Abstract
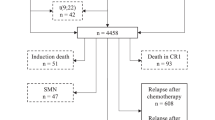
In this study, the long-term outcome of 1818 patients treated in five consecutive clinical trials (the cooperative study group for childhood acute lymphoblastic leukaemia (COALL) 82, 85, 89, 92 and 97) from 24 cooperating centres in Germany is reported. The probability of event-free survival (pEFS) improved significantly from the first two trials conducted in the 1980s (COALL 82 and COALL 85) to the three trials conducted in the 1990s (COALL 89, 92 and 97) (P=0.001). Through all COALL studies, age ⩾10 years and initial white blood cell count (WBC) ⩾50 × 109/l and pro-B immunophenotype were of significant prognostic relevance. A refinement of risk assessment has been achieved by in vitro drug sensitivity testing in COALL 92 and 97. In patients with very sensitive leukaemic cells, therapy could be reduced without loss of efficacy. In COALL 97, a further improvement in risk stratification was gained by the molecular assessment of minimal residual disease (MRD) under treatment, which proved to have a superior prognostic effect when compared with in vitro drug sensitivity testing. Importantly, the gradual reduction in central nervous system (CNS) irradiation led to a decreased incidence of brain tumours as a second malignancy. In general, the prevention of treatment-related late effects will be one of the major issues in future studies. It remains to be shown whether prolonged infusions of anthracyclines, which have been implemented into the COALL studies after equal efficacy compared with short-time infusions was confirmed, will be associated with fewer cardiac late effects. Another way to prevent late effects may be a more refined risk assessment allowing for a reduction in cumulative treatment burden. A great challenge in the future will be to improve the overall treatment results, which very likely can only be achieved by the identification of molecularly defined subgroups to which novel, rational therapeutic strategies can be applied.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Janka GE, Winkler K, Jürgens H, Göbel U, for the COALL Study Group. Early intensification therapy in high-risk childhood acute lymphocytic leukemia: lack of benefit from high-dose methotrexate. Haematol Blood Transfus 1987; 30: 456–460.
Jürgens H, Janka G, Ibrahim M, Tonert C, Winkler K, Göbel U . Prognostic significance of exposure to intermediate-dose methotrexate in children with standard risk ALL: the COALL 82/85 experience. Haematol Blood Transf 1992; 34: 338–342.
Harms DO, Janka-Schaub GE . Co-operative study group for childhood acute lymphoblastic leukemia (COALL), long-term follow up of trials 82, 85, 89 and 92. Leukemia 2000; 14: 2234–2239.
Janka-Schaub GE, Harms DO, den Boer ML, Veerman AJ, Pieters R . In vitro drug resistance as independent prognostic factor in the study COALL-O5-92 Treatment of childhood acute lymphoblastic leukemia; two-tiered classification of treatments based on accepted risk criteria and drug sensitivity profiles in study COALL-06-97. Klin Padiatr 1999; 211: 233–238.
Harms DO, Göbel U, Spaar HJ, Graubner U, Jorch N, Gutjahr P, et al., for the COALL Study Group. Thioguanine offers no advantage over mercaptopurine in maintenance treatment of childhood ALL: results of the randomized trial COALL-92. Blood 2003; 102: 2736–2740.
Escherich G, Göbel U, Jorch N, Spaar HJ, Janka-Schaub GE . Daunorubicin-induced cell kill with 1-hour versus 24-hour infusions a randomized comparison in children with newly diagnosed acute lymphoblastic leukemia. Klin Padiatr 2007; 219: 134–138.
Janka GE, Göbel U, Gutjahr P, Jürgens H, Spaar HJ, Winkler K, for the COALL Study Group. Possible adverse influence of administering intrathecal methotrexate (it-MTX) non-concurrently with cranial radiotherapy (CRT) in the treatment of childhood acute lymphocytic leukemia (ALL). Med Ped Oncol 1988a; 16: 394.
Janka GE, Winkler K, Göbel U, Graubner U, Gutjahr P, Haas RJ, et al., for the COALL Study Group. Rapidly rotating combination chemotherapy in childhood acute lymphoblastic leukemia: preliminary results of a randomized comparison with conventional treatment. Leukemia 1988b; 2: 73–78.
Janka-Schaub GE, Harms D, Göbel U, Graubner U, Gutjahr P, Haas RJ, et al., for the COALL Study Group. Randomized comparison of rotational chemotherapy in high-risk acute lymphoblastic leukaemia of childhood—follow up after 9 years. Eur J Pediatr 1996; 155: 640–648.
Kaplan EL, Meier P . Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Andersen PK, Borch-Johnsen K, Deckert T, Green A, Hougaard P, Keiding N et al. A Cox regression model for the relative mortality and its application to diabetes mellitus survival data. Biometrics 1985; 41: 921–932.
Kalbfleisch JD, Prentice RL . The Statistical Analysis of Failure Time Data, 2nd edn. Wiley Series in Probability and Statistics Wiley: New York, NY, 2002 439pp.
Graubner UB, Porzig S, Jorch N, Kolb R, Wessalowski R, Escherich G et al. Impact of reduction of therapy on infectious complications in childhood acute lymphoblastic leukemia. Ped Blood Cancer 2007; 50: 259–263.
van Dongen JJ, Seriu T, Panzer-Grumayer ER, Biondi A, Pongers-Willemse MJ, Corral L et al. Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood. Lancet 1998; 352: 1731–1738.
Conter V, Schrappe M, Arico M, Reiter A, Rizzari M, Dördelmann M et al. Role of cranial radiotherapy for childhood T-cell acute lymphoblastic leukaemia with high WBC count and good response to prednisone. J Clin Oncol 1997; 15: 2786–2791.
Vieira Pinheiro JP, Wenner K, Escherich G, Lanvers-Kaminsky C, Würthwein G, Janka-Schaub G et al. Serum asparaginase activities and asparagine concentrations in cerebrospinal fluid after a single infusion of 2.300 IU/m2 PEG-asparaginase in children with ALL treated according to protocol COALL 06-97. J Ped Blood Cancer 2006; 46: 18–25.
Kaatsch P, Reinisch I, Spix C, Berthold F, Janka-Schaub G, Mergenthaler A et al. Case control study on the therapy of childhood cancer and the occurrence of second malignant neoplasms in Germany. Cancer Causes Control 2009; 20: 965–980.
Baker KS, DeFor TE, Burns LJ, Ramsay NKC, Neglia JP, Robinson LL . New malignancies after blood or marrow stem-cell transplantation in children and adults: incidence and risk factors. J Clin Oncol 2003; 21: 1352–1358.
Löning L, Zimmermann M, Reiter A, Kaatsch P, Henze G, Riehm HJ et al. Secondary neoplasms subsequent to Berlin-Frankfurt-München therapy of acute lymphoblastic leukemia. Blood 2000; 95: 2770–2775.
Erb N, Harms D, Janka-Schaub GE . Pharmacokinetics and metabolism of thiopurines in children with acute lymphoblastic leukemia receiving 6-thioguanine versus 6-mercaptopurine. Cancer Chemother Pharmacol 1998; 42: 266–272.
Stoneham S, Lennard L, Coen P, Lilleyman J, Saha V . Veno-occlusive disease in patients receiving thiopurines during maintenance therapy for childhood acute lymphoblastic leukemia. Brit J Haematol 2003; 123: 100–102.
Broxon EH, Dole M, Wong R, Laya BF, Stork L . Portal hypertension develops in a subset of children with standard risk leukemia treated with oral 6-thioguanine during maintenance therapy. Ped Blood Cancer 2005; 44: 226–231.
Legha SS, Benjamin RS, Mackay B, Ewer M, Wallace S, Valdivieso M et al. Reduction of doxorubicin cardiotoxicity by prolonged continuous intravenous infusion. Ann Intern Med 1982; 96: 133–139.
Berrak SG, Ewer MS, Jaffe N, Pearson P, Ried H, Zietz HA et al. Doxorubicin cardiotoxicity in children: reduced incidence of cardiac dysfunction associated with continuous-infusion schedules. Oncol Rep 2001; 8: 611–614.
Pieters R, Huismans DR, Loonen AH, Hahlen K, van der Does-van den Berg A, van Wering ER et al. Relation of cellular drug resistance to long-term clinical outcome in childhood acute lymphoblastic leukaemia. Lancet 1991; 338: 399–403.
Kaspers GJ, Pieters R, Van Zantwijk CH, De Laat PA, De Waal FC, Van Wering ER et al. In vitro drug sensitivity of normal peripheral blood lymphocytes and childhood leukaemic cells from bone marrow and peripheral blood. Br J Cancer 1991; 64: 469–474.
Hongo T, Yajima S, Sakurai M, Horikoshi Y, Hanada R . In vitro drug sensitivity testing can predict induction failure and early relapse of childhood acute lymphoblastic leukemia. Blood 1997; 89: 2959–2965.
Frost BM, Nygren P, Gustafsson G, Forestier E, Jonsson OG, Kanerva J et al. Increased in vitro cellular drug resistance is related to poor outcome in high-risk childhood acute lymphoblastic leukaemia. Br J Haematol 2003; 122: 376–385.
Styczynski J, Wysocki M . Is the in vitro drug resistance profile the strongest prognostic factor in childhood acute lymphoblastic leukemia? J Clin Oncol 2004; 22: 963–964.
Den Boer ML, Harms DO, Pieters R, Kazemier KM, Gobel U, Korholz D et al. Patient stratification based on prednisolone-vincristine-asparaginase resistance profiles in children with acute lymphoblastic leukemia. J Clin Oncol 2003; 21: 3262–3268.
Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukaemia. European Organization for research and treatment of Cancer—Childhood Leukemia Cooperative Group. N Engl J Med 1998; 339: 591–598.
Flohr T, Schrauder A, Cazzaniga G, Panzer-Grümayer R, van der Velden V, Fischer S, et al., International BFM Study Group (I-BFM-SG). Minimal residual disease-directed risk stratification using real-time quantitative PCR analysis of immunoglobulin and T-cell receptor gene rearrangements in the international multicenter trial AIEOP-BFM ALL 2000 for childhood acute lymphoblastic leukemia. Leukemia 2008; 22: 771–782.
Acknowledgements
We are grateful to the Fördergemeinschaft Kinderkrebs-Zentrum Hamburg for financial support. We also thank all individuals who collaborated in the designated COALL trials in the past: Dr Pongratz (Augsburg), J Otte, N Jorch (Bielefeld), HJ Spaar, T Lieber (Bremen), Dr Siegert (Dresden), U Göbel, G Janssen (Düsseldorf), JF Beck, S Weigel (Greifswald), Dr Streitberger (Heide), W Nürnberger (Idar-Oberstein), C von Klinggräff, K Westerbeck (Kiel), P Thomas, S Völpel (Krefeld), G Weißbach, U Bierbach (Leipzig), P Gutjahr (Mainz), I Althaus (Mönchengladbach), R Roos, P Klose (München-Harlaching), U Graubner, I Schmidt, Dr Haas (München, LMU), Dr Drescher, H Müller, R Kolb (Oldenburg), J Wolff, O Peters (Regensburg), J Weber (Wiesbaden) and Dr Dohrn (Wuppertal). Finally, we are indebted to M den Boer, KM Kazemir, R Pieters (Rotterdam), GJ Kaspers (Amsterdam), U zur Stadt, D Grabowski, K Michael, S Bartel and A Clodius (Hamburg) for intellectual and technical support.
Author information
Authors and Affiliations
Consortia
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Supplementary information
Rights and permissions
About this article
Cite this article
Escherich, G., Horstmann, M., Zimmermann, M. et al. Cooperative study group for childhood acute lymphoblastic leukaemia (COALL): long-term results of trials 82,85,89,92 and 97. Leukemia 24, 298–308 (2010). https://doi.org/10.1038/leu.2009.249
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2009.249
Keywords
This article is cited by
-
Risk-adjusted therapy for pediatric non-T cell ALL improves outcomes for standard risk patients: results of JACLS ALL-02
Blood Cancer Journal (2020)
-
Results of two consecutive treatment protocols in Polish children with acute lymphoblastic leukemia
Scientific Reports (2020)
-
MRD response in a refractory paediatric T-ALL patient through anti-programmed cell death 1 (PD-1) Ab treatment associated with induction of fatal GvHD
Bone Marrow Transplantation (2017)
-
Methotrexate resistance in relation to treatment outcome in childhood acute lymphoblastic leukemia
Journal of Hematology & Oncology (2015)
-
CD44 and CD27 expression pattern in B cell precursor acute lymphoblastic leukemia and its clinical significance
Medical Oncology (2013)