-induced target blockade and predict response in chronic myeloid leukemia. J Clin Oncol 2007; 25: 4445–4451.
Picard S, Titier K, Etienne G, Teilhet E, Ducint D, Bernard MA et al. Trough imatinib plasma levels are associated with both cytogenetic and molecular responses to standard-dose imatinib in chronic myeloid leukemia. Blood 2007; 109: 3496–3499.
Larson RA, Druker BJ, Guilhot F, O’Brien SG, Riviere GJ, Krahnke T et al. Imatinib pharmacokinetics and its correlation with response and safety in chronic phase chronic myeloid leukemia: a subanalysis of the IRIS study. Blood 2008; 111: 4022–4028.
Marin D, Bazeos A, Mahon FX, Eliasson L, Milojkovic D, Bua M et al. Adherence is the critical factor for achieving molecular responses in patients with chronic myeloid leukemia who achieve complete cytogenetic responses on imatinib. J Clin Oncol 2010; 28: 2381–2388.
Noens L, van Lierde MA, De BR, Verhoef G, Zachee P, Berneman Z et al. Prevalence, determinants, and outcomes of nonadherence to imatinib therapy in patients with chronic myeloid leukemia: the ADAGIO study. Blood 2009; 113: 5401–5411.
Cortes JE, Baccarani M, Guilhot F, Druker BJ, Branford S, Kim DW et al. Phase III, randomized, open-label study of daily imatinib mesylate 400 mg versus 800 mg in patients with newly diagnosed, previously untreated chronic myeloid leukemia in chronic phase using molecular end points: tyrosine kinase inhibitor optimization and selectivity study. J Clin Oncol 2010; 28: 424–430.
Hughes TP, Kaeda J, Branford S, Rudzki Z, Hochhaus A, Hensley ML et al. Frequency of major molecular responses to imatinib or interferon alfa plus cytarabine in newly diagnosed chronic myeloid leukemia. N Engl J Med 2003; 349: 1423–1432.
Preudhomme C, Guilhot J, Nicolini FE, Guerci-Bresler A, Rigal-Huguet F, Maloisel F et al. Imatinib plus peginterferon alfa-2a in chronic myeloid leukemia. N Engl J Med 2010; 363: 2511–2521.
Hehlmann R, Lauseker M, Jung-Munkwitz S, Leitner A, Muller MC, Pletsch N et al. Tolerability-adapted imatinib 800 mg/d versus 400 mg/d versus 400 mg/d plus interferon-{alpha} in newly diagnosed chronic myeloid leukemia. J Clin Oncol 2011; 29: 1634–1642.
Baccarani M, Guilhot F, Larson RA, O’Brien SG, Druker BJ, on behalf of the IRIS Study Group. Outcomes by cytogenetic and molecular response at 12 and 18 months of imatinib in patients with newly diagnosed chronic myeloid leukemia (CML) in chronic phase (CP) in the IRIS trial. Blood 2006; 108 (Suppl): 606a (abstract 2138).
Marin D, Milojkovic D, Olavarria E, Khorashad JS, de Lavallade H, Reid AG et al. European LeukemiaNet criteria for failure or suboptimal response reliably identify patients with CML in early chronic phase treated with imatinib whose eventual outcome is poor. Blood 2008; 112: 4437–4444.
Khorashad JS, de LH, Apperley JF, Milojkovic D, Reid AG, Bua M et al. Finding of kinase domain mutations in patients with chronic phase chronic myeloid leukemia responding to imatinib may identify those at high risk of disease progression. J Clin Oncol 2008; 26: 4806–4813.
Soverini S, Colarossi S, Gnani A, Rosti G, Castagnetti F, Poerio A et al. Contribution of ABL kinase domain mutations to imatinib resistance in different subsets of Philadelphia-positive patients: by the GIMEMA Working Party on Chronic Myeloid Leukemia. Clin Cancer Res 2006; 12: 7374–7379.
White DL, Saunders VA, Dang P, Engler J, Venables A, Zrim S et al. Most CML patients who have a suboptimal response to imatinib have low OCT-1 activity: higher doses of imatinib may overcome the negative impact of low OCT-1 activity. Blood 2007; 110: 4064–4072.
White DL, Dang P, Engler J, Frede A, Zrim S, Osborn M et al. Functional activity of the OCT-1 protein Is predictive of long-term outcome in patients with chronic-phase chronic myeloid leukemia treated with imatinib. J Clin Oncol 2010; 28: 2761–2767.
Hiwase DK, Saunders V, Hewett D, Frede A, Zrim S, Dang P et al. Dasatinib cellular uptake and efflux in chronic myeloid leukemia cells: therapeutic implications. Clin Cancer Res 2008; 14: 3881–3888.
White DL, Saunders VA, Dang P, Engler J, Zannettino AC, Cambareri AC et al. OCT-1 mediated influx is a key determinant of the intracellular uptake of imatinib but not nilotinib (AMN107); reduced OCT-1 activity is the cause of low in vitro sensitivity to imatinib. Blood 2006; 108: 697–704.
Galimberti S, Cervetti G, Guerrini F, Testi R, Pacini S, Fazzi R et al. Quantitative molecular monitoring of BCR-ABL and MDR1 transcripts in patients with chronic myeloid leukemia during Imatinib treatment. Cancer Genet Cytogenet 2005; 162: 57–62.
Giannoudis A, Davies A, Lucas CM, Harris RJ, Pirmohamed M, Clark RE . Effective dasatinib uptake may occur without human organic cation transporter 1 (hOCT1): implications for the treatment of imatinib-resistant chronic myeloid leukemia. Blood 2008; 112: 3348–3354.
Davies A, Jordanides NE, Giannoudis A, Lucas CM, Hatziieremia S, Harris RJ et al. Nilotinib concentration in cell lines and primary CD34(+) chronic myeloid leukemia cells is not mediated by active uptake or efflux by major drug transporters. Leukemia 2009; 23: 1999–2006.
Mahon FX, Hayette S, Lagarde V, Belloc F, Turcq B, Nicolini F et al. Evidence that resistance to nilotinib may be due to BCR-ABL, Pgp, or Src kinase overexpression. Cancer Res 2008; 68: 9809–9816.
Brendel C, Scharenberg C, Dohse M, Robey RW, Bates SE, Shukla S et al. Imatinib mesylate and nilotinib (AMN107) exhibit high-affinity interaction with ABCG2 on primitive hematopoietic stem cells. Leukemia 2007; 21: 1267–1275.
Shukla S, Skoumbourdis AP, Walsh MJ, Hartz AM, Fung KL, Wu CP et al. Synthesis and characterization of a BODIPY Conjugate of the BCR-ABL kinase inhibitor Tasigna (nilotinib): evidence for transport of Tasigna and its fluorescent derivative by ABC drug transporters. Mol Pharm 2011; 8: 1292–1302.
Rosti G, Palandri F, Castagnetti F, Breccia M, Levato L, Gugliotta G et al. Nilotinib for the frontline treatment of Ph(+) chronic myeloid leukemia. Blood 2009; 114: 4933–4938.
Cortes JE, Jones D, O’Brien S, Jabbour E, Ravandi F, Koller C et al. Results of dasatinib therapy in patients with early chronic-phase chronic myeloid leukemia. J Clin Oncol 2010; 28: 398–404.
Cortes JE, Jones D, O’Brien S, Jabbour E, Konopleva M, Ferrajoli A et al. Nilotinib as front-line treatment for patients with chronic myeloid leukemia in early chronic phase. J Clin Oncol 2010; 28: 392–397.
Saglio G, Kim DW, Issaragrisil S, le CP, Etienne G, Lobo C et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med 2010; 362: 2251–2259.
Hughes TP, Hochhaus A, Saglio G, Kim DW, Jootar S, Coutre PD et al. ENESTnd update: continued superiority of nilotinib versus imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP). Blood 2010; 116 (Suppl) abstract 207.
Hochhaus A, Saglio G, le Coutre P, Kim DW, Etienne G, Zanichelli M et al. Superior efficacy of nilotinib compared with imatinib in newly-diagnosed patients with chronic myeloid leukemia in chronic (CML-CP): ENESTnd minimum 24-month follow-up. Haematologica 2011; 96 (Suppl 2) abstract 0484.
Saglio G, Kim DW, Issaragrisil S, le Coutre PD, Reiffers J, Lobo C et al. Nilotinib demonstrates superior efficacy compared with imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase: results from the international randomized phase III ENESTnd trial. Blood 2009; 114 (Suppl) abstract LBA1.
Kantarjian H, Shah NP, Hochhaus A, Cortes J, Shah S, Ayala M et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 2010; 362: 2260–2270.
Hochhaus A, Shah N, Cortes J, Baccarani M, Bradley-Garelik MB, Zhu C et al. Efficacy and safety of dasatinib compared with imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP): minimum 24-month follow-up from the DASISION trial. Haematologica 2011; 96 (Suppl 2) abstract 1011.
Porkka K, Baccarani M, Cortes J, Hochhaus A, Kantarijan H, Shah N et al. Pleural effusion in patients with chronic-phase chronic myeloid leukemia (CML-CP) who received first-line dasatinib in the DASISION trial: patient characteristics, management, and outcomes. Haematologica 2011; 96 (Suppl 2) abstract 0692.
Radich JP, Kopecky KJ, Kamel-Reid S, Stock W, Paietta E, Wadleigh M et al. A randomized phase II trial of dasatinib 100 mg vs imatinib 400 mg in newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP): the S0325 intergroup trial. Blood 2010; 116 (Suppl) abstract LBA6.
Kreutzman A, Juvonen V, Kairisto V, Ekblom M, Stenke L, Seggewiss R et al. Mono/oligoclonal T- and NK-cells are common in chronic myeloid leukemia patients at diagnosis and expand during dasatinib therapy. Blood 2010; 116: 772–782.
Nagata Y, Ohashi K, Fukuda S, Kamata N, Akiyama H, Sakamaki H . Clinical features of dasatinib-induced large granular lymphocytosis and pleural effusion. Int J Hematol 2010; 91: 799–807.
Mustjoki S, Ekblom M, Arstila TP, Dybedal I, Epling-Burnette PK, Guilhot F et al. Clonal expansion of T/NK-cells during tyrosine kinase inhibitor dasatinib therapy. Leukemia 2009; 23: 1398–1405.
Kim DH, Kamel-Reid S, Chang H, Sutherland R, Jung CW, Kim HJ et al. Natural killer or natural killer/T cell lineage large granular lymphocytosis associated with dasatinib therapy for Philadelphia chromosome positive leukemia. Haematologica 2009; 94: 135–139.
Schiffer CA, Cortes JE, Saglio G, le Coutre P, Bahceci E, Dejardin D et al. Association of lymphocytosis following treatment with dasatinib with response and outcome. J Clin Oncol 2010; 28 (15s) abstract 6553.
Schiffer CA, Cortes JE, Saglio G, le Coutre P, Guilhot F, Bahceci E et al. Lymphocytosis following first-line treatment for CML in chronic phase with dasatinib is associated with improved responses: a comparison with imatinib. Blood 2010; 116 (Suppl) abstract 358.
Gambacorti-Passerini C, Kim DW, Kantarjian HM, Brummendorf TH, Dyagil I, Griskevicius L et al. An ongoing phase 3 study of bosutinib (SKI-606) versus imatinib in patients with newly diagnosed chronic phase chronic myeloid leukemia. Blood 2010; 116 (Suppl) abstract 208.
Simonsson B, Porkka K, Richter J . Second-generation BCR-ABL kinase inhibitors in CML. N Engl J Med 2010; 363: 1673–1675.
Akard LP . Second-generation BCR-ABL kinase inhibitors in CML. N Engl J Med 2010; 363: 1672–1673; author reply 1673–1675.
Giles FJ, Abruzzese E, Rosti G, Kim DW, Bhatia R, Bosly A et al. Nilotinib is active in chronic and accelerated phase chronic myeloid leukemia following failure of imatinib and dasatinib therapy. Leukemia 2010; 24: 1299–1301.
Quintas-Cardama A, Kantarjian H, Jones D, Nicaise C, O’Brien S, Giles F et al. Dasatinib (BMS-354825) is active in Philadelphia chromosome-positive chronic myelogenous leukemia after imatinib and nilotinib (AMN107) therapy failure. Blood 2007; 109: 497–499.
Baranska M, Lewandowski K, Gniot M, Iwola M, Lewandowska M, Komarnicki M . Dasatinib treatment can overcome imatinib and nilotinib resistance in CML patient carrying F359I mutation of BCR-ABL oncogene. J Appl Genet 2008; 49: 201–203.
Kim D, Kim DW, Cho BS, Goh HG, Kim SH, Kim WS et al. Structural modeling of V299L and E459K Bcr-Abl mutation, and sequential therapy of tyrosine kinase inhibitors for the compound mutations. Leuk Res 2009; 33: 1260–1265.
Paquette RL, Shah NP, Sawyers CL, Martinelli G, John N, Chalukya M et al. PHA-739358, an aurora kinase inhibitor, induces clinical responses in chronic myeloid leukemia harboring T315I mutations of BCR-ABL. Blood 2007; 110 (Suppl) abstract 1030.
Shah P, Asatiani E, Cortes J, Paquette RL, Pinilla-Ibarz J, Kasap C et al. Interim results from a phase I clinical trial of the BCR-ABL inhibitor XL228 in drug-resistant Ph+ leukemias. Haematologica 2008; 93 (Suppl 1): 47–48 (abstract 120).
Gontarewicz A, Balabanov S, Keller G, Colombo R, Graziano A, Pesenti E et al. Simultaneous targeting of Aurora kinases and Bcr-Abl kinase by the small molecule inhibitor PHA-739358 is effective against imatinib-resistant BCR-ABL mutations including T315I. Blood 2008; 111: 4355–4364.
Gontarewicz A, Brummendorf TH . Danusertib (formerly PHA-739358)—a novel combined pan-Aurora kinases and third generation Bcr-Abl tyrosine kinase inhibitor. Recent Results Cancer Res 2010; 184: 199–214.
Cortes J, Paquette R, Talpaz M, Pinilla J, Asatiani E, Wetzler M et al. Preliminary clinical activity in a phase I trial of the BCR-ABL/IGF-1R/aurora kinase inhibitor XL228 in patients with Ph+ leukemias with either failure to multiple TKI therapies or with T315I mutation. Blood 2008; 112 (Suppl) abstract 3232.
Rivera VM, Xu Q, Wang F, Snodgrass J, O’Hare T, Corbin AS et al. Potent antitumor activity of AP24534, an orally active inhibitor of Bcr-Abl variants including T315I, in in vitro and in vivo models of chronic myeloid leukemia (CML). Blood 2007; 110 (Suppl) abstract 1032.
O’hare T, Shakespeare WC, Zhu X, Eide CA, Rivera VM, Wang F et al. AP24534, a pan-BCR-ABL inhibitor for chronic myeloid leukemia, potently inhibits the T315I mutant and overcomes mutation-based resistance. Cancer Cell 2009; 16: 401–412.
Cortes J, Talpaz M, Deininger M, Shah N, Flinn IW, Mauro MJ et al. A phase 1 trial of oral AP24534 in patients with refractory chronic myeloid leukemia and other hematologic malignancies: first results of safety and clinical activity against T315I and resistant mutations. Blood 2009; 114 (Suppl): 267 (abstract 643).
Van Etten RA, Chan WW, Zaleskas VM, Evangelista P, Lazarides K, Peng C et al. DCC-2036: a novel switch pocket inhibitor of ABL tyrosine kinase with therapeutic efficacy against BCR-ABL T315I in vitro and in a CML mouse model. Blood 2007; 110 (Suppl) abstract 463.
Van Etten RA, Chan WW, Zaleskas VM, Walz C, Evangelista P, Lazarides K et al. Switch pocket inhibitors of the ABL tyrosine kinase: distinct kinome inhibition profiles and in vivo efficacy in mouse models of CML and B-lymphoblastic leukemia induced by BCR-ABL T315I. Blood 2008; 112 (Suppl) abstract 576.
Baccarani M, Druker BJ, Cortes-Franco J, Hughes TP, Kim DW, Pane F et al. 24 months update of the TOPS study: a phase III, randomized, open-label study of 400 mg/d versus 800 mg/d of imatinib mesylate in patients with newly diagnosed, previously untreated chronic myeloid leukemia in chronic phase. Blood 2009; 114 (Suppl): 142–143 (abstract 337).
Baccarani M, Rosti G, Castagnetti F, Haznedaroglu I, Porkka K, Abruzzese E et al. Comparison of imatinib 400 mg and 800 mg daily in the front-line treatment of high-risk, Philadelphia-positive chronic myeloid leukemia: a European LeukemiaNet Study. Blood 2009; 113: 4497–4504.
McWeeney SK, Pemberton LC, Loriaux MM, Vartanian K, Willis SG, Yochum G et al. A gene expression signature of CD34+ cells to predict major cytogenetic response in chronic-phase chronic myeloid leukemia patients treated with imatinib. Blood 2010; 115: 315–325.
Hosoya N, Sanada M, Nannya Y, Nakazaki K, Wang L, Hangaishi A et al. Genomewide screening of DNA copy number changes in chronic myelogenous leukemia with the use of high-resolution array-based comparative genomic hybridization. Genes Chromosomes Cancer 2006; 45: 482–494.
Koh Y, Kim DY, Park SH, Jung SH, Park E, Kim HJ et al. GSTT1 copy number gain is a poor predictive marker for escalated-dose imatinib treatment in chronic myeloid leukemia: genetic predictive marker found using array comparative genomic hybridization. Cancer Genet Cytogenet 2010; 203: 215–221.
Nadarajan VS, Phan CL, Ang CH, Liang KL, Gan GG, Bee PC et al. Identification of copy number alterations by array comparative genomic hybridization in patients with late chronic or accelerated phase chronic myeloid leukemia treated with imatinib mesylate. Int J Hematol 2011; 93: 465–473.
Mahon FX, Rea D, Guilhot J, Guilhot F, Huguet F, Nicolini F et al. Discontinuation of imatinib in patients with chronic myeloid leukaemia who have maintained complete molecular remission for at least 2 years: the prospective, multicentre Stop Imatinib (STIM) trial. Lancet Oncol 2010; 11: 1029–1035.
Ross DM, Branford S, Seymour JF, Schwarer AP, Arthur C, Bartley PA et al. Patients with chronic myeloid leukemia who maintain a complete molecular response after stopping imatinib treatment have evidence of persistent leukemia by DNA PCR. Leukemia 2010; 24: 1719–1724.
Graham SM, Jorgensen HG, Allan E, Pearson C, Alcorn MJ, Richmond L et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 2002; 99: 319–325.
Copland M, Hamilton A, Elrick LJ, Baird JW, Allan EK, Jordanides N et al. Dasatinib (BMS-354825) targets an earlier progenitor population than imatinib in primary CML but does not eliminate the quiescent fraction. Blood 2006; 107: 4532–4539.
Jorgensen HG, Allan EK, Jordanides NE, Mountford JC, Holyoake TL . Nilotinib exerts equipotent antiproliferative effects to imatinib and does not induce apoptosis in CD34+ CML cells. Blood 2007; 109: 4016–4019.
Lemoli RM, Salvestrini V, Bianchi E, Bertolini F, Fogli M, Amabile M et al. Molecular and functional analysis of the stem cell compartment of chronic myelogenous leukemia reveals the presence of a CD34− cell population with intrinsic resistance to imatinib. Blood 2009; 114: 5191–5200.
Chen Y, Hu Y, Zhang H, Peng C, Li S . Loss of the Alox5 gene impairs leukemia stem cells and prevents chronic myeloid leukemia. Nat Genet 2009; 41: 783–792.
Naka K, Hoshii T, Hirao A . Novel therapeutic approach to eradicate tyrosine kinase inhibitor resistant chronic myeloid leukemia stem cells. Cancer Sci 2010; 101: 1577–1581.
Zhao C, Chen A, Jamieson CH, Fereshteh M, Abrahamsson A, Blum J et al. Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature 2009; 458: 776–779.
Zhao C, Blum J, Chen A, Kwon HY, Jung SH, Cook JM et al. Loss of beta-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell 2007; 12: 528–541.
Ito K, Bernardi R, Morotti A, Matsuoka S, Saglio G, Ikeda Y et al. PML targeting eradicates quiescent leukaemia-initiating cells. Nature 2008; 453: 1072–1078.
Chen Y, Hu Y, Michaels S, Segal D, Brown D, Li S . Inhibitory effects of omacetaxine on leukemic stem cells and BCR-ABL-induced chronic myeloid leukemia and acute lymphoblastic leukemia in mice. Leukemia 2009; 23: 1446–1454.
Copland M, Pellicano F, Richmond L, Allan EK, Hamilton A, Lee FY et al. BMS-214662 potently induces apoptosis of chronic myeloid leukemia stem and progenitor cells and synergizes with tyrosine kinase inhibitors. Blood 2008; 111: 2843–2853.
Zhang B, Strauss AC, Chu S, Li M, Ho Y, Shiang KD et al. Effective targeting of quiescent chronic myelogenous leukemia stem cells by histone deacetylase inhibitors in combination with imatinib mesylate. Cancer Cell 2010; 17: 427–442.
Essers MA, Offner S, Blanco-Bose WE, Waibler Z, Kalinke U, Duchosal MA et al. IFNalpha activates dormant haematopoietic stem cells in vivo. Nature 2009; 458: 904–908.
Acknowledgements
The authors have contributed significantly to this paper, take full responsibility for the content of this article and confirm that it reflects their viewpoint and medical expertise. StemScientific, funded by Bristol-Myers Squibb, provided writing and editing support. Bristol-Myers Squibb did not in any way influence the content of the paper nor did the authors receive financial compensation for authoring the article. This study was supported by NIH Grants HL082978-01 (MD), CA04963920A2 (MD), 1R01CA129611 (PJS), Leukemia and Lymphoma Society grant 7036-01 (MD). MD is a Scholar in Clinical Research of the Leukemia and Lymphoma Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M Deininger is a paid consultant for Novartis, Bristol-Myers Squibb and ARIAD. PJ Shami has participated in Advisory Boards and Speaker Bureau for Novartis.
Rights and permissions
About this article
Cite this article
Shami, P., Deininger, M. Evolving treatment strategies for patients newly diagnosed with chronic myeloid leukemia: the role of second-generation BCR–ABL inhibitors as first-line therapy. Leukemia 26, 214–224 (2012). https://doi.org/10.1038/leu.2011.217
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.217
Keywords
This article is cited by
-
Chronic myeloid leukemia stem cells and molecular target therapies for overcoming resistance and disease persistence
International Journal of Hematology (2018)
-
Assessment of bone marrow lymphocytic status during tyrosine kinase inhibitor therapy and its relation to therapy response in chronic myeloid leukaemia
Journal of Cancer Research and Clinical Oncology (2016)
-
Long-term outcome of chronic myeloid leukemia patients treated frontline with imatinib
Leukemia (2015)
-
A review of the European LeukemiaNet recommendations for the management of CML
Annals of Hematology (2015)
-
Managing chronic myeloid leukaemia in the elderly with intermittent imatinib treatment
Blood Cancer Journal (2015)